Professional Documents
Culture Documents
Composition of The Human Body
Uploaded by
raschensOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Composition of The Human Body
Uploaded by
raschensCopyright:
Available Formats
Composition of the human body
Composition of the human body
The composition of the human body can be looked at from the point of view of either mass composition, or atomic composition. To illustrate both views, the human body is ~70% water, and water is ~11% hydrogen by mass but ~67% hydrogen by atomic percent. Thus, most of the mass of the human body is oxygen, but most of the atoms in the human body are hydrogen atoms. Both mass-composition and atomic composition figures are given below. Almost 99% of the mass of the human body is made up of the six elements oxygen, carbon, hydrogen, nitrogen, calcium, and phosphorus. Only about 0.85% is composed of another five elements: potassium, sulfur, sodium, chlorine, and magnesium. All are necessary to life. The remaining elements are trace elements, of which more than a dozen are thought to be necessary for life, or play an active role in health (e.g., fluorine, which hardens dental enamel but seems to have no other function). Not all elements which are found in the human body in trace quantities play a role in life. Some of these elements are thought to be simple bystander contaminants without function (examples: caesium, titanium), while many others are thought to be active toxins, depending on amount (cadmium, mercury, radioactives). The possible utility and toxicity of a few elements at levels normally found in the body (aluminum) is debated. Functions have been proposed for trace amounts of cadmium and lead, but these are almost certainly toxic in amounts normally found in the body. There is evidence that one element normally considered a toxin (arsenic) is essential in ultratrace quantities, even in mammals. Some elements that are clearly used in lower organisms and plants (arsenic, silicon, boron, nickel, vanadium) are probably needed by mammals also, but in far smaller doses. Two halogens used abundantly by lower organisms (fluorine and bromine) are presently known to be used by mammals only opportunistically. However, a general rule is that elements found in active biochemical use in lower organisms are often eventually found to be used in some way, by higher organisms.[citation needed]
Elemental composition
The average 70kg adult human body contains approximately 6.7 x 1027 atoms and contains at least detectable traces of 60 chemical elements. About 24 or 25 of these elements are thought to play an active positive role in life and health in humans.[1] The relative amounts of each element vary by individual, with the largest contributor due to fat/muscle/bone body composition ratio differences from person to person. The numbers in the table are averages of different numbers reported by different references. The human body is ~65% water, and water is ~11% hydrogen by mass but ~67% hydrogen by atomic percent.
Atomic number 8 Element Mass Oxygen Percent of [2][3][4][5][6][7] 65 Mass [8] (kg) 43 Atomic percent 24 [9] Positive health role in mammals Group
Yes (water, electron acceptor) /No (Reactive Oxygen Species) Yes (organic compounds are hydrocarbon derivatives) Yes (e.g. water) Yes (e.g. DNA and amino acids) Yes (e.g. Calmodulin and Hydroxylapatite in bones) Yes (e.g. DNA and phosphorylation) Yes (e.g. Na+/K+-ATPase)
16
Carbon
18
16
12
14
1 7 20
