Professional Documents
Culture Documents
Modul Galus Chem 2014
Uploaded by
Juni FarhanaCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Modul Galus Chem 2014
Uploaded by
Juni FarhanaCopyright:
Available Formats
MODUL CHEMISTRY 2014
SUCCESS IN CHEMISTRY SPM 2014
SMK DR MEGAT KHAS
KEEP FIGHTING TILL THE END NEVER GIVE UP!!!
0
MODUL CHEMISTRY 2014
SET 1
ATOMIC STRUCTURE EMPRICAL FORMULAE
MODUL CHEMISTRY 2014
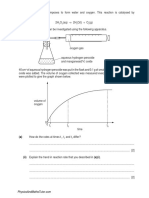
EXPERIMENT INVOLVING HEATING AND COOLING CURVE-NAPHTHALENE SPM ZONE
EXPERIMENT NAPHTALENE- study the precaution during experiment: Favourite question:
i- What is the purposed of using water bath in the experiment because naphthalene is flammable to ensure an even heating ii- How to ensure the naphthalene undergoes even heating - stir the naphthalene continuously iii. How to avoid supercooling - Stir the substance/ naphthalene continuously
GRAPH OF HEATING AND COOLING CURVE Favourite question: i- State the meaning of Melting point?
is temperature at which solid changes to liquid ii- Why temperature remains constant from t1 to t2 Heat absorbed to overcome the intermolecular forces iii- State the melting point/ What is the melting point of naphthalene 78 (refer to the graph given) iv State the physical state between t1 to t2 solid and liquid ( refer to graph given)
Temperature / C
Suhu / C A T t
2
v. Why temperature remains constant from t1 to t2 Heat loss to the surroundings is exactly balanced by the heat enegy liberated as the t particles attract one another to form a solid. 1 vi. Type of particles v- Draw the arrangement of particles of naphthalene at point t3 and t2
C / t Time min 3 Ma sa / min
MODUL CHEMISTRY 2014
ATOM, MOLECULES AND ION Favourite question: i- State the type of particles atom/ molecules / ion ii.- What is physical state of magnesium at room temperature solid iii- State the type of particles of magnesium at room temperature atom SPM ZONE
The sub atomic particles in atom : proton, electron, neutron The meaning of proton : The number of protons in its atoms The meaning of nucleon number: The total number of neutrons and protons in its atom. State the meaning of isotopes the atom with same number of protons but different number of neutrons/ neucleon
The uses of isotopes:- iodine 131- used in thyroid disease (medicine) cobalt 60- cancer treatment carbon 14 to estimate age of fossil (archaeology) phosporus-32: as fertilizer (agricultural) uranium 235 : nuclear in power stations State the meaning of valence electron are electron in the outermost occupied shell i. Write the electron arrangement of P 2.5 ii. Draw the electron arrangement of atom P
Atom P Q R S T Proton Number 7 10 17 17 19 Nucleon number 14 20 35 37 39
iii.Draw the electron arrangement of ion P
iv- State the number of neutron of atom P 7
MODUL CHEMISTRY 2014
TRIAL CHEMISTRY SPM 1
Diagram 1 shows the cooling curve of acetamide beginning from 90o C. Rajah 1 menunjukkan graf penyejukan asetamida bermula daripada 90oC.
Temperature / C Suhu / C
90 A
C 0 t1
DIAGRAM 1 RAJAH 1 Based on Diagram 1, answer the following questions. Berdasarkan Rajah 1, jawab soalan-soalan berikut. (a) What is the type of particles in acetamide? Apakah jenis zarah dalam asetamida? . [1 mark] (b) State the time when acetamide starts to freeze. Nyatakan masa apabila asetamida mula membeku. . [1 mark] (c) State the freezing point of acetamide. Nyatakan takat beku bagi asetamida. .
t2
t3
Time / min Masa / min
MODUL CHEMISTRY 2014
(d) Why does the temperature of acetamide remains constant from t1 to t2? Mengapa suhu asetamida tidak berubah dari t1 hingga t2? . .[ 2 MARK] (e) Draw the arrangement of particles of acetamide at points A and C in the spaces provided. Lukiskan susunan zarah bagi asetamida pada titik A dan C di ruangan yang disediakan.
Point A Titik A
Point C Titik C
[2 marks]
(f) State the physical state of acetamide at these points: Nyatakan keadaan fizik asetamida pada masa: (i) 0 to t1: (ii) t2 to t3: [2 marks]
MODUL CHEMISTRY 2014
MODUL CHEMISTRY 2014
The table below shows four substances with their respective chemical formula. Substance Iodine Potassium chloride Naphthalene Magnesium Chemical formula I2 KCl C10H8 Mg [3 marks]
(a) State one substance from the table above that exists as (i) atoms.
_________________________________________________________________ (ii) molecules. _________________________________________________________________ (iii) ions. _______________________________________________________________________
MODUL CHEMISTRY 2014
3 Diagram 3.1 shows the heating curve of solid naphthalene, C10H8. Rajah 3.1 menunjukkan lengkung pemanasan pepejal naftalena, C10H8. Temperature / 0C
T1
B C
T0 A
0
Diagram 2.1 Rajah 2.1
Time / s
(a)
(i)
Name the process involved in this experiment. Namakan proses yang terlibat dalam eksperimen ini. .............................................................................................................. [1 mark] [1 markah ]
(ii)
State the type of particle present in naphthalene, C10H8. Nyatakan jenis zarah yang terdapat dalam naftalena, C10H8 . ......................................................................................................... [1 mark] [1 markah]
(b)
Explain why there is no change in temperature from B to C Terangkan mengapa tiada perubahan suhu dari B ke C ................................................................................................................................... .................................................................................................................................. [2 marks] [2 markah]
MODUL CHEMISTRY 2014
(c) State how the movement of naphthalene particles changes from C to D during heating. Nyatakan bagaimana pergerakan zarah-zarah naftalena berubah semasa pemanasan dari C ke D. ................................................................................................................. [1 mark] [1 markah] (d) Diagram 2.2 shows the atomic model proposed by Neils Bohr. Rajah 2.2 menunjukkan model atom yang dicadangkan oleh Neils Bohr.
