Professional Documents
Culture Documents
Table of Thermo Relations
Uploaded by
billy435Original Description:
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Table of Thermo Relations
Uploaded by
billy435Copyright:
Available Formats
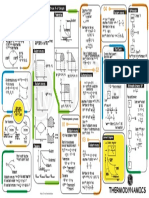
Thermodynamic Relations
∂S ∂U ∂H ∂A ∂G
∂T
p
Cp
T
C p− p ∂V
∂T p
Cp −S − p
∂V
∂T p
−S
∂T
v
CV
T
CV
C V V
∂p
∂T V
−S −S V ∂p
∂T V
∂p
T
− ∂V
∂T p
−T
∂V
∂T p
−p
∂V
∂p T
−T ∂V
∂T p
V −p
∂V
∂p T
V
∂p
V
CV ∂T
T ∂p V
CV
∂T
∂p V
CV ∂T
∂p V
V −S
∂T
∂p V
−S
∂T
∂p V
V
∂V
T
∂p
∂T V
T ∂p
∂T V
−p T
∂p
∂T V
V
∂p
∂V T
−p V
∂p
∂V T
∂V p
C p ∂T
T ∂V p
Cp ∂T
∂V p
−p Cp ∂T
∂V p
−S
∂T
∂V p
−p −S
∂T
∂V p
How to use this table.
The top row gives the variable in the top of the partial derivative. For example, to find ∂H
∂p V
, go to the column with ∂ H in the top
and down to the row with
∂p V
. You quickly see that
∂H
∂p V
=C V
∂T
∂p V
V . Notice that some relations are much simpler than
others, which is why we use certain thermodynamic potentials for certain independent variables.
Additional Relations
=
1 ∂V
V ∂T p
=
−1 ∂V
V ∂p T
C p −C V =
2T V
∂ G /T
∂T
p
−H
= 2
T
You might also like
- Replacement lights upgrade Elantra '18Document86 pagesReplacement lights upgrade Elantra '18Ahmed RagabNo ratings yet
- Lessons Learned - Risk Management Issues in Genetic Counseling (2007)Document151 pagesLessons Learned - Risk Management Issues in Genetic Counseling (2007)AditiNo ratings yet
- DEWA Electrical Installation Regulations Section 1 OverviewDocument123 pagesDEWA Electrical Installation Regulations Section 1 Overviewsiva_nagesh_280% (5)
- MechanismDocument17 pagesMechanismm_er100No ratings yet
- Oracle Time and Labor - Data SheetDocument5 pagesOracle Time and Labor - Data Sheetbilaltanoli@gmail.comNo ratings yet
- Peptan - All About Collagen Booklet-1Document10 pagesPeptan - All About Collagen Booklet-1Danu AhmadNo ratings yet
- CHECK LIST FOR HIGH RISE BUILDING NOCDocument15 pagesCHECK LIST FOR HIGH RISE BUILDING NOCNedunuri.Madhav Murthy100% (2)
- Zrínyi 2019 4.o Megyei FDocument2 pagesZrínyi 2019 4.o Megyei FFruzsi KNo ratings yet
- Rules & Guidelines of Elliott WaveDocument12 pagesRules & Guidelines of Elliott WaveNd Reyes100% (2)
- Austin's Cover Letter Example - Cultivated CultureDocument1 pageAustin's Cover Letter Example - Cultivated CultureYash SNo ratings yet
- Analog To Digital Conversion (ADC)Document62 pagesAnalog To Digital Conversion (ADC)Asin PillaiNo ratings yet
- Epidemiological Cutoff Values For Antifungal Susceptibility TestingDocument36 pagesEpidemiological Cutoff Values For Antifungal Susceptibility Testingdadrrui100% (1)
- 2018 Cambridge Lower Second Progression Test Science Stage 8 QP Paper 2 - tcm143-430409Document16 pages2018 Cambridge Lower Second Progression Test Science Stage 8 QP Paper 2 - tcm143-430409AnisahNo ratings yet
- Jawaban Soal Persamaan Fundamental-28Document2 pagesJawaban Soal Persamaan Fundamental-28NurNo ratings yet