Hydrogen Nitrogen Calcium
10 3 1.4
7 1.8 1.0
63 0.58 0.24
1 15 2
15 19
Phosphorus Potassium
1.1 0.25
0.78 0.14
0.14 0.033
15 1
Composition of the human body
2
0.25 0.15 0.15 0.05 0.006 0.0037 0.14 0.10 0.095 0.019 0.0042 0.0026 0.038 0.037 0.024 0.0070 0.00067 0.0012 Yes (e.g.Cysteine, Methionine, Biotin, Thiamine) Yes (e.g. Na+/K+-ATPase) Yes (e.g. Cl-transporting ATPase) Yes (e.g. binding to ATP and other nucleotides) Yes (e.g. Hemoglobin, Cytochromes) Yes/No (topically hardens teeth; toxic in higher amounts) Yes (e.g. Zinc finger proteins) Yes (probable) No (?) No (?) No (?) No (?) (toxic in higher amounts) Yes (e.g. copper proteins) No(?) (toxic?) No(?) (toxic in higher amounts) No 0.0000012 6.0e-7 7.5e-7 No? (toxic) No(?) Yes (e.g. thyroxine, triiodothyronine) No 0.0000030 4.5e-8 0.0000015 8.9e-8 0.0000015 8.9e-8 0.0000015 Yes (probable) Yes (toxic in higher amounts) Yes (e.g. urease) Yes (not confirmed) Yes (e.g. Mn-SOD) Yes (not confirmed). Toxic in higher amounts Yes (not confirmed). Toxic in high amounts. Useful medically (mood stabilizer). No (toxic) No Yes (e.g. the molybdenum oxotransferases, Xanthine oxidase and Sulfite oxidase) No (?) 3.0e-7 Yes (e.g. vitamin B12) No (toxic) No (toxic) No 3.0e-7 No No 2 14 17 4 13 16 10 6 7 15 1 16 1 17 2 8 17
16 11 17 12 26 9
Sulfur Sodium Chlorine Magnesium Iron* Fluorine
30 14 37 38 35 82 29 13 48 58 56 50 53 22 5 34 28 24 25 33 3
Zinc Silicon Rubidium Strontium Bromine Lead Copper Aluminium Cadmium Cerium Barium Tin Iodine Titanium Boron Selenium Nickel Chromium Manganese Arsenic Lithium
0.0032 0.002 0.00046 0.00046 0.00029 0.00017 0.0001 0.000087 0.000072 0.000057 0.000031 0.000024 0.000016 0.000013 0.000069 0.000019 0.000014 0.0000024 0.000017 0.000026 0.0000031
0.0023 0.0010 0.00068 0.00032 0.00026 0.00012 0.000072 0.000060 0.000050 0.000040 0.000022 0.000020 0.000020 0.000020 0.000018 0.000015 0.000015 0.000014 0.000012 0.000007 0.000007
0.00031 0.0058 0.000033 0.000033 0.000030 0.0000045 0.0000104 0.000015 0.0000045
12 14 1 2 17 14 11 13 12
80 55 42
Mercury Caesium Molybdenum
0.000019 0.0000021 0.000013
0.000006 0.000006 0.000005
8.9e-8 1.0e-7 4.5e-8
12 1 6
32 27 51 47 41 40 57
Germanium Cobalt Antimony Silver Niobium Zirconium Lanthanum 0.0000021 0.000011 0.000001 0.00016 0.0006 0.000137
0.000005 0.000003 0.000002 0.000002 0.0000015 0.000001 8e-7
14 9 15 11 5 4
Composition of the human body
3
0.000012 7e-7 7e-7 6e-7 5e-7 5e-7 4e-7 0.000014 2e-7 2e-7 2e-7 0.000026 1.1e-7 1e-7 1.3e-7 1e-7 5.0e-8 2.0e-8 5e-9 1e-17 3.6e-8 3e-14 4.5e-8 1e-17% 3.0e-9 1.2e-8 3.0e-7 No No No No No (toxic) No No No No Yes (not confirmed) No (toxic) No (toxic) No No No (toxic) No (toxic) 6 2 2 16 13 3 15 13 13 11 3 5 5
52 31 39 83 81 49 79 21 73 23 90 92 62 74 4 88
Tellurium Gallium Yttrium Bismuth Thallium Indium Gold Scandium Tantalum Vanadium Thorium Uranium Samarium Tungsten Beryllium Radium
*Iron = ~3 g in men, ~2.3 g in women The elements needed for life are relatively common in the Earth's crust, and conversely most of the common elements are necessary for life. An exception is aluminium, which is the third most common element in the Earth's crust (after oxygen and silicon), but seems to serve no function in living cells. Rather, it is harmful in large amounts.[citation needed] Transferrins can bind aluminium.[10] Periodic table highlighting dietary elements[1]
H Li Be B Al Ti V Cr Mn Fe Ru Os Hs Co Ni Cu Zn Ga In Tl C Si Ge Sn Pb Fl Tm N P As Sb Bi O S Se Te Po F Cl Br I At He Ne Ar Kr Xe Rn
