Professional Documents
Culture Documents
Form 5 Lesson 7 Structure
Uploaded by
Aidah AmirOriginal Description:
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Form 5 Lesson 7 Structure
Uploaded by
Aidah AmirCopyright:
Available Formats
OMEGA TUITION CENTRE
Students name : ____________________________________ ________________________ Teachers Name : Mr Chew Chin Kuen Subject : Chemistry Form 5 Class Attend : 5 Chemistry 6 Chapter 1 : Rate of reaction lesson 7 : 2nd Feb 2013 (2.30 4.30 PM)
Paper 2
As catalyst
Manganese (IV) oxide / Iron (III) oxide
Rate of reaction is inversely proportional with time, as the rate of reaction decreased with time
reaction is completed
mol of KClO3 = MV / 1000 ; mol of KClO3 = (1.0)(4.0)/1000 = 0.004 mol From equation 2 mol KClO3 = 1 mol of O2 mol of O2 = 0.002 mol V of O2 = mol x Vm ; V of O2 = 0.002 mol x 24 dm3 / mol = 0.048 dm3 @ 48 cm3
Rate
= (48 cm3 - 0) / (45 s - 0) = 1.067 cm3 / s
all hydrochloric acid in the mixture is used up
Mass of CO2 / g
Time / s at the first 60 rate = 3.5 - 0.0 / 60 - 0 = 0.583 g / s
By using marble in powder form / increase the temperature / add concentration of HCl
Fe (s) + H2SO4 (aq) FeSO4 (aq) + H2 (g)
; mol = 3.0 / 56 = 0.0536 mol mol of H2SO4 = MV / 1000 ; mol = (0.5)(30) / 1000 mol = 0.015 mol (limitant) since 1 mol of H2SO4 = 1 mol of H2 ; mol of H2 = 0.015 mol Vgas = mol x Vm @ Vgas = 0.015 x 24 Vgas = 0.36 dm3 @ 360 cm3
mol of Fe = mass / Ar
Even though the type of acid used are different, but the mol of H+ is the same for both H2SO4 and HCl, since H2SO4 is a diproctic acid, while HCl is a monoproctic acid
Rate of reaction in C is higher than A, since catalyst is add into the mixture, CuSO4 helps to lower the activation energy by providing alternative routes for reaction to occur
The temperature used in Exp B is higher than A. This will increase the kinetic energy of particles hence caused the collision between reactants become more rapids
Mg (s) + 2 H+ (aq) Mg2+ (aq) + H2 (g)
Since ethanoic acid is weak acid while HCl is strong acid. the concentration of H+ in HCl is higher, hence caused a higher frequency of effective collision
Since the amount of H+ given by H2SO4 is 2 times greater than amount of H+ given by HCl. Higher the concentration of H+, higher the frequency of effective collision
Rate of reaction in S is higher than Q, since catalyst is add into the mixture, CuSO4 helps to lower the activation energy by providing alternative routes for reaction to occur
Time taken for the reaction to occur will be lower than 35 second, as temperature increase, rate of reaction also increase. This is due to, particles has higher kinetic energy which caused a higher frequency of effective collision.
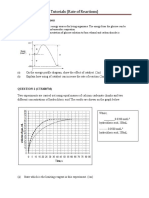
Carbon dioxide gas is liberated when limestone reacts with dilute acids. Figure below shows the volume of carbon dioxide gas collected at room conditions during the duration of the reaction.
(a)
Write an equation for the reaction of limestone with dilute hydrochloric acid.
CaCO3 (s) + 2 HCl (aq) CaCl2 (aq) + CO2 (g) + H2O (l) .
(b) (i) How long did the reaction last?
210 s
(ii) (iii)
70 cm What is the total volume of carbon dioxide gas evolved? .
Calculate the average speed of the reaction.
Rate = 70 / 210 = 0.33 cm3 / s
(c) Calculate the rate of reaction at t = 60 s and t = 90 s. At 60 s, rate = (80 - 40) / (120 - 30) = 0.444 cm3 / s At 90 s, rate = (80 - 50) / (165 - 30) = 0.222 cm3 / s
(d)
Calculate the mass of limestone reacted.
mol = 70 / 24000 cm3 / mol = 0.00292 mol From the chemical equation ; 1 mol of CO2 = 1 mol of CaCO3 Hence mol of CaCO3 = 0.00292 mol Mass of CaCO3 = mol x RMM ; mol = 0.00292 mol x [40 + 12 + 3(16)] mass = 0.292 g Using mol of CO2 = V / Vm ; (e) The concentration of the hydrochloric acid is 0.25 mol dm-3. Calculate the volume of hydrochloric acid reacted. From mol of CO2 reacted = 0.00292 mol ; Since 2 mol HCl = 1 mol CO2 ; mol of HCl = 0.00146 mol Volume of HCl = mol x 1000 / M ; VHCl = 0.00146 x 1000 / 0.25 VHCl = 5.84 cm3
Figure below shows the set-up used to investigate the reaction between calcium carbonate and hydrochloric acid.