X Y
Diagram 2.2 Rajah 2.2 i ) Name X and Y. Namakan X dan Y. X : ...................................... Y : .......................................... [1 mark] [1 markah] ii) Which subatomic particles are involved in a chemical reaction? Zarah-zarah sub-atom yang manakah terlibat dalam tindak balas kimia? ....................................................................................................... [1 mark] [1 markah]
MODUL CHEMISTRY 2014
(e) Table 2 shows the number of protons and neutrons of four different atoms. Jadual 2 menunjukkan bilangan proton dan neutron bagi empat atom yang berlainan.
Atom W X Y Z
Number of protons Bilangan proton 16 16 3 19
Number of neutrons Bilangan neutron 17 16 4 20
i)
Which atoms are isotopes? Atom-atom yang manakah adalah isotop? ................................................................................................. [1 mark] [1 markah]
ii)
Give a reason for your answer in (e) (i). Berikan satu sebab bagi jawapan anda di (e) (i).
..................................................................................................................... ..................................................................................................................... [2 marks] [2 markah] TRIAL KELANTAN SPM 2007 1. Table 1 shows proton number and nucleon number for atom W, X and Y. Jadual 1 menunjukkan nombor proton dan nombor nukleon bagi atom W, X dan Y. Atom W X Y Proton number / Nombor Proton 11 17 17 TABLE / JADUAL 1 Nucleon number / Nombor Nukleon 23 35 37
10
MODUL CHEMISTRY 2014
(a) (i) What is meant by proton number? Apakah yang dimaksudkan dengan nombor proton ? ............................................................... ......................................... (ii) What is the number of neutron in atom W ? Apakah bilangan neutron dalam atom W? ................................................................................... (b) Which atoms are isotopes? Explain why. Atom atom yang manakah adalah isotop? Jelaskan mengapa. .................................................................................................................................. ........................................................................................................................... (c) Write the electron arrangement of atom X . Tuliskan susunan elektron bagi atom X. ............................................................................................................. [1 mark] [2 marks] [1 mark] [1 mark]
(d)(i) Draw the electron arrangement of atom Y. In your diagram , show the number of protons and the number of neutrons in the nucleus. Lukiskan susunan elektron atom Y. Dalam rajah anda, tunjukkan bilangan proton dan bilangan neutron dalam nukleus.
[2 marks] (ii) What is the number of valence electron of atom Y? Apakah bilangan elektron valen bagi atom Y? ...........................................................................................................................[1 mark] Write the formula of ion Y. Tuliskan formula ion Y. ................................................................................................................................[1 mark] (e) Write the symbol of atom X in the form of Tuliskan simbol bagi atom X dalam bentuk
A Z A Z
X. X.
....................................................................................................................[1 mark]
11
MODUL CHEMISTRY 2014
TRIAL PAHANG SPM . a) Atom S T U V Number of protons Bilangan proton 16 16 17 18 Number of neutrons Bilangan neutron 17 18 18 17 Table 1
(i)
What is meant by the proton number and nucleon number of an atom? Apakah yang dimaksudkan dengan nombor proton dan nombor nukleon bagi atom? Proton number Nombor proton .............................................................................................. [1 mark] Nucleon number Nombor nucleon
.. [1 mark] A (ii) Represent atom S in the form of Z X Tuliskan atom S dengan perwakilan atom unsur
A Z
.. [1 mark] (iii) Which of the atoms are isotopes? Atom-atom yang manakah isotop? . [1 mark] (iv) State a reason for your answer in (d)(iii) Nyatakan alasan bagi jawapan anda di (d)(iii)
[1 mark]
12
MODUL CHEMISTRY 2014
EMPRICAL FORMULAE: EXPERIMENT OF MAGNESIUM OXIDE AND COPPER OXIDE
copper oxide
Dry hydrogen gas
EXPERIMENT 1
EXPERIMENT II
i- Why must magnesium ribbon be cleaned with sandpaper? To remove the oxide layer on the surface of magnesium oxide ii- Explain why the crucible must be closed immediately when the magnesium ribbon starts to burn? To prevent the white fumes of Magnesium oxides from escaping iii- Explain why the crucible lid must be opened at intervals during the experiment? To allow oxygen in the air to enter into the crucible for complete combustion iv. How to make sure all the magnesium has reacted completelywith oxygen? Repeating the heating, cooling and weighing process until a constant mass is achieved v. Why the empirical formula of cooper oxide can not determined using this technique? because the copper oxide is less reactive metal Mass(g) 54.6 59.4 62.6
Mass of crucible + lid Mass of crucible + lid + magnesium ribbon Mass of crucible + lid + magnesium oxide vi- Calculate the mass of magnesium 59.4 54.6 = 4.8 g vii. Calculate the mass of oxygen 62.6 59.4 = 3.2 g
13
MODUL CHEMISTRY 2014
copper oxide
Dry hydrogen gas
Asbestos crucible
EXPERIMENT 1
i-. Suggest two substances to produce the hydrogen gas in this experiment
Zinc and Hydrochloric acid ii- Name the substance used to dry hydrogen gas Calcium chloride iii- The combustion tube must be filled up with hydrogen gas. Describe how to confirm that hydrogen gas has filled up the combustion tube. Let the dry hydrogen gas flow into the combustion tube for few minutes. The collected gas is tested with glowing wooden splinter until no pop sound is heard indicating that hydrogen gas has been removed. iv- Explain why the hydrogen gas is still passed through the apparatus during the cooling of the combustion tube. To prevent the metal formed from being oxidized by hydrogen in air Mass of combustion tube + asbestos crucible Mass of combustion tube + asbestos crucible + M oxide Mass of combustion tube + asbestos crucible + M [Relative atomic mass: M,207; O,16] a) Calculate the mass of i) M 56.70 54.63 = 2.07 g = 54.63 g = 58.30 g = 56.70 g
ii) Oxygen 58.30 56.70 = 1.6g
b) Calculate the mole ratio of atom M and oxygen atom M = 2.07 g = 0.1 mol 207 atom oxygen = 1.6 g = 0.1 mol 16
EMPRICAL FORMULA AND MOLECULAR FORMULA c) Determine the empirical formula of M oxide simplest ratio M = 0.1 = 1 simplest ratio O = 0.1 =1 0.1 14 0.1 MO
MODUL CHEMISTRY 2014
i- State the meaning of Emprical Formula The chemical formula which shows the simplest ratio of the number of atoms for each element in a compound ii- State the meaning of Molecular formulae Formula that shows the actual number of each types of atom in a molecule for each element in a compound iii Determine the empirical formular of the following
Mass of carbon: Jisim karbon Mass of oxygen: Jisim oksigen Mass of hydrogen: Jisim hydrogen Relative molecular mass: Jisim molekul relative 26.67 g 71.11 g 2.22 g 90 carbon: 40.00% Karbon Hydrogen: 6.66 % Hidrogen Oxygen: 53.33 % hydrogen Relative molecular mass: 180 Jisim molekul relative Relative atomic mass of H = 1, C = 12 and O = 16
Relative atomic mass of H = 1, C = 12 and O = 16
i-
Element Mass (g) Number of moles
Carbon 26.67 26.67 12
Oxygen 71.11 71.11 16
Hydrogen 2.22 2.22 1
Ratio of number of moles Simplest Ratio
2.22 2.22 = 1 2.22
4.44 4.44 = 2 2.22
2.22 2.22 =1 2.22
Emprical Formula = CO2H ii- Determine the molecular formula of this compound. (Emprical formula )n= Relative Molecular mass (CO2H)n = 90 (12+2(16)+1)n = 90 (12+32+1)n =90 45n = 90 n= 90 =2 45 Molecular formula = (CO2H)2 = C2O4H2
15
MODUL CHEMISTRY 2014
iii.