Na Mg K Ca Sc Y *
Rb Sr
Zr Nb Mo Tc Hf Ta W Re Bh
Rh Pd Ag Cd Ir Pt Au Hg
Cs Ba La
Fr Ra Ac ** Rf Db Sg *
Mt Ds Rg Cn Uut Gd Tb Dy Ho Er
Uup Lv Uus Uuo Yb No Lu Lr
Ce Pr Nd Pm Sm Eu U Np
** Th Pa
Pu Am Cm Bk Cf Es Fm Md
The four organic basic elements
Quantity elements
Essential trace elements
Function suggested from active handling in mammals, but no specific identified biochemical function
Composition of the human body
Composition by molecule type
The composition can also be expressed in terms of chemicals, such as: Water Proteins including those of hair, connective tissue, etc. Fats (or lipids) Apatite in bones Carbohydrates such as glycogen and glucose DNA Dissolved inorganic ions such as sodium, potassium, chloride, bicarbonate, phosphate Gases such as oxygen, carbon dioxide, nitrogen oxide, hydrogen, carbon monoxide, methanethiol. These may be dissolved or present in the gases in the lungs or intestines. Ethane and pentane are produced by oxygen free radicals.[11] Many other small molecules, such as amino acids, fatty acids, nucleobases, nucleosides, nucleotides, vitamins, cofactors. Free radicals such as superoxide, hydroxyl, and hydroperoxyl. The composition of the human body can be viewed on an atomic and molecular scale as shown in this article. The estimated gross molecular contents of a typical 20-micrometre human cell is as follows:[]
Molecule Water Other Inorganics Lipids Other Organics Protein RNA DNA Percent of Mass Mol.Weight (daltons) Molecules Percent of Molecules 65* 1.5 12 0.4 20 1.0 0.1 18* N/A N/A N/A N/A N/A 1e11 1.74e14* 1.31e12 8.4e11 7.7e10 1.9e10 5e7 46* 98.73* 0.74 0.475 0.044 0.011 3e-5 3e-11
Water: Obviously the amount of water is highly dependent on the level of hydration. DNA: A human cell also contains mitochondrial DNA. Sperm cells contain less mitochondrial DNA than other cells. A mammalian red blood cell contains no nucleus and thus no DNA.
Materials and tissues
Body composition can also be expressed in terms of various types of material, such as: Muscle Fat Bone and teeth Brain and nerves Connective tissue Blood 7% of body weight. Lymph Contents of digestive tract, including intestinal gas Urine Air in lungs
Composition of the human body
Composition by cell type
There are many species of bacteria and other microorganisms that live on or inside the healthy human body. In fact, 90% of the cells in (or on) a human body are microbes, by number[12][13] (much less by mass or volume). Some of these symbionts are necessary for our health. Those that neither help nor harm us are called commensal organisms.
References
[1] Ultratrace minerals. Authors: Nielsen, Forrest H. USDA, ARS Source: Modern nutrition in health and disease / editors, Maurice E. Shils ... et al.. Baltimore : Williams & Wilkins, c1999., p. 283-303. Issue Date: 1999 URI: (http:/ / hdl. handle. net/ 10113/ 46493) [2] Thomas J. Glover, comp., Pocket Ref, 3rd ed. (Littleton: Sequoia, 2003), p. 324 (), which in [3] turn cites Geigy Scientific Tables, Ciba-Geigy Limited, Basel, Switzerland, 1984. [5] Distribution of elements in the human body (by weight) (http:/ / www. daviddarling. info/ encyclopedia/ E/ elbio. html) Retrieved on 2007-12-06 [7] ) [8] J. Emsley, The Element, 3rd ed., Oxford: Clarendon Press, 1998. [9] Neilsen, cited (http:/ / hdl. handle. net/ 10113/ 46493) [11] Douglas Fox, "The speed of life" (http:/ / www. newscientist. com/ article/ mg18024195. 500-the-speed-of-life. html), New Scientist, No 2419, 1 November 2003.