The volume of carbon dioxide was recorded at 10-minute intervals under room conditions. Some of the calcium carbonate remained undissolved at the end of the experiment. The results of the experiment are given in the following table. Time (min) Volume of CO2 collected (cm3) (a)
2-
0 0
20 700
40 975
60 1140
80 1175
100 1200
120 1200
Write an ionic equation for the reaction between calcium carbonate and hydrochloric acid.
3 2 2 . (b) (i) Complete the table below.
CO
(s) + 2 H+ (aq) + CO (g) + H O (l)
Time interval (min) 0 - 20 20 - 40 40 - 60 60 - 80
Volume of gas collected (cm3)
700 275 165 25
(ii) Explain the variation in the volumes of carbon dioxide collected during these intervals. Volume of the gas collected decrease with the interval time due to the amount of reactants used for the reaction become smaller with time. Hence the amount of product obtained will be lower with time (c) Why did the reaction stop after 100 minutes?
You might also like
- Rate NotesDocument16 pagesRate NotesMegan GohNo ratings yet
- Ap Unit7 WorksheetDocument4 pagesAp Unit7 Worksheetburcak gecNo ratings yet
- 4.3 Reaction Rates and Reversible ReactionsDocument18 pages4.3 Reaction Rates and Reversible ReactionsVictor VC100% (5)
- 64edf0f4e41caDocument6 pages64edf0f4e41caDanzell JonathanNo ratings yet
- Revision StoichiometryDocument12 pagesRevision StoichiometryFangru CaoNo ratings yet
- RTS Chemistry SPM Question Bank Chapter 10Document8 pagesRTS Chemistry SPM Question Bank Chapter 10dobbybibiNo ratings yet
- GCE O Levels (Singapore) - Speed of ReactionDocument3 pagesGCE O Levels (Singapore) - Speed of ReactionChong56No ratings yet
- HKDSE CHEMISTRY - Book 4A AnsDocument48 pagesHKDSE CHEMISTRY - Book 4A AnsSteven Chu100% (1)
- Ans SL MC Test r2 The Amount of Chemical ChangeDocument9 pagesAns SL MC Test r2 The Amount of Chemical ChangeALINo ratings yet
- 5 Solution Stoichiometry (S)Document11 pages5 Solution Stoichiometry (S)Mr TanNo ratings yet
- RTS Chemistry SPM Question Bank Chapter 10Document8 pagesRTS Chemistry SPM Question Bank Chapter 10Scorched ZenNo ratings yet
- Tutorial 1 Rate of ReactionDocument2 pagesTutorial 1 Rate of ReactionchenanNo ratings yet
- Unit 4 (Mole) PAPER 4Document118 pagesUnit 4 (Mole) PAPER 4Muhammad Hasnain SikandarNo ratings yet
- Chemistry As Revision Questions F332Document23 pagesChemistry As Revision Questions F332LilliNo ratings yet
- Chemistry Topic One QuestionsDocument30 pagesChemistry Topic One QuestionsAruba Dhaduk100% (1)
- Chemistry Past Paper Ch1.1Document20 pagesChemistry Past Paper Ch1.1Raymond ChanNo ratings yet
- Quantitative ChemistryDocument32 pagesQuantitative ChemistryElena EngiNo ratings yet
- C4.8 CalculationsChemsheets GCSE 1105 (Titrations 1)Document2 pagesC4.8 CalculationsChemsheets GCSE 1105 (Titrations 1)Bee AbeilleNo ratings yet
- Topic 6/16 Kinetics (Rates of Reactions)Document14 pagesTopic 6/16 Kinetics (Rates of Reactions)Oyinkansola OsiboduNo ratings yet
- (Answer Key) Calculation Exercise - 元素の貓 - 免費dse化學練習Document6 pages(Answer Key) Calculation Exercise - 元素の貓 - 免費dse化學練習Belladonna Lee100% (1)
- Chem F.6 Full Report 1Document11 pagesChem F.6 Full Report 1stephenliyuting_1992100% (2)
- Music 2Document18 pagesMusic 2JonathanNgNo ratings yet
- WORKSHEET - Chemical Formulae and EquationsDocument7 pagesWORKSHEET - Chemical Formulae and EquationsMaisha Islam100% (1)
- 2009 HCI Prelim P2Document15 pages2009 HCI Prelim P2Felicia LimNo ratings yet
- Chem G-9 Lesson 7 IGCSE Qs - Rates of ReactionDocument24 pagesChem G-9 Lesson 7 IGCSE Qs - Rates of ReactionKarim WaelNo ratings yet
- Chapter 1: Rate of Reaction: Larning Task 1.2 Problem SolvingDocument29 pagesChapter 1: Rate of Reaction: Larning Task 1.2 Problem Solvingamin_zamanNo ratings yet
