Professional Documents
Culture Documents
HCL PDF
Uploaded by
skrim240Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
HCL PDF
Uploaded by
skrim240Copyright:
Available Formats
CHEM 332L
Physical Chemistry Lab
Revision 2.2
The Vibrational-Rotational Spectrum of HCl
In this experiment we will examine the fine structure of the vibrational fundamental line for
H
35
Cl in order to determine several spectroscopic constants; the Rotational Constants B
0
and B
1
,
the Centrifugal Distortion Constant D
e
, and the Fundamental Vibrational Band Origin
.
Inclusion of a few literature values for Rotational Constants B
v
for higher vibrational states
v=2,3,4, will allow us to determine the Equilibrium Rotational Constant B
e
. Inclusion of
literature values for the 1
st
thru 4
th
vibrational overtones will allow us to determine further
spectroscopic constants; the Equilibrium Fundamental Vibrational Frequency
and the
Anharmonicity Constant
. Finally, we will assume the HCl molecule is behaving
approximately as two masses connected by a stiff spring of constant k rotating with internuclear
distance r. We will then extract these molecular parameters from the appropriate spectroscopic
constants.
First, we must recognize that Hydrogen Chloride is a diatomic gaseous substance. Because it is a
diatomic molecule, it will have one vibrational mode and one rotational mode of
motion. Thus, we can model HCl molecules as vibrating rotors.
We begin by examining the vibrational motion of the molecule and treat the
atoms of the molecule as being connected by a Hookean spring of constant k. In
this case, the Hamiltonian operator for the system is written as:
(Eq. 1)
Solving the Schrodinger wave equation
= E for the energy yields:
E
v
= hv (v + ) v = 0, 1, 2, (Eq. 2)
where v = (1/2t) is the Fundamental Vibrational Frequency of the system and is its
reduced mass. v is the quantum number associated with the vibrational energy state E
v
. In
P a g e | 2
anticipation of discussing spectral lines arising from with energy transitions, we can define the
vibrational spectral "Term" as:
G(v) =
= (v + ) (Eq. 3)
where = v/c. h and c are Planck's Constant and the speed of light, respectively. If we allow
for non-Hookean behavior, then:
G(v) =
(v + ) -
(v + )
2
(Eq. 4)
where
is an anharmonicity constant. This anharmonicity correction lowers the energy of
each vibrational state because the parabolic potential of Hookean behavior tends to broaden and
flatten and become more Morse like. The anharmonicity correction becomes more severe at
higher vibrational states where the deviation from Hookean behavior is more pronounced.
A couple of points must be kept in mind when
considering equation 4. First, anharmonicity
will cause the fundamental frequency of the
oscillator v to depend on which vibrational
state the system is in. Thus, we define an
equilibrium fundamental frequency
; this
equals v at the bottom of the potential well
where anharmonicity vanishes. Second,
and
of equation 4 are spectroscopic constants
that can be measured regardless of the model
used for representing the molecule. On the
other hand, the spring constant k of equation 2
depends on the nature of the model used to
represent the molecule. This must be kept in mind when we extract values of k from the
measured spectroscopic constant
.
Now, vibrational spectral lines will occur when a photon is absorbed causing an energy transition
from E
v''
to E
v'
, where v'' is the lower quantum state and v' is the upper. In terms of spectral
Terms, this transition is represented as:
A = G(v') - G(v'') (Eq. 5)
The fundamental line vibrational spectral line
will be given by A
= G(1) - G(0).
Overtones A
, A
, etc. are quantum
forbidden by the selection rule Av = 1 and so
are very weak. (Hot Bands, 1 2, 2 3, etc. are
also very weak because higher vibrational states
will not be significantly populated due to
P a g e | 3
insufficient thermal energy in the system.) Schematically, the spectrum will appear as:
Using high resolution spectrometers, it is observed each of these lines exhibits more detailed
structure.
For instance, at fairly low resolution, the Fundamental Line appears as a doublet. Using a higher
resolution spectrometer, it is observed this line exhibits fine structure. At higher resolution yet,
each fine structure line is observed to split into a pair of lines.
The fine structure can be explained by considering the rotational motion of the HCl molecule.
The splitting of each fine structure line can be explained by considering the sample to be made
up of a mixture H
35
Cl and H
37
Cl molecules.
So, turning to the rotational motion of the HCl molecule, we treat the molecule as a rigid rotor.