Article Sources and Contributors
Article Sources and Contributors
Composition of the human body Source: http://en.wikipedia.org/w/index.php?oldid=543997864 Contributors: 0paco0, 1exec1, AznBurger, Betacommand, Carstensen, Chem8700, Cmglee, Cutelyaware, Double sharp, Download, Dratman, Epbr123, Eric Kvaalen, Eric Norby, Essent, Evagrim, Fishal, Frietjes, G716, Glacialfox, Hamiltondaniel, Iain.meek83, Icairns, Jmh649, Kzyn, Littlealien182, MeekMark, Memstone, Michael Hardy, Narayansg, Narrul, Nergaal, One-ply, Paul A, Philosophia X Known, Regstuff, Sbharris, Sp, SpaceFlight89, SpecMode, Spinningspark, Szczureq, TYelliot, Timwi, Wikipelli, Woohookitty, Wtshymanski, Zemyla, , 81 anonymous edits
License
Creative Commons Attribution-Share Alike 3.0 Unported //creativecommons.org/licenses/by-sa/3.0/
You might also like
- Minerals and Their Significance To A Human BodyDocument3 pagesMinerals and Their Significance To A Human BodyengghomNo ratings yet
- Nutrition BiochemDocument45 pagesNutrition BiochemKesha Marie TalloNo ratings yet
- Diseases of The Endocrine SystemDocument8 pagesDiseases of The Endocrine SystemMichelle HuangNo ratings yet
- Reproductive System Study GuideDocument6 pagesReproductive System Study GuideJames DaurayNo ratings yet
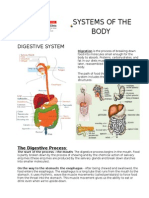
- Systems of The Body (Digestive)Document3 pagesSystems of The Body (Digestive)KryztalGhail LlanoraNo ratings yet
- Cations, Anions, and the Human BodyDocument1 pageCations, Anions, and the Human BodyKathryn Dominique PachecoNo ratings yet
- Energy System NotesDocument3 pagesEnergy System Notesapi-267326015100% (1)
- Human Health and DiseasesDocument24 pagesHuman Health and DiseasesAnujith AnuNo ratings yet
- Protocols@Internal Medicine@2018-2019Document54 pagesProtocols@Internal Medicine@2018-2019Sujeet ShahNo ratings yet
- MusculoskeletalDocument119 pagesMusculoskeletalsimplyrosalyn100% (5)
- Food Energy: Energy Usage in The Human BodyDocument11 pagesFood Energy: Energy Usage in The Human Bodyzharell1019No ratings yet
- Nutrition PaperDocument4 pagesNutrition Paperapi-433705780No ratings yet
- 2 1 3 A Feedback Student Response Sheet (Revised 10 2 14)Document7 pages2 1 3 A Feedback Student Response Sheet (Revised 10 2 14)api-28182463470% (10)
- Obstetrics Notes by DR Kamal Deep PDFDocument33 pagesObstetrics Notes by DR Kamal Deep PDFKrisha PatelNo ratings yet
- Human Respiration HandoutDocument4 pagesHuman Respiration HandoutJulius GutierrezNo ratings yet
- Chapter25 Urinary SystemDocument9 pagesChapter25 Urinary Systemkikajet23No ratings yet
- HormonesDocument29 pagesHormonesCandy Chieng67% (3)
- Body System ChecklistDocument6 pagesBody System Checklistapi-422967453No ratings yet
- List of Common Medical AbbreviationsDocument3 pagesList of Common Medical AbbreviationsJayakumar SankaranNo ratings yet
- Chapter 3 - Genetics (Fundamentals of Anatomy and Physiology For Nursing and Healthcare Students)Document87 pagesChapter 3 - Genetics (Fundamentals of Anatomy and Physiology For Nursing and Healthcare Students)Mary Grace MartinNo ratings yet
- Sources Hormone FunctionDocument2 pagesSources Hormone FunctionKatherine Joy MaderajeNo ratings yet
- Introduction To Human Body Anatomy PhysiologyDocument85 pagesIntroduction To Human Body Anatomy PhysiologyTalha AbbasiNo ratings yet
- Skeletal System Skeletal Anatomy: (Typical)Document10 pagesSkeletal System Skeletal Anatomy: (Typical)anon_660872041No ratings yet
- Name: ID:: Madiha Sayed Nagy. 51859Document12 pagesName: ID:: Madiha Sayed Nagy. 51859drng48No ratings yet
- Trivia UpdatedDocument11 pagesTrivia Updatedapi-242113272No ratings yet