- Basic Engineering CorrelationDocument15 pagesBasic Engineering CorrelationJM CartañoNo ratings yet
- Y1 P2 Summative Topics 1.1 1.2 11.1Document7 pagesY1 P2 Summative Topics 1.1 1.2 11.124zaltayNo ratings yet
- Revision of KS4 Calculations in Chemistry For KS5 WorksheetDocument6 pagesRevision of KS4 Calculations in Chemistry For KS5 WorksheetniaNo ratings yet
- Worksheet Revision Calculations ks5Document6 pagesWorksheet Revision Calculations ks5James YangNo ratings yet
- IGCSE Prep - 3Document17 pagesIGCSE Prep - 3Yoel Friady HutabaratNo ratings yet
- Class Test 1 Rate of Reaction For Edexcel A2 ChemistryDocument7 pagesClass Test 1 Rate of Reaction For Edexcel A2 Chemistryjeffydaniel1972No ratings yet
- Tutorials (Rate of Reactions) : QUESTION 1 (2006 CT3 Jan)Document7 pagesTutorials (Rate of Reactions) : QUESTION 1 (2006 CT3 Jan)Subesh ShanmugamNo ratings yet
- Topic-1.1 Formulae, Equations and Amount of SubstancesDocument20 pagesTopic-1.1 Formulae, Equations and Amount of SubstancesAneeka KamalNo ratings yet
- Application of Rate ReactionDocument10 pagesApplication of Rate ReactionRahmawati PutrianasariNo ratings yet
- 9 Thermochemistry (S)Document23 pages9 Thermochemistry (S)Mr TanNo ratings yet
- Determination of The Valency of MagnesiumDocument7 pagesDetermination of The Valency of MagnesiumJiaxinOoiNo ratings yet
- Chemistry Experiment 1Document8 pagesChemistry Experiment 1Shirah CoolNo ratings yet
- CH 2Document43 pagesCH 2Tamanna GaurNo ratings yet
- Ap Unit7 Worksheet AnswersDocument5 pagesAp Unit7 Worksheet Answersburcak gecNo ratings yet
- AS Level Topic 5 TestDocument10 pagesAS Level Topic 5 TestMorvan BarnesNo ratings yet
- Mole Concept Moles Equations and MolarityDocument18 pagesMole Concept Moles Equations and MolarityNageya paulNo ratings yet
- CHEM101 2008 SetB-1Document6 pagesCHEM101 2008 SetB-1shishir kafleNo ratings yet
- Tutorial-Manual CH1002Document18 pagesTutorial-Manual CH1002Gift Chulu100% (2)
- Class 9 - 1st Term Calculation WorksheetDocument5 pagesClass 9 - 1st Term Calculation Worksheetabdulhadisaqib290No ratings yet
- Gas Liquid AbsorptionDocument9 pagesGas Liquid AbsorptionShashwat OmarNo ratings yet
- Chemistry Form 5 Chapter 1 - Rate of ReactionDocument63 pagesChemistry Form 5 Chapter 1 - Rate of ReactionSiti Nursyafiqah100% (7)
- Name of Student Tsui Yee Grade Y10 TrustDocument25 pagesName of Student Tsui Yee Grade Y10 Trustapi-361490645No ratings yet
- Expt01 HCL and NaOH AnsDocument3 pagesExpt01 HCL and NaOH AnsaragpdNo ratings yet
- As Expt 2.1 (4) - Enthalpy of Formation of CaCO3Document4 pagesAs Expt 2.1 (4) - Enthalpy of Formation of CaCO3Diddled_skittles100% (1)
- Succeed I Can WorksheetDocument8 pagesSucceed I Can WorksheetCorinne Amelia SimNo ratings yet
- Naskah Murid Modul 1-Rate of ReactionDocument14 pagesNaskah Murid Modul 1-Rate of ReactionIza MohdSabriNo ratings yet
- Moles TestDocument5 pagesMoles TestMahedyNo ratings yet
- Acid N Salt RevisionDocument6 pagesAcid N Salt RevisionTennarasu PannirselvamNo ratings yet
- DP10 Online Quiz RevisedDocument8 pagesDP10 Online Quiz RevisedjackyqinsjNo ratings yet
- As Unit 1 Chapter 1 Past PapersDocument20 pagesAs Unit 1 Chapter 1 Past PapersK K Chamath Aachinthya0% (1)
- Topic 1 Quantitative SLHL Test ADocument9 pagesTopic 1 Quantitative SLHL Test APak Hei Marcus CHOWNo ratings yet
- Revision Exercise (Organic Chemistry) : Pahang 2010Document5 pagesRevision Exercise (Organic Chemistry) : Pahang 2010Sammul IgnatiusNo ratings yet
- Effective PresentationsDocument2 pagesEffective PresentationsSammul IgnatiusNo ratings yet
- Students Matching F 5Document10 pagesStudents Matching F 5faezahsapixNo ratings yet