Again, we write the system's Hamiltonian operator:
(Eq. 6)
P a g e | 4
Solving the Schrodinger wave equation
= E for the energy yields:
E
l
=
l (l + 1) l = 0, 1, 2, (Eq. 7)
where I = r
2
is the moment of inertia of the molecule. We define the rotational spectral "Term"
as:
F(J) =
= B J (J + 1) (Eq. 8)
where B = h/8t
2
cI is the system's Rotational Constant. (Note: In spectroscopic notation, the
quantum number l is denoted by the symbol J.) If we allow for centrifugal distortion as the
molecule begins to "spin" faster and faster, then the Term becomes:
F(J) = B
e
J (J + 1) - D
e
J
2
(J + 1)
2
(Eq. 9)
where D
e
is the Centrifugal Distortion Constant. Again, B
e
and D
e
are spectroscopically
determinable constants that are independent of molelcular model.
Expected values of "I" suggest these terms will lie very close together and that the spectra lines:
A = F(J') - F(J'') (Eq. 10)
will lie in the microwave region. Here the selection rule for allowed transitions is AJ = 1.
"Hot" bands are not a problem because the energy levels lie close enough
together that thermal population of upper rotational states is
considerable. Therefore, in microwave spectroscopy, energy transitions,
as pictured to the left, will occur.
These rotational states will lie on top the vibrational states, giving us
spectral Terms T(v,J) = G(v) + F(J). This allows us to describe the
fine structure lines as being due to transitions of the form:
A = T(v',J') - T(v'',J'') (Eq. 11)
Inserting our expressions for G(v) and F(J) into this equation, we obtain
the following general result for the spectral lines:
A = (v' - v'')
[(v' - v'')(v' + v'' + 1)]
+ B
v'
J'(J' + 1) - B
v''
J''(J'' + 1) - D
e
J'
2
(J' + 1)
2
+ D
e
J''
2
(J'' + 1)
2
P a g e | 5
(Eq. 12)
Note that because the molecule "stretches" slightly as it moves into higher vibrational states, due
to anharmonicity, the internuclear distance r will depend on vibrational quantum number v
(denoted as r
v
), hence "I" will depend on the vibrational quantum number and therefore "B" will
also depend on the vibrational quantum number (denoted as B
v
). We define a vibration-rotation
Coupling Constant as o
e
such that the nature of B's dependence on v is:
B
v
= B
e
- o
e
(v + ) (Eq. 13)
A similar problem occurs for the distortion constant D, except the coupling is weak enough we
can neglect it.
With all the overlayed rotational fine-structure on the vibrational band, how do we define the
"position" of the vibrational spectral line? Well, formally, we define the line's Band Origin
as:
= (v' - v'')
[(v' - v'')(v' + v'' + 1)] (Eq. 14)
If we are dealing with the fundamental line, then v'' = 0 and v' = 1. Then:
A
- 2
+ B
1
J'(J' + 1) - B
0
J''(J'' + 1) - D
e
J'
2
(J' + 1)
2
+ D
e
J''
2
(J'' + 1)
2
(Eq. 15)
Now, one of two situations can occur; AJ = J'-J'' = +1 or AJ = J'-J'' = -1. If AJ = +1, we are said
to be in the R-branch of the line; which occurs at higher wavenumbers. If AJ = -1, we are in the
P-branch; at lower wavenumbers.
P a g e | 6
Thus, for the R-branch, we have:
R(J'') = A
10,AJ=+1
=
- 2
+ (B
1
+ B
0
) (J''+1)
+ (B
1
- B
0
) (J''+1)
2
- 4D
e
(J''+1)
3
(Eq. 1)
and, for the P-branch, we have:
P(J'') = A
10,AJ=-1
=
- 2
- (B
1
+ B
0
) J'' + (B
1
- B
0
) J''
2
+ 4D
e
J''
3
(Eq. 17)
We now turn to the question of how to extract the spectroscopic constants from the measured
positions of the fine-structure lines; R(0), R(1), R(2), ., P(1), P(2), P(3), .
First, how do we extract the rotational constants B
0
and B
1
and the distortion constant D
e
?
Equations 16 and 17 can be re-arranged to obtain the following forms:
= B
1
- 2 D
e
(J''
2
+ J'' + 1) (Eq. 18)
= B
0
- 2 D
e
(J''
2
+ J'' + 1) (Eq. 19)
Thus, measurements of R(J'') and P(J''), when plotted according to equations 18 and 19, and the
results subjected to a linear least square analysis, will yield the desired constants.