- The ABC's in Pediatric Assessment and Care: Cymbeline Joan L. Pooten, RN NUM - Pediatric ICUDocument39 pagesThe ABC's in Pediatric Assessment and Care: Cymbeline Joan L. Pooten, RN NUM - Pediatric ICUggrrk7No ratings yet
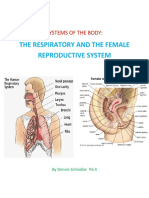
- Systems of The BodyDocument10 pagesSystems of The BodyKatrina SchindlarNo ratings yet
- Arq AmberDocument4 pagesArq AmberMaqsood AnsariNo ratings yet
- CH 7 Notes Cellular RespirationDocument3 pagesCH 7 Notes Cellular RespirationCJ100% (1)
- The Endocrine SystemDocument20 pagesThe Endocrine SystemJonathan TabbunNo ratings yet
- Biology of Human Disease WorksheetDocument2 pagesBiology of Human Disease WorksheetCorwin DavisNo ratings yet
- Bowel 1 EliminationDocument33 pagesBowel 1 EliminationKhumaeyraBundaSetiyaniWindratiNo ratings yet
- Human Biology PASS Mid-Tri Exam Buster 2021Document12 pagesHuman Biology PASS Mid-Tri Exam Buster 2021Robert100% (1)
- Endocrine System Workbook 2015Document8 pagesEndocrine System Workbook 2015api-298277560100% (1)
- Easy Tips for a Healthy DietDocument11 pagesEasy Tips for a Healthy Dietdiop1959No ratings yet
- Medical Terminology SyllabusDocument3 pagesMedical Terminology SyllabusAzza RezkNo ratings yet
- 3.14 Chapter 3 Water and Electrolytes Balance and ImblanceDocument140 pages3.14 Chapter 3 Water and Electrolytes Balance and ImblanceShourav SarkarNo ratings yet
- Phytoecdysteroids RkleinDocument11 pagesPhytoecdysteroids RkleintrungkunmingNo ratings yet
- Clinical Obstetrics Gynecology CoverDocument2 pagesClinical Obstetrics Gynecology CoverElla Wilson AteporNo ratings yet
- Mock Final Exam for Anatomy & PhysiologyDocument15 pagesMock Final Exam for Anatomy & PhysiologyAryan PatelNo ratings yet
- Behavior Change ProjectDocument10 pagesBehavior Change Projectapi-519752101No ratings yet
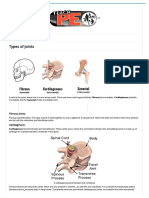
- Types of Joints - The Skeleton & Bones - Anatomy & Physiology PDFDocument3 pagesTypes of Joints - The Skeleton & Bones - Anatomy & Physiology PDFPankaj VermaNo ratings yet
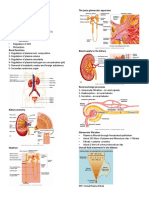
- Fisiologi 1 - Introducton Renal PhysiolgyDocument5 pagesFisiologi 1 - Introducton Renal PhysiolgyHachi Nini Shop IINo ratings yet
- First Year Curriculum OverviewDocument48 pagesFirst Year Curriculum Overviewseo167No ratings yet
- Meghana ResumeDocument2 pagesMeghana Resumeapi-488531273No ratings yet
- Roth 10e Nclex Chapter 05Document4 pagesRoth 10e Nclex Chapter 05jennaaahhhNo ratings yet
- APC Cardio Catalina-1Document21 pagesAPC Cardio Catalina-1Carmelo ScavoneNo ratings yet
- General Human Physiology and BiochemistryDocument9 pagesGeneral Human Physiology and BiochemistryleeminhoangrybirdNo ratings yet
- Introduction of AnatomyDocument18 pagesIntroduction of Anatomyjames nginyangiNo ratings yet
- Basal Metabolic RateDocument23 pagesBasal Metabolic RateMRITUNJAY KUMAR SHARMANo ratings yet
- Ventricles of BrainDocument16 pagesVentricles of BrainAzeemNo ratings yet
- Anatomy and Physiology of Nervous SystemDocument91 pagesAnatomy and Physiology of Nervous SystemSanhati Ghosh Banerjee100% (1)
- DiabetesDocument25 pagesDiabetesLawrence LarsonNo ratings yet
- Understanding The Heart - IntroductionDocument11 pagesUnderstanding The Heart - IntroductionChelsea Green Publishing100% (1)
- Dna and Rna Powerpoint 2Document46 pagesDna and Rna Powerpoint 2api-267309851No ratings yet
- Muscular System Web QuestDocument5 pagesMuscular System Web Questaidibwahab0% (1)
- Anatomical TermsDocument82 pagesAnatomical TermsRichelle Ann Gatchola EvangelistaNo ratings yet