Element Mass (g) Number of moles Carbon 40.00 Hydrogen 6.66 Oxygen 53.33
Ratio of number of moles
Emprical Formula = ii- Determine the molecular formula of this compound.
TRIAL PAHANG SPM 2011 (b) An organic compound Q consists of 64.87% of carbon, 13.51% of hydrogen and 21.62% of oxygen. [Relative atomic mass: H,1 ; C,12 ;O, 16] Suatu sebatian organik Q mengandungi karbon 64.87%, hidrogen 13.51% dan oksigen 21.62%. [Jisim atom relatif: H,1 ; C,12 ; O, 16] (i) Determine the empirical formula of compound Q. Tentukan formula empirik bagi sebatian Q.
[3 marks] (ii) If the relative molecular mass of compound Q is 74, find the molecular formula of compound Q. Jika jisim molekul relatif bagi sebatian Q adalah 74, cari formula molekul bagi sebatian Q.
16
MODUL CHEMISTRY 2014
TRIAL SPM
Diagram 3 shows the set-up of apparatus to determine the empirical formula of magnesium oxide. Rajah 3 menunjukkan susunan radas bagi menentukan formula empirik magnesium oksida.
Diagram 3 Rajah 3 The following are the results of the experiment Di bawah adalah keputusan daripada eksperimen Mass of crucible + Jisim mangkuk pijar Mass of crucible + Jisim mangkuk pijar lid + penutup lid + magnesium ribbon + penutup + pita magnesium
54.6 g 59.4 g 62.6 g
Mass of crucible + lid + magnesium oxide Jisim mangkuk pijar + penutup + magnesium oksida [Relative atomic mass: Mg,24; O,16] [Jisim atom relatif: Mg, 24; O, 16]
(a)
Calculate the mass of Kira jisim bagi
(i)
Magnesium [1 marks] [1markah]
(ii)
Oxygen [1 marks] [1markah]
(b)
Calculate the number of moles of Kira bilangan mol bagi
(i)
Magnesium [1 marks] [1markah]
(ii)
Oxygen [1 marks] [1markah
17
MODUL CHEMISTRY 2014
(c) Determine the empirical formula of magnesium oxide formed. Tentukan formula empirik magnesium oksida yang terbentuk.
[1 mark] [1 markah] (d) Write the chemical equation between magnesium and oxygen in this experiment. Tuliskan persamaan kimia bagi tindak balas magnesium dan uksigen dalam eksperimen ini. .. [1 mark] [1 markah] (e) Why must magnesium ribbon be cleaned with sandpaper? Mengapakah magnesium perlu dibersihkan dengan kertas pasir ? ... .. [1 mark] [1 Markah] (f) Explain why Terangkan mengapa (i) the crucible must be closed immediately when the magnesium ribbon starts to burn? Mangkuk pijar perlu ditutup serta merta apabila pita magnesium mula terbakar ? .. ...................................................................... [1 mark] [1 Markah] the crucible lid must be opened at intervals during the experiment? Penutup mangkuk pijar dibuka sekali sekala semasa eksperimen ? .. .. [1 mark] [1 Markah] (g) How to make sure all the magnesium has reacted completely with oxygen? Bagaimana memastikan semua magnesium telah lengkap bertindak balas dengan oksigen? .. ... [1 mark] [1 Markah]
(ii)
18
MODUL CHEMISTRY 2014
TRIAL CHEMISTRY KEDAH SPM 2008 2. Diagram 2 shows the apparatus set-up to determine the empirical formula of oxide metal M. Rajah 2 menunjukkan susunan radas untuk menentukan formula empirik bagi oksida logam M.
M oxide Oksida M Dry hydrogen gas Gas hidrogen kering Combustion tube Tiub pembakaran
DIAGRAM 2 (a) (i) Name two chemicals used to prepare hydrogen gas in the laboratory. Namakan dua bahan yang digunakan untuk menyediakan gas hidrogen dalam makmal. ....................................................................................................................... [2 marks] (ii) Write a chemical equation for the reaction in (a)(i). Tuliskan persamaan kimia bagi tindak balas di (a)(i). ... [1 mark] (b) Table 2 shows the result of the experiment: Jadual 2 menunjukkan keputusan eksperimen itu: Mass of combustion tube + asbestos paper Jisim tiub pembakaran + kertas asbestos Mass of combustion tube + asbestos paper + M oxide before heating Jisim tiub pembakaran + kertas asbestos + oksida M sebelum pembakaran Mass of combustion tube + asbestos paper + product after heating Jisim tiub pembakaran + kertas asbestos + hasil selepas pembakaran [Relative atomic mass of O=16, M=55] (i) Determine the empirical formula of M oxide. Tentukan formula empirik bagi oksida M. 39.25 g 47.95 g 44.75 g
Asbestos paper Kertas asbestos
[3 marks]
19
MODUL CHEMISTRY 2014
(c) State one precaution that must be taken when carrying out the experiment. Nyatakan satu langkah berjaga-jaga yang mesti diambil semasa menjalankan eksperimen itu. ................................................................................................................................. [1 mark] (d) Can the empirical formula of magnesium oxide be determined by the same method? Explain your answer. Bolehkah formula empirik bagi magnesium oksida ditentukan dengan kaedah yang sama? Terangkan jawapan anda. ................................................................................................................................. ................................................................................................................................. [2 marks] TRIAL JOHOR SPM 2008 2 Diagram 2 shows the set up of an experiment to determine the empirical formula of copper(II) oxide. Rajah 2 di bawah menunjukkan susunan radas bagi eksperimen yang dijalankan untuk menentukan formula empirik kuprum(II) oksida.