Next, to determine the band origin we use another manipulation of equations 16 and 17:
+ (B
1
- B
0
) (J'' + 1)
2
(Eq. 20)
Another linear least square analysis is needed to obtain this constant.
To obtain
and
we will need additional information concerning the overtone lines for the
case where v'' = 0. We can rearrange equation 14 to yield the following.
(v' + 1) (Eq. 21)
Again, a linear least square analysis will give us the desired constants. Overtone data for H
35
Cl
is:
Overtone
[cm
-1
]
0 - 2 5667.9841
0 - 3 8346.782
0 - 4 10922.81
0 - 5 13396.19
P a g e | 7
Next, we can use our measured values of B
0
and B
1
, along with additional data, and a linear least
square analysis according to equation 13, to determine B
e
. Needed data for H
35
Cl is:
v B [cm
-1
]
2 9.834663
3 9.534909
4 9.2360
5 8.942
Finally, once the spectroscopic constants
and B
e
are determined, we can use our model for the
HCl molecule to determine the molecular parameters k and r
e
.
v
e
=
c = (1/2t) (Eq. 22)
B
e
= h / 8t
2
cr
e
2
(Eq. 23)
(See development above.) Here r
e
represents the equilibrium internuclear distance when the
molecule is not vibrating. When the molecule moves into vibrational states v = 0,1,2, it begins
to stretch, due to the anharmonicity of the potential, and r increases. Hence, the internuclear
distance (really root-mean-square internuclear distance
) will depend on the vibrational state
v. From the rotational constants B
0
and B
1
, the root-mean-square internuclear distance between
the H and Cl atoms in vibrational states v=0 and v=1,
and
respectively, can be
determined.
B
v
= h /8t
2
c
(Eq. 24)
Thus, we will experimentally determine the positions of all the fine-structures lines R(J'') and
P(J'') of the fundamental band for H
35
Cl. We will use this information to extract the
spectroscopic constants B
0
, B
1
, D
e
and
. We will supplement the fundamental line data with
additional spectroscopic data to determine
and B
e
. Finally, we will invoke our
molecular model for HCl and determine the constants k and r
e
,
, and
.
P a g e | 8
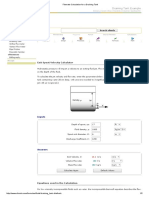
Procedure
Obtain a high resolution Infrared spectrum of HCl gas. You will use a 10 cm gas cell as pictured
below.
The cell will be filled with HCl gas via one of the methods listed:
i) Use a "lecture" gas bottle, as pictured on page one of this laboratory, to fill the cell
directly.
ii) Treat Sodium Chloride with concentrated Sulfuric Acid to produce the Hydrogen
Chloride gas:
NaCl(s) + H
2
SO
4
(l) HCl(g) + NaHSO
4
(s)
You can do this by dropping, very slowly, ~30 mL of conc. H
2
SO
4
into about 5g of the
solid NaCl as pictured below.
P a g e | 9
Your laboratory instructor will indicate which method of filling the gas cell will be used.
Once your gas cell is filled with HCl, your laboratory instructor will demonstrate the use of the
IR spectrometer. Take the spectrum with an appropriate resolution to see the fine structure in the
fundamental line. Measure the positions of R(J'') and P(J'') spectral lines for H
35
Cl.
P a g e | 10
Data Analysis
The following calculations will require linear least squares analyses that are of fairly high
precision. Track your significant figures closely and make sure you perform an error
analysis for each directly measured quantity.
1. From your data, determine the spectroscopic constants:
B
0
, B
1
and
2. Use the supplemental overtone data to determine the spectroscopic constants:
and
3. Use the supplemental data for rotational constants to determine the spectroscopic constant
B
e
.
4. Determine the molecular parameters k, r
e
,
and
.
5. Use the following data to determine k for alternate isotopic substitutions of HCl.
Molecule v
e
[cm
-1
]
D
35
Cl 2145.1630
D
37
Cl 2141.82
6. Use the following data to determine r
e
for alternate isotopic substitutions of HCl.
Molecule B
e
[cm
-1
]
H
37
Cl 10.578
D
35
Cl 5.448794
D
37
Cl 5.432
P a g e | 11
References
Arnaiz, Francisco J. "A Convenient Way to Generate Hydrogen Chloride in the Freshman Lab"
J. Chem. Ed. 72 (1995) 1139.
Herzberg, G. Spectra of Diatomic Molecules Van Nostrand, Princeton, New Jersey, 1950.
Levine, Ira N. Physical Chemistry McGraw-Hill, Boston, 2009.