- Stages of ShockDocument7 pagesStages of ShockNicholas TagleNo ratings yet
- Elements in Our Body and in TechnologyDocument46 pagesElements in Our Body and in TechnologySherilyn Closa BunagNo ratings yet
- Mesmerism and Its OpponentsDocument403 pagesMesmerism and Its OpponentsraschensNo ratings yet
- Division of A StringDocument26 pagesDivision of A StringraschensNo ratings yet
- Rationale of MesmerisimDocument254 pagesRationale of Mesmerisimraschens100% (1)
- The Didactical ChallengeDocument315 pagesThe Didactical ChallengeraschensNo ratings yet
- The Principles of MR Harrison S Timekeep PDFDocument63 pagesThe Principles of MR Harrison S Timekeep PDFraschensNo ratings yet
- A System of Mechanical Philosophy PDFDocument737 pagesA System of Mechanical Philosophy PDFraschensNo ratings yet
- The Circle of The Mechanical Arts PDFDocument711 pagesThe Circle of The Mechanical Arts PDFraschensNo ratings yet
- Turning and Mechanical Manipulation PDFDocument499 pagesTurning and Mechanical Manipulation PDFraschensNo ratings yet
- 18 Man Is A Compound Electrical MagnetDocument192 pages18 Man Is A Compound Electrical MagnetKeith WarlickNo ratings yet
- The Sacred CircleDocument611 pagesThe Sacred CircleraschensNo ratings yet
- A Rudimentary Treatise On Clock and Watc PDFDocument307 pagesA Rudimentary Treatise On Clock and Watc PDFraschensNo ratings yet
- Mechanical Philosophy Horology and Astro PDFDocument597 pagesMechanical Philosophy Horology and Astro PDFraschensNo ratings yet
- COMPLETE Letters of Petrus PeregrinusDocument76 pagesCOMPLETE Letters of Petrus PeregrinusDan BeesonNo ratings yet
- Euclid ElementsDocument449 pagesEuclid ElementsraschensNo ratings yet
- Dinural Variation of Terrestrial MagnetismDocument42 pagesDinural Variation of Terrestrial MagnetismraschensNo ratings yet
- CNCDocument26 pagesCNCraschensNo ratings yet
- Kepler's Laws of Planetary MotionDocument11 pagesKepler's Laws of Planetary MotionraschensNo ratings yet
- The Power Source: The 25 Amp & 60 Amp RectifierDocument0 pagesThe Power Source: The 25 Amp & 60 Amp RectifierraschensNo ratings yet
- Kepler's Laws of Planetary MotionDocument11 pagesKepler's Laws of Planetary MotionraschensNo ratings yet
- Home etching circuit boards with toner transfer in under 40 stepsDocument5 pagesHome etching circuit boards with toner transfer in under 40 stepsraschensNo ratings yet
- Euclid ElementsDocument571 pagesEuclid ElementsraschensNo ratings yet
- Chinese ChronologyDocument290 pagesChinese ChronologyraschensNo ratings yet
- Knowledge: ManualDocument36 pagesKnowledge: ManualraschensNo ratings yet
- The Art of ChannelingDocument14 pagesThe Art of ChannelingEarthcat100% (6)
- The Word Made Manifest Through Sacred GeometryDocument384 pagesThe Word Made Manifest Through Sacred GeometryDerek Gerlach100% (15)
- Embellishing Your Woodturning With Inlay TechniquesDocument5 pagesEmbellishing Your Woodturning With Inlay TechniquesraschensNo ratings yet
- Cosmic Law - Patterns in The Universe by Dean BrownDocument67 pagesCosmic Law - Patterns in The Universe by Dean BrownTaur100% (9)
- COMPLETE Letters of Petrus PeregrinusDocument76 pagesCOMPLETE Letters of Petrus PeregrinusDan BeesonNo ratings yet
- Composition of The Human BodyDocument6 pagesComposition of The Human BodyraschensNo ratings yet
- Dinural Variation of Terrestrial MagnetismDocument42 pagesDinural Variation of Terrestrial MagnetismraschensNo ratings yet
- SS316 Butt WeldingDocument124 pagesSS316 Butt Weldingdevinder_bamrahNo ratings yet