Copper(II) oxide Kuprum(II) oksida
H2
Heat Panaskan Anhydrous calcium chloride, CaCl2 Kalsium klorida kontang
DIAGRAM 2 (a) The empty box in Diagram 2 is the set up of apparatus to prepare the hydrogen gas. Complete the diagram. Di dalam kotak kosong pada Rajah 2 di atas merupakan susunan radas untuk menyediakan gas hidrogen. Lengkapkan Rajah 2 tersebut. [2 marks] (b) State the function of the anhydrous calcium chloride? [ 1 mark] Nyatakan fungsi kalsium klorida kontang ? ..........................................
20
MODUL CHEMISTRY 2014
(c) Write a chemical equation to show the reaction taking place in (a). Tuliskan persamaan kimia untuk tindak balas yang berlaku di (a). ............................................... [2 marks] (d) State one precaution that need be taken before the copper(II) oxide is heated. Nyatakan satu langkah berjaga-jaga yang perlu diambil sebelum memanaskan kupum(II) oksida. .. [1 mark] (e) Table 2 shows the results of an experiment carried out by a student. Jadual 2 menunjukkan keputusan bagi eksperimen yang dilakukan oleh pelajar. Mass of combustion tube + porcelain boat Jisim tiub pembakaran + bot porselin Mass of combustion tube + porcelain boat + copper(II) oksida Jisim tiub pembakaran + bot porselin + kuprum(II) oksida Mass of combustion tube + porcelain boat + copper Jisim tiub pembakaran + bot porselin + kuprum Mass of copper Jisim kuprum Mass of oxygen Jisim oksigen TABLE 2 (i) Complete Table 2. Lengkapkan Jadual 2. [1 mark] (ii) Calculate the number of moles of copper and oxygen in this reaction. [Relative atomic mass for Cu = 64, O = 16] Kirakan bilangan mol kuprum dan oksigen dalam tindak balas ini. [Jisim atom relatif bagi Cu= 64, O = 16] 30.24g 32.26g 31.86g
[2 marks] (iii) Determine the empirical formula of copper(II) oxide. Tentukan formula empirik kuprum(II) oksida
21
MODUL CHEMISTRY 2014
(f) Give an example of another metal, which the empirical formula of that metal oxide can be determined by the same method. Berikan satu contoh logam lain di mana formula empirik bagi logam oksidanya boleh ditentukan dengan kaedah yang sama seperti di atas. .... [1 mark] TRIAL JOHOR 2 Diagram 2 shows the set -up apparatus of an experiment to determine the empirical formula of copper (II) oxide. Rajah 2 menunjukkan susunan radas eksperimen untuk menentukan formula empirik bagi kuprum (II) oksida.
Hydrogen gas Gas hidrogen
Copper(II) oxide Kuprum(II) oksida
Porcelain dish Mangkuk porselin Heat Panaskan
Anhydrous calcium chloride, CaCl2 Kalsium klorida kontang, CaCl2
Diagram 2 (a) What is the meaning of chemical formula? Apakah maksud formula kimia? ................................................ [1 mark] Name an acid and metal that can be used to prepare hydrogen gas in this experiment. Namakan suatu asid dan logam yang boleh digunakan untuk menyediakan gas hidrogen dalam eksperimen ini. [1 mark] .................................................. Write a balanced chemical equation for the reaction between the acid and the metal in (b) (i). Tuliskan persamaan kimia yang berimbang untuk tindak balas yang berlaku di antara asid dan logam di (b) (i). ........................................
(b)
(i)
(ii)
22
MODUL CHEMISTRY 2014
(c) State one precaution that must be taken before the copper (II) oxide is heated. Nyatakan satu langkah berjaga-jaga yang perlu diambil sebelum kupum(II) oksida dipanaskan. ..... [1 mark] [1 markah] Table 2 shows the results of an experiment carried out by a student. Jadual 2 menunjukkan keputusan eksperimen yang dilakukan oleh pelajar. Mass of combustion tube + porcelain dish Jisim tiub pembakaran + mangkuk porselin Mass of combustion tube + porcelain dish + copper (II) oxide Jisim tiub pembakaran + mangkuk porselin + kuprum(II) oksida Mass of combustion tube + porcelain dish + copper Jisim tiub pembakaran + mangkuk porselin + kuprum 30.24g 32.26g 31.86g
(d)
(i)
Calculate the number of moles of copper in this reaction. [Relative atomic mass : Cu = 64] Kirakan bilangan mol kuprum dalam tindak balas ini. [Jisim atom relatif :i Cu= 64]
[1 mark] [1 markah]
(ii)
Calculate the number of moles of oxygen in this reaction. [Relative atomic mass : O = 16] Kirakan bilangan mol kuprum dalam tindak balas ini. [Jisim atom relative: O= 16]
[1 mark] [1 markah] (iii) Determine the empirical formula of copper (II) oxide. Tentukan formula empirik kuprum (II) oksida. [2 marks]
23
MODUL CHEMISTRY 2014
(e)
Name another metal oxide which uses the same method to determine the empirical formula. Namakan suatu oksida logam yang lain di mana formula empiriknya ditentukan dengan kaedah yang sama. .... [1 mark] [1 markah]
(f)
M is a reactive metal. Suggest a method to determine the empirical formula of the oxide of M. M merupakan suatu logam reaktif. Cadangkan satu kaedah untuk menentukan formula empirik bagi oksida M. ........................................................................................................................................... [1 mark] [1 markah]
24
MODUL CHEMISTRY 2014
SET 2
PERIODIC TABLE CHEMICAL BONDING
25
MODUL CHEMISTRY 2014
PERIODIC TABLE A B E F C D G
i- State one element that exist as metal? B ii- State element that exist as mono atom A iii- Choose an element that is a halogen/diatomic D or G iv- shaded the Transition Elements in the table above vi- State the name that represent vertical column Group vii- State the name represent horizontal column Period viii- State one special characteristic of transition elements formed coloured ion / used as catalyst x- State the element that can form ionic compound B and D / E and C xi- State the element that can form covalent compound C and D / C and G xii- State the element that can form amphoteric oxides F xiii- Arrange A, B,C,D,E, F, and G according to the increase in size of the atoms A, D, C.B, G, F, E xiiii- State the element that react vigorously with water B x- Write the chemical equation when element in (xiii) react with water B + H2O BOH + H2 xi. State the formulae ion for E E2+
26
MODUL CHEMISTRY 2014
POSITION OF ELEMENT IN GROUP Favourite question:
Group 17 since it has 7 valence electron occupied in outermost shell Period 3 since it has 3 shell occupied by electron
GROUP 18 Favourite question: i- Why group 18 is chemically inert/ not reactive? because group 18 has stable octet arrangement which is do not share, lose or release electron ii- State the uses of element in group 18 Helium to fill weather ballons Neon to fill advertising light bulbs Argon uses to fill electric bulbs
27
MODUL CHEMISTRY 2014
TRIAL JOHOR SPM 2013 3 Diagram 3 shows part of the Periodic Table of the Elements. D, E, G, L, M, and J do not represent the actual symbol of the elements. Rajah 3 menunjukkan sebahagian daripada Jadual Berkala Unsur. D, E, K, L, M dan J tidak mewakili simbol sebenar unsur-unsur berkenaan.