- Students Matching F 5Document10 pagesStudents Matching F 5faezahsapixNo ratings yet
- The Disappearing Spoon: And Other True Tales of Madness, Love, and the History of the World from the Periodic Table of the ElementsFrom EverandThe Disappearing Spoon: And Other True Tales of Madness, Love, and the History of the World from the Periodic Table of the ElementsRating: 4 out of 5 stars4/5 (146)
- Periodic Tales: A Cultural History of the Elements, from Arsenic to ZincFrom EverandPeriodic Tales: A Cultural History of the Elements, from Arsenic to ZincRating: 3.5 out of 5 stars3.5/5 (137)
- Is That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeFrom EverandIs That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeRating: 5 out of 5 stars5/5 (4)
- Organic Chemistry for Schools: Advanced Level and Senior High SchoolFrom EverandOrganic Chemistry for Schools: Advanced Level and Senior High SchoolNo ratings yet
- Handbook of Formulating Dermal Applications: A Definitive Practical GuideFrom EverandHandbook of Formulating Dermal Applications: A Definitive Practical GuideNo ratings yet
- Process Plant Equipment: Operation, Control, and ReliabilityFrom EverandProcess Plant Equipment: Operation, Control, and ReliabilityRating: 5 out of 5 stars5/5 (1)
- Chemistry for Breakfast: The Amazing Science of Everyday LifeFrom EverandChemistry for Breakfast: The Amazing Science of Everyday LifeRating: 4.5 out of 5 stars4.5/5 (90)
- Phase Equilibria in Chemical EngineeringFrom EverandPhase Equilibria in Chemical EngineeringRating: 4 out of 5 stars4/5 (11)
- Monkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeFrom EverandMonkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeRating: 4 out of 5 stars4/5 (1)
- Taste: Surprising Stories and Science About Why Food Tastes GoodFrom EverandTaste: Surprising Stories and Science About Why Food Tastes GoodRating: 3 out of 5 stars3/5 (20)
- Essential Oil Chemistry Formulating Essential Oil Blends that Heal - Aldehyde - Ketone - Lactone: Healing with Essential OilFrom EverandEssential Oil Chemistry Formulating Essential Oil Blends that Heal - Aldehyde - Ketone - Lactone: Healing with Essential OilRating: 5 out of 5 stars5/5 (1)
- The Elements We Live By: How Iron Helps Us Breathe, Potassium Lets Us See, and Other Surprising Superpowers of the Periodic TableFrom EverandThe Elements We Live By: How Iron Helps Us Breathe, Potassium Lets Us See, and Other Surprising Superpowers of the Periodic TableRating: 3.5 out of 5 stars3.5/5 (22)
- The Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsFrom EverandThe Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsRating: 5 out of 5 stars5/5 (3)
- The Periodic Table of Elements - Post-Transition Metals, Metalloids and Nonmetals | Children's Chemistry BookFrom EverandThe Periodic Table of Elements - Post-Transition Metals, Metalloids and Nonmetals | Children's Chemistry BookNo ratings yet
- Bioplastics: A Home Inventors HandbookFrom EverandBioplastics: A Home Inventors HandbookRating: 4 out of 5 stars4/5 (2)
- Process Steam Systems: A Practical Guide for Operators, Maintainers, and DesignersFrom EverandProcess Steam Systems: A Practical Guide for Operators, Maintainers, and DesignersNo ratings yet
- Physical and Chemical Equilibrium for Chemical EngineersFrom EverandPhysical and Chemical Equilibrium for Chemical EngineersRating: 5 out of 5 stars5/5 (1)
- Science Goes Viral: Captivating Accounts of Science in Everyday LifeFrom EverandScience Goes Viral: Captivating Accounts of Science in Everyday LifeRating: 5 out of 5 stars5/5 (1)
- Chemistry: a QuickStudy Laminated Reference GuideFrom EverandChemistry: a QuickStudy Laminated Reference GuideRating: 5 out of 5 stars5/5 (1)