- Compatibility of Materials With Liquid Oxygen (Impact Sensitivity Threshold and Pass-Fail Techniques)Document12 pagesCompatibility of Materials With Liquid Oxygen (Impact Sensitivity Threshold and Pass-Fail Techniques)Ahmed AlzubaidiNo ratings yet
- METALLODRUGSDocument21 pagesMETALLODRUGSAditya Verma100% (1)
- HLB Values and Saa Functionality: Technical Brief 2010 Volume 1Document2 pagesHLB Values and Saa Functionality: Technical Brief 2010 Volume 1ArangaNo ratings yet
- UsersGuide deltaECDocument285 pagesUsersGuide deltaECHakan AteşNo ratings yet
- ASTM F1089-18 Standard Test Method For Corrosion of Surgical InstrumentsDocument1 pageASTM F1089-18 Standard Test Method For Corrosion of Surgical InstrumentsMarketing ProjectNo ratings yet
- Literature Review On Carbon Dioxide Capture by AbsorptionDocument21 pagesLiterature Review On Carbon Dioxide Capture by AbsorptionTU_MTECH_ENV11No ratings yet
- ENSCO DS6 - Unit320P04DS6 - SNDS6320D - Mud Pump D - Abnormal - 25506970Document5 pagesENSCO DS6 - Unit320P04DS6 - SNDS6320D - Mud Pump D - Abnormal - 25506970mohamed hamedNo ratings yet
- A510A510M-13 Standard Specification For General Requirements For Wire Rods and Coarse Round Wire, Carbon Steel, and Alloy SteelDocument6 pagesA510A510M-13 Standard Specification For General Requirements For Wire Rods and Coarse Round Wire, Carbon Steel, and Alloy Steeltjt4779100% (2)
- Aetheric Battery - Copy #1Document6 pagesAetheric Battery - Copy #1Keresztúri FerencNo ratings yet
- Aapt United States Physics Team AIP 2010: Do Not Distribute This PageDocument15 pagesAapt United States Physics Team AIP 2010: Do Not Distribute This PagelagostinhaNo ratings yet
- Algebra:: N N N N N N 1B 2B ZoneDocument6 pagesAlgebra:: N N N N N N 1B 2B Zonevenedick sapongayNo ratings yet
- GtryeDocument66 pagesGtryePholNo ratings yet
- Applied HydrogeologyDocument10 pagesApplied HydrogeologyJuliana Quinderé Carneiro0% (1)
- MCQ Gad 2Document8 pagesMCQ Gad 2AlhanNo ratings yet
- Ultrasonic Testing Equipment Manufacturers - Canopus InstrumentsDocument1 pageUltrasonic Testing Equipment Manufacturers - Canopus InstrumentscanopusinstrumentsNo ratings yet
- Transmission Insulator SpecificationDocument0 pagesTransmission Insulator Specificationrenjithas2005No ratings yet
- Synthesis of Flavone from 2-HydroxyacetophenoneDocument11 pagesSynthesis of Flavone from 2-Hydroxyacetophenonearefeena100% (2)
- LED Lighting Systems - LEEDDocument9 pagesLED Lighting Systems - LEEDErvin LiquidwaxNo ratings yet
- Molecules of LifeDocument38 pagesMolecules of LifeYana PanlilioNo ratings yet
- CASSIDA FLUID GL 220 SDSDocument5 pagesCASSIDA FLUID GL 220 SDSFajar EkoNo ratings yet
- How The Concept of The Element Evolved From Ancient Greek To The PresentDocument23 pagesHow The Concept of The Element Evolved From Ancient Greek To The PresentPineda, Sean AlfredNo ratings yet
- Bi Rotator FormDocument17 pagesBi Rotator FormBùi Hắc HảiNo ratings yet
- 7 Paint DefectsDocument29 pages7 Paint DefectsMohamed Nouzer100% (2)
- CHEM111 Week 4.2 - Chemical Bonds and Compounds Part 2 - Covalent Bond ModelDocument17 pagesCHEM111 Week 4.2 - Chemical Bonds and Compounds Part 2 - Covalent Bond ModelKharl Roei SalvadorNo ratings yet
- Datos Bibliográficos A 35ºCDocument6 pagesDatos Bibliográficos A 35ºCANA VILA FERREIRONo ratings yet
- Chapter-2-LEVEL MEASUREMENTDocument26 pagesChapter-2-LEVEL MEASUREMENTMohammed YusufNo ratings yet
- Tutorial 1 - AnswersDocument8 pagesTutorial 1 - AnswersRaymond Kakala100% (6)
- Microbial Test KitDocument3 pagesMicrobial Test KitLutfi HidayatNo ratings yet
- Material Science QuestionsDocument40 pagesMaterial Science QuestionsLucky KapoorNo ratings yet