Levine, Ira N. Quantum Chemistry Prentice Hall, Englewood Cliffs, New Jersey, 1991.
Meyer, Charles F. and Levin, Aaron A. "On the Absorption Spectrum of Hydrogen Chloride"
Phys. Rev. 34 (1929) 44.
Pickworth, J. and Thompson, H.W. "The Fundamental Vibration-Rotation Band of Deuterium
Chloride" Proc. R. Soc. London Ser. A 218 (1953) 218.
Rank, D.H.; Eastman, D.P; Rao, B.S.; and Wiggins, T.A. "Rotational and Vibrational Constants
of the HCl
35
and DCl
35
Molecules" J. Opt. Soc. Am. 52 (1961) 1.
Sime, Rodney J. Physical Chemistry: Methods, Techniques, and Experiments Saunders College
Publishing, Philadelphia, 1990.
Tipler, Paul A. Physics Worth Publishers, New York, 1976.
Van Horne, B.H. and House, C.D. "Near Infrared Spectrum of DCl" J. Chem. Phys. 25 (1956)
56.
You might also like
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5794)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (895)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (588)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (345)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (121)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (400)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- Sl. No. Name of The Sponsor Location Contact NoDocument12 pagesSl. No. Name of The Sponsor Location Contact NoSampath R0% (1)
- Brosur Evo FranklinDocument2 pagesBrosur Evo FranklinFargan ThanNo ratings yet
- Essential Variables API 1104Document4 pagesEssential Variables API 1104Madidj_2014No ratings yet
- EPA Method 3101Document3 pagesEPA Method 3101skrim240No ratings yet
- Semester I: Room Timetable - FST CSL1Document2 pagesSemester I: Room Timetable - FST CSL1skrim240No ratings yet
- Writing The Conclusion To Your Literature Review: See Example BelowDocument1 pageWriting The Conclusion To Your Literature Review: See Example Belowskrim240No ratings yet
- R 3528Document3 pagesR 3528skrim240No ratings yet
- Semester I: Course Timetable - CHEM 2470, Introduction To Analytical ChemistryDocument3 pagesSemester I: Course Timetable - CHEM 2470, Introduction To Analytical Chemistryskrim240No ratings yet
- Semester I: Room Timetable - FFA DAEE Computer LabDocument1 pageSemester I: Room Timetable - FFA DAEE Computer Labskrim240No ratings yet
- ZincEDTA PDFDocument22 pagesZincEDTA PDFskrim240No ratings yet
- Summer: Course Timetable - CHEM 2015, SpectrosDocument1 pageSummer: Course Timetable - CHEM 2015, Spectrosskrim240No ratings yet
- Summer: Course Timetable - CHEM 2260, Basic Organic Chemistry IDocument1 pageSummer: Course Timetable - CHEM 2260, Basic Organic Chemistry Iskrim240No ratings yet
- Summer: Course Timetable - CHEM 2360, Basic Physical ChemistryDocument1 pageSummer: Course Timetable - CHEM 2360, Basic Physical Chemistryskrim240No ratings yet
- K A Ó B K: Kinetics of Multiple ReactionsDocument5 pagesK A Ó B K: Kinetics of Multiple Reactionsskrim240No ratings yet
- Chemical Examples For The Fit Equations: ExampleDocument11 pagesChemical Examples For The Fit Equations: Exampleskrim240No ratings yet
- Lecture 9 Introductory Kinetics PDFDocument78 pagesLecture 9 Introductory Kinetics PDFskrim240No ratings yet
- LABILEDocument5 pagesLABILEskrim240No ratings yet
- Beers LawDocument5 pagesBeers Lawskrim240No ratings yet
- Experiment 3 Synthesis, Characterization and Ligand Exchange Equilibria of Potassium Tris (Oxalato) Ferrate (Iii)Document7 pagesExperiment 3 Synthesis, Characterization and Ligand Exchange Equilibria of Potassium Tris (Oxalato) Ferrate (Iii)skrim240100% (1)
- Civ V BNW PC Online Manual EngDocument17 pagesCiv V BNW PC Online Manual Engskrim240No ratings yet
- Chlorine Thiosulfate TitrationDocument3 pagesChlorine Thiosulfate Titrationskrim240No ratings yet