L
D
M J
G
Using the letters in the Periodic Table of the Elements in Diagram 3, answer the following questions. Dengan menggunakan huruf-huruf yang terdapat dalam Jadual Berkala pada Rajah 3, jawab soalansoalan berikut. (a) (i) State the position of element E in the Periodic Table. Nyatakan kedudukan unsur E dalam Jadual Berkala. . [2 marks] [2 markah] (ii) Choose the element which exhibit different oxidation numbers in its compounds. Pilih unsur yang mempunyai pelbagai nombor pengoksidaan di dalam sebatiannya.
(b)
..... [1 mark] [1 markah] Element D combines with element L to form a compound. Unsur D bergabung dengan unsur L untuk membentuk suatu sebatian. (i) Write the chemical formula of this compound. Tuliskan formula kimia bagi sebatian ini. ..................................................................... [1 mark] [1 markah] State one physical property of this compound. Nyatakan satu sifat fizik sebatian ini. . [1 mark
(ii)
28
MODUL CHEMISTRY 2014
(c) D and E have the same chemical properties. D dan E mempunyai sifat-sifat kimia yang sama. (i) Which element is more reactive? Unsur yang manakah yang lebih reaktif? ........................................................................................................................................... [1 mark] [1 markah] Explain your answer in (c) (i). Terangkan jawapan anda dalam (c) (i). . . [2 marks] [2 markah] Which element exists as diatomic molecules? Unsur yang manakah membentuk molekul dwiatom? ..[1 mark] [1 markah] (ii) Name the chemical bond in these diatomic molecules. Namakan ikatan kimia dalam molekul dwiatom ini. .................................................................. [1 mark] [1 markah]
(ii)
(d)
(i)
29
MODUL CHEMISTRY 2014
1 Diagram 1 shows the position of several elements P, Q, R, S, T, U and W. Rajah 1 menunujukkan kedudukan beberapa unsur-unsur P, Q, R, S, T, U and W.
P Q R S T V U W
DIAGRAM 1 RAJAH 1 Using the symbols P, Q, R, S, T, U and W, answer the following questions. Dengan menggunakan simbol P, Q, R, S, T, U dan W, jawab soalan-soalan (a) State the element which you would classify under Group 2 Nyatakan unsur yang boleh dikelaskan di bawah Kumpulan 2 .... [1 mark] [1 markah] (b) Write the electron arrangement of atom V. Tuliskan susunan elektron bagi atom V. .. [1 mark] [ markah] (c) Which of the elements has the biggest atomic radius? Unsur yang manakah mempunyai jejari atom paling besar? ... [1 markah] [1markah]
berikut.
(d)
(i) Compare the electronegativity of elements Q, T, V and U. Bandingkan keelektronegatifan bagi Q, T, V dan U. .. [1 mark] [1 markah]
30
MODUL CHEMISTRY 2014
(ii) Explain your answer in (b)(i). Terang jawapan anda di (b)(i). .. [2 marks] [2 markah] (e) State the ion formed by Q. Nyatakan ion yang terbentuk oleh Q. ............................................................................ [1 mark] [1 markah] (f) Compare the reactivity of Q and R. Bandingkan kereaktifan di antara Q dan R. .. [1 mark] [markah] (g) Why is W chemically unreactive? Kenapa W tidak reaktif secara kimia? ............................................................................... [1 mark] [1 markah ] (h) Give one special characteristic of S. Beri satu ciri istimewa bagi S. .... [1 mark] [ 1 markah]
31
MODUL CHEMISTRY 2014
3. Diagram 2 shows part of the Periodic Table of Elements. A, B, C, D, E, F, G and H do not represent the
actual symbol of the elements. A B E F C D G
DIAGRAM 2 Using the letters in the Periodic Table of Elements in Diagram 2, answer the following questions. (a) (i) Write the electron arrangement of atom F. ................................................ [1 mark] (ii) Give the formula of ion F. ................................................ [1 mark] (b) Arrange the elements A, B, C, D, E, F and G according to the atomic size in ascending order. ................................................... [1 mark] (c) Element A exists as a monoatomic gas. Explain why. ................................................... ............................................... [1 mark] (d) Element D is more reactive than element G. Explain why. ................................................................. ................................................................. [2 marks] (e) E reacts with D to form a compound. (i) What is the type of the compound formed? . ................................................................ [1 mark]
32
MODUL CHEMISTRY 2014
(ii) Draw the diagram of electron arrangement for the compound formed.
TRIAL TERENGGANU 4.Diagram 2 shows the symbols of atom for element U, V, W and X. Rajah 2 menunjukkan simbol- simbol atom bagi unsur U, V, W dan X.
7 3
12 6
19 9
20 10
DIAGRAM / RAJAH 2 (a) Write the electron arrangement of atom W. Tuliskan susunan elektron bagi atom W. ..............................................................................................................................[1 mark] (b) (i) Element U, V, W and X are placed in the same period in the Periodic Table. State the period. Unsur U, V, W dan X berada dalam kala yang sama dalam Jadual Berkala. Nyatakan kala itu.