- The First Law of Thermodynamics (Continued) (Lecture 03) : - Prashant Uday ManoharDocument27 pagesThe First Law of Thermodynamics (Continued) (Lecture 03) : - Prashant Uday ManoharHRIDAY MAHESHWARINo ratings yet
- Relaciones Termodinámicas TablasDocument2 pagesRelaciones Termodinámicas TablasEstefanía RomeroNo ratings yet
- ESE-2018 Mains Test Series: Mechanical Engineering Test No: 2Document33 pagesESE-2018 Mains Test Series: Mechanical Engineering Test No: 2Er Kapil KashyapNo ratings yet
- Gibbs Free Energy and Chemical PotentialDocument73 pagesGibbs Free Energy and Chemical PotentialSHER AfghanNo ratings yet
- CHM 373 Solutions To UPC Chapter #3: July 3, 2013Document7 pagesCHM 373 Solutions To UPC Chapter #3: July 3, 2013Janmejay GoenkaNo ratings yet
- Procesos TermodinamicosDocument1 pageProcesos Termodinamicossifsalma09No ratings yet
- Thermodynamics Properties of FluidsDocument70 pagesThermodynamics Properties of FluidsCesar BravoNo ratings yet
- Watchtower-Control and GT PTBDocument15 pagesWatchtower-Control and GT PTBGustava LafavaNo ratings yet
- Problem Set 7Document2 pagesProblem Set 7Ykhay ElfanteNo ratings yet
- Formulario ExamenDocument2 pagesFormulario ExamenMohamed ErraquiouiNo ratings yet
- Pauta Control 1-S03Document3 pagesPauta Control 1-S03NachoNo ratings yet
- Introduction To The Equilibrium PhaseDocument8 pagesIntroduction To The Equilibrium PhaseJksgNo ratings yet
- DinamicaDocument2 pagesDinamicaAlan FloresNo ratings yet
- Chem 201 Lecture NotesDocument2 pagesChem 201 Lecture NotesProtect btsrvNo ratings yet
- Constraint MotionDocument5 pagesConstraint MotionnithinjothimuruganNo ratings yet
- Compressibility PDFDocument2 pagesCompressibility PDFJuan Daniel CabreraNo ratings yet
- 5.60 Thermodynamics & Kinetics: Mit OpencoursewareDocument5 pages5.60 Thermodynamics & Kinetics: Mit OpencoursewarecaptainhassNo ratings yet
- Short title for document on LC1 LS/flDocument1 pageShort title for document on LC1 LS/flBatsha PonnusamyNo ratings yet
- Formulario Termodinamica Equaciones BasicasDocument1 pageFormulario Termodinamica Equaciones BasicasDivad_28No ratings yet
- Rientra Duct FlowAirS2012slidesDocument45 pagesRientra Duct FlowAirS2012slideso_mars_2010No ratings yet
- 0.1 Derivation of Unsteady Bernoulli's Equation: 2.016 HydrodynamicsDocument1 page0.1 Derivation of Unsteady Bernoulli's Equation: 2.016 HydrodynamicsIhab OmarNo ratings yet
- HW 1Document3 pagesHW 1kako3163No ratings yet
- Rumus FisikaDocument6 pagesRumus FisikaEvita CarolineNo ratings yet
- GLBB Di Perce Pat Berubah Berat Bergerak Secara GravitasiDocument5 pagesGLBB Di Perce Pat Berubah Berat Bergerak Secara GravitasiSalsabila Putri AndriantiNo ratings yet
- Clones 5m1Document6 pagesClones 5m1keman91No ratings yet
- Du Tds PDV DH Tds VDP Da SDT PDV DG SDT VDP: Chapter 11 SummaryDocument4 pagesDu Tds PDV DH Tds VDP Da SDT PDV DG SDT VDP: Chapter 11 SummaryGitanjali TomarNo ratings yet