- A Volumetric Analysis (Redox Titration) of Hypochlorite in BleachDocument5 pagesA Volumetric Analysis (Redox Titration) of Hypochlorite in Bleachskrim240No ratings yet
- Module 2: Infrared SpectrosDocument5 pagesModule 2: Infrared SpectrosAngela ReyesNo ratings yet
- Solvent Sbps 1425 - HPCLDocument2 pagesSolvent Sbps 1425 - HPCLBharat ChatrathNo ratings yet
- Centricut Catalogue 2019 - Laser - MazakDocument42 pagesCentricut Catalogue 2019 - Laser - MazakIvan YordanovNo ratings yet
- Remediation Project 1Document26 pagesRemediation Project 1api-508660724No ratings yet
- Module 6 - Ash, Water, Extractive and Crude Fiber Content DeterminationDocument23 pagesModule 6 - Ash, Water, Extractive and Crude Fiber Content DeterminationPRINCESS NOELLEE TRAYVILLANo ratings yet
- Spin Atomic Model: Role of Electron Spin in Forming Atoms and MoleculesDocument8 pagesSpin Atomic Model: Role of Electron Spin in Forming Atoms and MoleculesGhanshyam JadhavNo ratings yet
- IOGP S-616 - 2022 - Supp. Specification To API SPEC 5L & ISO 3183 Line PipeDocument187 pagesIOGP S-616 - 2022 - Supp. Specification To API SPEC 5L & ISO 3183 Line PipeFerryNo ratings yet
- Magnetic Particle Test (MT) : Level IiDocument18 pagesMagnetic Particle Test (MT) : Level IiAhmed El Morsy100% (1)
- D 3002 DesignDocument20 pagesD 3002 DesignmargaretramosNo ratings yet
- A Simplified Method For The Cultivation of Extreme Anaerobic Archaea Based SULFIDE 2000 !!!!Document6 pagesA Simplified Method For The Cultivation of Extreme Anaerobic Archaea Based SULFIDE 2000 !!!!Vera Brok-VolchanskayaNo ratings yet
- مستند بلا عنوانDocument4 pagesمستند بلا عنوانHamzah A. LaftaNo ratings yet
- Engi 9625 Assignment 1Document6 pagesEngi 9625 Assignment 1nuvanNo ratings yet
- Capsules and Its Technology An OverviewDocument8 pagesCapsules and Its Technology An OverviewRonald Reyes LeonNo ratings yet
- Lab 4: An Acyclic Process ObjectivesDocument1 pageLab 4: An Acyclic Process ObjectivesFnur FatihahNo ratings yet
- Vacancy-Induced Fano Resonances in Zigzag Phosphorene NanoribbonsDocument9 pagesVacancy-Induced Fano Resonances in Zigzag Phosphorene NanoribbonsAntonio GaleoteNo ratings yet
- EVS UNIT 2 NotesDocument21 pagesEVS UNIT 2 NotesNivashini VindhyaNo ratings yet
- GEE ElectrodeDocument17 pagesGEE ElectrodeSatyamEngNo ratings yet
- Units Earned Exam % No. of Test Items: ST NDDocument1 pageUnits Earned Exam % No. of Test Items: ST NDkenneth senadosNo ratings yet
- Certificate of Analysis: Product Number: H7250 Batch Number: WXBC3017VDocument1 pageCertificate of Analysis: Product Number: H7250 Batch Number: WXBC3017VAnonymous pCIauPOGNo ratings yet
- International Journal of Mining, Reclamation and EnvironmentDocument17 pagesInternational Journal of Mining, Reclamation and EnvironmentMargaritha Alexanderina FrancisNo ratings yet
- CuproBraze 34Document2 pagesCuproBraze 34Vinod SaleNo ratings yet
- Green Roof Energy SimulationDocument11 pagesGreen Roof Energy SimulationAnas Khalid TahirNo ratings yet
- Pharmaceutical Industry BR 9108227 en Us 2102 1Document12 pagesPharmaceutical Industry BR 9108227 en Us 2102 1PhongNo ratings yet
- Design For Temperature-Controlled Solar Heated Chick BrooderDocument7 pagesDesign For Temperature-Controlled Solar Heated Chick Brooderchristian emeka okaforNo ratings yet
- Lab Manual 02Document158 pagesLab Manual 02Stephen VivekNo ratings yet
- MUY COMPLETO - CNC CNF Towards The Scale-Up Production of Cellulose Nanocomposites Using Melt ProcessingDocument63 pagesMUY COMPLETO - CNC CNF Towards The Scale-Up Production of Cellulose Nanocomposites Using Melt ProcessingDerek ZoolanderNo ratings yet
- Flowrate Calculation For A Draining TankDocument2 pagesFlowrate Calculation For A Draining TankAnonymous bHh1L1No ratings yet