.....................................................................................................................[1 mark] (ii) Compare the atomic size of element V and X. Bandingkan saiz atom unsur V dan X. .....................................................................................................................[1 mark] (iii) Explain your answer in (b) (ii) Terangkan jawapan anda dalam (b) (ii). ................................................................................................................................... ..................................................................................................................[2 marks ] (c) Element V can react with element W to form a compound. Unsur V boleh bertindak balas dengan unsur W membentuk satu sebatian. (i) Write the chemical formula for the compound. Tuliskan formula kimia bagi sebatian itu. ..................................................................................................................[1 mark ]
33
MODUL CHEMISTRY 2014
2 Figure 2.1 shows the chemical symbols which represent elements R, S and T. (Rajah 2.1 menunjukkan simbol kimia yang mewakili unsur-unsur R, S dan T.) 35 R 17 (a) (i) 12 S
39 T
6
FIGURE 2.1
19
Write the electron arrangement of atom T. (Tuliskan susunan elektron bagi atom T.) ... [1 mark] State the period and the group for element T in the Periodic Table. (Nyatakan kala dan kumpulan bagi unsur T dalam Jadual Berkala.) Period (Kala) : . Group (Kumpulan) : .............................. [2 marks]
(ii)
(b)
Atoms of R and S can react to form a compound. (Atom R dan atom S boleh bertindak balas untuk membentuk satu sebatian.) (i) Name the type of bond in the compound formed between atoms R and S. (Nyatakan jenis ikatan dalam sebatian yang terbentuk antara atom R dan atom S.) .. [1 mark] Give one physical property of the compound in (b)(i). (Beri satu sifat fizik bagi sebatian di (b)(i).)
(ii)
(c)
. [1 mark] Atoms of R and T can also react to form a compound. Draw the electron arrangement for the compound formed. (Atom R dan atom T juga boleh bertindak balas untuk membentuk satu sebatian. Lukiskan susunan elektron bagi sebatian yang terbentuk.)
34
MODUL CHEMISTRY 2014
35
MODUL CHEMISTRY 2014
CHAPTER 5 Favourite question: Formation of ionic and covalent compound Element Unsur W X Y Z Electron arrangement Susunan elektron 2.4 2.6 2.8.1 2.8.2
Using the information in Table 10.2, choose any two elements and explain how ionic compund and covalent compound are formed. i- ionic compound .( Z and X// Y and X) Z = 2.8.2 Formula ion : Z+2 Z+2 X-2 Z2X2 1. 2. 3. 4. 5. 6. 7. ZX X = 2.6 Formula ion : X-2
Atom Z releases two electrons to atom X and achieve a stable octet electron arrangement. Z 2+ formed//Z positive ion formed Atom X receives two electrons and achieve a stable octet electron arrangement. X 2- formed// X negative ion formed. Z 2+ and X 2- are attracted by a strong electrostatic forces // ionic bond Z and X form ionic compound with formula ZX
ii- covalent bond (W and X) W = 2.4 Formula ion : W-4 X = 2.6 Formula ion = X-2 W-4 X-2
W2X4 WX2 1.atom W needs 4 electrons to achieve a stable octet electron arrangement 2. Atom X needs 2 electrons to achieve a stable octet electron arrangement 3. One atom of W contribute four electrons to be shared with two atoms X 4. Two atoms X will contribute two electrons each to be share with one atom W 5. One atom W shares four pairs of electrons with two atoms X, forming two double covalent bonds // The formula of the compound is WX2/ or can deduce from the diagram
36
MODUL CHEMISTRY 2014
i- State the physical properties of ionic compound -conduct electricity in molten and aqueous solution - soluble in water - has higher melting point and boiling point - insoluble in organic solvent ii- State the properties of covalent compound -can not conduct electricity in both molten and aqueous solution - insoluble in water - has low melting point and boiling point -vaporize easily(meruap) - soluble in organic solvent Tolong ingat 2 properties sahaja bagi setiap ionic and covalent compound !!!
iii- State the meaning of anion the ion which has negative charge iv- State the meaning of cation the ion which has positive charge
37
MODUL CHEMISTRY 2014
TRIAL KEDAH SPM (a) Figure 9 shows the symbols which represent elements X and Y
Element X and Y react to form ionic bond. Describe the formation of an ionic compound [ 8 marks]
[ 8 marks] 38
MODUL CHEMISTRY 2014
39
MODUL CHEMISTRY 2014
b) Table 10.1 shows the electron arrangement diagram of compound X and Y. Jadual 10. menunjukkan gambarajah susunan elektron bagi sebatian X dan Y.
Compound Sebatian
Electron arrangement diagram Gambarajah susunan elektron
+
X
P+
R-
Table 10.1
Jadual 10.1
(i)
Based on Table 10.1, determine the types of compound X and Y. Berdasarkan Jadual 10.1, tentukan jenis sebatian X dan Y. [2 marks]
Compound X and compound Y have different physical properties. Choose one of the physical properties that can differentiate between compound X and Y Explain the difference in the physical properties between compound X and Y. Sebatian X dan sebatian Y mempunyai sifat fizik yang berbeza. Plih satu sifat fizik tersebut yang dapat membezakan antara sebatian X and Y. Terangkan perbezaan sifat fizik tersebut di antara sebatian X dan Y. [2 marks]
40
MODUL CHEMISTRY 2014
c) Table 10.2 shows the electron arrangement for atoms W, X, Y and Z. d) Jadual 10.2 menunjukkan susunan elektron bagi atom-atom W, X, Y and Z.
Element Unsur W X Y Z
Electron arrangement Susunan elektron 2.4 2.6 2.8.1 2.8.2 Table 10.2
Jadual 10.2
Using the information in Table 10.2, choose any two elements and explain how ionic compund and covalent compound are formed. Gunakan maklumat dalam Jadual 10.2, pilih mana-mana dua unsur dan terangkan bagaimana sebatian ion dan sebatian kovalen terbentuk
[10 marks]
41
MODUL CHEMISTRY 2014
SET 3
ELECTROCHEMISTRY
42
MODUL CHEMISTRY 2014
CHAPTER 6 Favourite question: i- State the meaning of electrolytes the chemical substances that can conduct electricity either in molten and aqueous solution ii- State the meaning of electrolysis the process whereby a compound is decomposed into its constituent elements when electric current passes through an electrolyte iii- Why ionic compound can conduct electricity in molten and aqueous solution because the ion are free to move to carry out the electric current iv- Why in solid state ionic compound cannot conduct electricity because they are held in fixed position therefore the ion are not free to move v- Why covalent compound cannot conduct electricity either in molten and aqueous solution because they do not contains ion which cannot carry electric current ANALYSING IN AQUEOUS SOLUTION
i- State the energy change in electrolysis cell Electrical change to chemical charge ii-State the application of electrolysis in industries -Extraction of ores metal - Purification of metals - Electroplating of metals iii.Why the colour of copper(II) sulphate is remain unchanged because the concentration of the blue Cu2+ ion remain unchanged since anode dissolved to form Cu2+ ion iv. State three factor affected the aqueous solution -position of ions in electrochemical series - concentration of ions - types of electrodes v.Why the ion Cu+2 selectively discharge at cathode since ion Cu+2 has lower position compared to H+ ion in electrochemical series vi. Why the ion Cl- selectively discharged at anode since ion Cl- has higher concentration compared to OH- ion in electrochemical series vii. How to confirm the gas is oxygen? Put the glowing wooden splinter inside the mouth of test tube. Relight glowing wooden splinter.