- Equations of State ExplainedDocument11 pagesEquations of State ExplainedMehabaw FikrieNo ratings yet
- Get Lost NerdDocument2 pagesGet Lost NerdSathvikaNo ratings yet
- Ejercicios Entrega Tema 5Document4 pagesEjercicios Entrega Tema 5Andrea Garcia EstellesNo ratings yet
- ˆ θ = Deg = = RR sin θdθdϕ: x ˆ y−yˆ x r A R (הפילקה לע הרידגמ תיווזהש חטשה A)Document4 pagesˆ θ = Deg = = RR sin θdθdϕ: x ˆ y−yˆ x r A R (הפילקה לע הרידגמ תיווזהש חטשה A)Naama GloorNo ratings yet
- l6 ThermoDocument68 pagesl6 ThermoJahnavi RameshNo ratings yet
- Statistical Molecular Thermodynamics: Christopher J. CramerDocument7 pagesStatistical Molecular Thermodynamics: Christopher J. CramerLuciano Sánchez AramburuNo ratings yet
- Electrical MeasurementDocument100 pagesElectrical MeasurementAsif RazaNo ratings yet
- TP 07Document1 pageTP 07iago villaNo ratings yet
- Sports-Improve Your EnglishDocument6 pagesSports-Improve Your EnglishGwen GalaNo ratings yet
- FA Az: E Eil ZNDocument2 pagesFA Az: E Eil ZNIsham IbrahimNo ratings yet
- Thermodynamics: Mind MapsDocument1 pageThermodynamics: Mind MapsRiya BhardwajNo ratings yet
- Juara UmumDocument1 pageJuara UmumKerin BerlianaNo ratings yet
- The Seven Generals of ThermodynamicsDocument1 pageThe Seven Generals of ThermodynamicsAnthony Macalindong100% (2)
- PR Co 0001 enDocument6 pagesPR Co 0001 enNikola VeleskiNo ratings yet
- CH 4Document26 pagesCH 4林孟群No ratings yet
- NOTAS PARA PARCIAL3 FísicaDocument4 pagesNOTAS PARA PARCIAL3 FísicaJuan David Caicedo BarrantesNo ratings yet
- Linear Motion GraphsDocument2 pagesLinear Motion GraphsNajwa Huda ShaharilNo ratings yet
- Suvat EquationsDocument1 pageSuvat EquationshosannaNo ratings yet
- StreamlinedDocument1 pageStreamlinedkinki_sammixNo ratings yet
- Ice 2Document2 pagesIce 2Nuttapat TeerakNo ratings yet
- PHYSchartDocument2 pagesPHYSchartSkander HamadiNo ratings yet
- Maximizing SEO Value in Short TitlesDocument1 pageMaximizing SEO Value in Short Titlesovidiu2014No ratings yet
- Solfeo 1 - Tenor SaxDocument1 pageSolfeo 1 - Tenor SaxSoniaVanesaMahechaTriviñoNo ratings yet
- Selena Scott RotatedDocument4 pagesSelena Scott RotatedJeremy TurnageNo ratings yet
- Derivation of Root Means Square (RMS Voltage)Document1 pageDerivation of Root Means Square (RMS Voltage)Wilson (Electrical Engineer)No ratings yet
- Ap Academy 3Document3 pagesAp Academy 3José Luis Bolívar AguilarNo ratings yet
- Nat-Test List Moji Goi BunpouDocument19 pagesNat-Test List Moji Goi BunpouTaufikurahman MaulanaNo ratings yet
- Jurnal Harian PBL RS HajiDocument4 pagesJurnal Harian PBL RS Hajirsuhajisurabaya diklatNo ratings yet
- U U U U Shear Viscosity Bulk Viscosity P Pressure Mass Density C K Thermal ConductivityDocument9 pagesU U U U Shear Viscosity Bulk Viscosity P Pressure Mass Density C K Thermal ConductivityNe Vem Quiche ShakiraNo ratings yet
- Commissioner of Customs V Hypermix Feeds Corporation: IS S U E F A C T S C M O 2 7 - 2 0 0 3Document1 pageCommissioner of Customs V Hypermix Feeds Corporation: IS S U E F A C T S C M O 2 7 - 2 0 0 3Reyshanne Joy B MarquezNo ratings yet
- Image Formation in Plane Mirrors: Ray DiagramsDocument3 pagesImage Formation in Plane Mirrors: Ray DiagramsSouvik BanerjeeNo ratings yet
- Stress-Busting Plan for Life's ChallengesDocument3 pagesStress-Busting Plan for Life's Challengesliera sicadNo ratings yet
- NotesTransl 108 (1985) Larsen, Who Is This GenerationDocument20 pagesNotesTransl 108 (1985) Larsen, Who Is This GenerationluzuNo ratings yet
- trac-nghiem-ngu-am-am-vi-hoc-tieng-anh-đã chuyển đổiDocument18 pagestrac-nghiem-ngu-am-am-vi-hoc-tieng-anh-đã chuyển đổiNguyễn ThiênNo ratings yet
- Rustia V Cfi BatangasDocument2 pagesRustia V Cfi BatangasAllen GrajoNo ratings yet
- (Salim Ross) PUA 524 - Introduction To Law and The Legal System (Mid Term)Document4 pages(Salim Ross) PUA 524 - Introduction To Law and The Legal System (Mid Term)Salim RossNo ratings yet
- Thermal Physics KPN MurthyDocument151 pagesThermal Physics KPN MurthyRithish BarathNo ratings yet
- Manage a micro business with BSB30315Document3 pagesManage a micro business with BSB30315Theo A W JacksonNo ratings yet
- Advisory Circular: Aircraft Maintenance Engineer Licence - Examination Subject 2 Aircraft Engineering KnowledgeDocument44 pagesAdvisory Circular: Aircraft Maintenance Engineer Licence - Examination Subject 2 Aircraft Engineering KnowledgejashkahhNo ratings yet
- CANAVAN' and VESCOVI - 2004 - CMJ X SJ Evaluation of Power Prediction Equations Peak Vertical Jumping Power in WomenDocument6 pagesCANAVAN' and VESCOVI - 2004 - CMJ X SJ Evaluation of Power Prediction Equations Peak Vertical Jumping Power in WomenIsmenia HelenaNo ratings yet
- Ivf Market in IndiaDocument15 pagesIvf Market in IndiaSunil Tak100% (1)
- Fixed Prosthodontics Provisional Materials: Making The Right Selection.Document7 pagesFixed Prosthodontics Provisional Materials: Making The Right Selection.veloso.rossana0% (1)
- Advantages of Computerizing and Networking ComputersDocument5 pagesAdvantages of Computerizing and Networking ComputersIn BioNo ratings yet
- APA - 7thed - UCW Student Paper TemplateDocument4 pagesAPA - 7thed - UCW Student Paper Templatesimerpreet3No ratings yet
- Health Benefits of Kidney BeansDocument17 pagesHealth Benefits of Kidney BeansShyneAneeshNo ratings yet
- ICS Technical College Prospectus 2024 Edition 1Document36 pagesICS Technical College Prospectus 2024 Edition 1samuel287kalumeNo ratings yet
- Radial Drill Catalog-110620Document14 pagesRadial Drill Catalog-110620Anto SiminNo ratings yet
- ZO 503 Physiological Chemistry by Dr.S.S.KunjwalDocument22 pagesZO 503 Physiological Chemistry by Dr.S.S.KunjwalAbhishek Singh ChandelNo ratings yet
- KOREADocument124 pagesKOREAchilla himmudNo ratings yet