43
MODUL CHEMISTRY 2014
viii. How to identify the gas is hydrogen? Put the glowing wooden splinter near the mouth of test tube. Pop sound will heard. viiii. How to identify the gas is chlorine decolourised the blue litmus paper x. How to identify the iodine gas Flow the gas in test tube containing starch solution. white solution changes to dark blue solution xii. Observation involving oxygen gas and hydrogen gas The colorless bubbles gas released xiii. Observation involving chlorine gas the greenish yellow gas released with chocking smell xiiii. Observation involving iodine and bromine gas brown gas released with chocking smell xiv. State the observation of copper plate at cathode brown solid layer deposited at cathode/ cathode become thicker xv. State the observation of copper plate at anode anode dissolved in electrolyte/ anode become thinner xvi. State the observation of silver/lead plate at cathode grey solid layer deposited at cathode/ cathode become thicker xv. State the observation of silver/ lead plate at anode anode dissolved in electrolyte/ anode become thinner
ANALYSING OF VOLTAIC CELL
i- State the energy change in voltaic cell Chemical change to electric charge ii- What is function of salt bridge to allow the movement of ion so that the electric circuit is completed iii- What is function of porous pot? to prevent two electrolyte from mixing iv- State the negative terminal and positive terminal between zinc plate and cooper plate Negative terminal-Zinc plate and positive terminal- Cooper plate v. Why zinc act as negative terminal Since the position of Zinc is higher than position of Copper in electrochemical series
44
MODUL CHEMISTRY 2014
4 Diagram 4 shows the set-up of apparatus to investigate the electrolysis of sodium chloride solution with carbon electrodes. Rajah 4 menunjukkan susunan radas untuk mengkaji elektrolisis larutan natrium klorida dengan menggunakan elektrod-elektrod karbon.
Test tube Tabung Uji Electrolytic Cell Sel Elektrolisis
0.001 mol dm-3 sodium chloride solution larutan natrium klorida 0.001 mol dm-3
Electrode P Elektrod P
Electrode Q Elektrod Q
Diagram 4 Rajah 4 (a) What is the energy change in the electrolytic cell above? Apakah perubahan tenaga dalam sel elektrolisis di atas ......................................................................................................................................... [1 mark] (b) Write the formulae of all ions present in sodium chloride solution. Tuliskan formula bagi semua ion yang hadir dalam larutan natrium klorida. ...................................................................................................................................... [1 mark] (i) Name the gas collected at electrode P. Namakan gas yang terkumpul pada elektrod P ........................................................... ...................... [1 mark] [1 markah]
(c)
45
MODUL CHEMISTRY 2014
(ii) How do you identify the gas collected in (c)(i)? Bagaimanakah anda mengesahkan gas yang terkumpul di (c)(i)? ............................................. .... [2 marks] [2 markah] (d) (i) What is the product formed at electrode P if the experiment is repeated using 2.0 mol dm-3 sodium chloride solution? Apakah hasil yang akan terbentuk di elektrod P jika eksperimen ini di ulangi dengan menggunakan larutan natrium klorida 2.0 mol dm-3? .................................. .. [1 mark] [1 markah] Explain your answer in (d)(i). Terangkan jawapan anda di (d)(i). .... .... .. [2 marks] [2 markah] (iii) Write the half equation for the reaction at electrode P. Tuliskan persamaan setengah bagi tindak balas di elektrod P. ....... [1 mark] [1 markah] (e) Suggest one metal that can replace the carbon electrodes in this experiment to obtain the same result as above. Cadangkan satu logam yang boleh menggantikan elektrod karbon dalam eksperimen tersebut untuk mendapatkan keputusan yang sama seperti di atas. ................................................. [1 mark] [1 markah]
(ii)
46
MODUL CHEMISTRY 2014
TRIAL SPM
2.Diagram 2 shows the set up of apparatus to investigate the electrolysis of silver nitrate solution with carbon electrodes and copper(II) sulphate solution with copper electrodes. Rajah 2 menunjukkan susunan radas bagi satu eksperimen untuk mengkaji elektrolisis larutan argentum nitrat dengan elektrod karbon dan elektrolisis larutan kuprum(II) sulfat dengan elektrod-elektrod kuprum.
Carbon electrode Elektrod karbon Silver nitrate solution Larutan argentum nitrat
Copper electrode Elektrod kuprum Copper(II) sulphate solution Larutan kuprum(II) sulfat
Cell 1 Sel 1 Diagram 2 Rajah 2 (a) What is the energy change in electrolysis? Apakah perubahan tenaga dalam elektrolisis?
Cell 2 Sel 2
[ 1 mark] (b) Write the formulae of all the ions present in silver nitrate solution. Tuliskan formula bagi semua ion yang terdapat dalam larutan argentum nitrat. ............................................................................................................................................... [ 1 marks] (c) In the electrolysis of Cell 1 Dalam elektrolisis Sel 1 (i) What is the observation at electrode B? Apakah pemerhatian pada elektrod B? [ 1 mark]
47
MODUL CHEMISTRY 2014
(ii) Write the half equation for the reaction at electrode B. Tulis persamaan setengah yang berlaku pada elektrod B.
(d)
. [ 1 mark] In the electrolysis of Cell 2. Dalam elektrolisis Sel 2. (i) What is the observation at electrode C? Apakah pemerhatian pada elektrod C? ..................................... [1 mark] Write the half equation for the reaction at electrode C. Tulis persamaan setengah yang berlaku pada elektrod C.
(ii)
(e )
.. [1 mark] What are the processes that occur at electrodes A and D? Apakah proses yang berlaku pada elektrod A dan D? Electrode A Electrode D :.. :. [2 marks]
(f )
State which cell the concentration of electrolyte remains unchanged. Nyatakan sel manakah kepekatan elektrolitnya tidak berubah. ............... [1 mark]
(g )
State one application of electrolysis in industrial. Nyatakan satu aplikasi elektrolisis dalam industri.. .............. [1 mark]
48
MODUL CHEMISTRY 2014
TRIAL SPM
Diagram 1 shows the set-up of apparatus to investigate the electrolysis of 0.0001 mol dm3 of hydrochloric acid and 1.0 mol dm3 of potassium iodide using carbon electrodes. Rajah 1 menunjukkan susunan radas untuk mengkaji elektrolisis 0.0001 mol dm3 hidroklorik asid dan 1.0 mol dm3 kalium iodida dengan menggunakan elektrod karbon.
Electrode X [Elektrod X] Electrode W [Elektrod W Hydrochloric acid [asid hidroklorik]
Electrode Y [Elektrod Y] Electrode Z [Elektrod Z] Potassium iodide solution [Larutan kalium iodida]
Diagram 1[Rajah 1]
(a)
(i) Identify all the electrodes which serve as anode and cathode. Kenalpastikan semua elektrod yang berfungsi sebagai anod dan katod Anode [anod]:.................................................................................................................... Cathode [katod]:................................................................................................................ [2 marks] (ii) Name the product that is formed at electrode W. Namakan hasil yang terbentuk pada elektrod W. ..... [1 mark] (iii) Write the half equation for the reaction that occurs at electrode W. Tuliskan setengah persamaan bagi tindak balas yang berlaku di elektrod W. .. [1mark]
49
MODUL CHEMISTRY 2014
(b) (i) State the observation at electrode Y. Nyatakan pemerhatian pada elektrod Y. [1 mark]
(ii) Name the product formed in b(i) Namakan hasil yang terbentuk di b(i) ............................................................................................................................................ [ 1 mark] (iii) Suggest a test to confirm the product in b(ii). Cadangkan satu ujian untuk mengesahkan hasil yang terbentuk di b(ii). ..... .................................................. [2 marks] (c) In a separate experiment, the 0.0001 mol dm3 of hydrochloric acid is replaced with 1.0 moldm3 of hydrochloric acid and electrolysis is carried out. Dalam experimen yang berlainan, asid hidroklorik yang berkepekatan 0.0001 mol dm3 digantikan dengan asid hidroklorik yang berkepekatan 1.0 mol dm3 dan elektrolisis dijalankan.. (i) State the product that will form at electrode W. Nyatakan hasil yang akan terbentuk pada elektrod W. ..... [1 mark] (ii) Explain your answer in c(i). Terangkan jawapan anda dalam c(i). ............................................................................................................................................ ..... . [1 mark]
50
MODUL CHEMISTRY 2014
TRIAL SPM
Diagram 3.2 shows the set up of the apparatus used to electroplate an iron key with silver metal. Rajah 3.2 menunjukkan susunan radas yang digunakan untuk menyadur satu kunci besi dengan logam perak.
Electrolyte R Elektrolit R
Diagram / Rajah 3.2
Based on Diagram 3.2 : Berdasarkan Rajah 3.2 : (i) State the correct position of the iron key either electrodes P or Q. Nyatakan kedudukan yang betul bagi kunci besi samada elektrod P atau Q. .......................... [1 mark] (ii) Name a suitable solution that can be used as electrolyte R. Namakan satu larutan yang sesuai digunakan sebagai elektrolit R. .......................... [1 mark] (iii) Suggest one step that must be taken to get a better electroplated key. Cadangkan satu langkah yang perlu diambil untuk mendapatkan saduran yang baik pada kunci tersebut. .......................... [1 mark]
51
MODUL CHEMISTRY 2014
(c) Diagram 3.3 shows the set-up of apparatus of a purification process. Rajah 3.3 menunjukkan susunan radas bagi suatu proses penulenan.
Electrical source Sumber tenaga elektrik
A X Y
Impure copper plate Kepingan kuprum tak tulen
Pure copper plate Kepingan kuprum tulen
Copper(II) sulphate solution Larutan kuprum(II) sulfat Diagram / Rajah 3.3 (i) Identify the anode and the cathode. Kenal pasti anod dan katod. X : ............ Y : ............ [1 mark] (ii) During this process, the intensity of the blue colour of copper(II) sulphate remains unchanged. Explain why. Semasa proses ini, didapati keamatan warna biru larutan kuprum(II) sulfat tidak berubah. Terangkan mengapa. .................................................................. ................................................................ ................................................................. [2 marks]
52
MODUL CHEMISTRY 2014
TRIAL SPM JOHOR
1.The diagram below shows the apparatus set up to study the effect of concentration of electrolyte on the products of electrolysis
Experiment I : Electrolysis of very dilute sodium chloride solution Experiment II : Electrolysis of concentrated sodium chloride solution In Experiment I (i) What can be observed at the anode ? [ 1 mark] (ii) Write an equation to represent the reaction that takes place at the anode. [ 1 mark] (iii) What is the redox process that takes place at the anode ? . [ 1 mark] In Experiment II, (i) Which electrode will give a different compared to Experiment I ? . [ 1 mark] (ii) Name the product formed at the electrode in (b) (i). .. [ 1 mark] (iii) Write an equation for the reaction that takes place in (b) (i). [ 1 mark]
53
MODUL CHEMISTRY 2014
5. Diagram 3.1 shows the set-up of apparatus of an electrolytic cell.In beaker A, energy is change from chemical energy to electrical energy. Rajah 3.1 menunjukkan susunan radas bagi sel elektrolisis.Perubahan tenaga yang berlaku dalam bikar A ialah dari tenaga kimia kepada tenaga elektrik V
Magnesium plate [Plat magnesium] ZnSO4(ak) Beaker A [Bikar A] (a) (i)
Zinc plate [Plat zink]
Carbon rod [Rod karbon] CuSO4(aq) Beaker B [Bikar B]
Name all the cations present in the solution of beaker B. Namakan semua kation yang hadir dalam larutan di bikar B.
.................................................................................................................. [1 mark] (ii) Mark the negative electrode and positive electrode in beaker B Tandakan elektrod negatif dan positif dalam bikar B [1 mark] What is the expected observation in the copper (II) sulphate solution? Apakah pemerhatian yang dijangkakan pada larutan kuprum (II) sulfat?
(b) (i)
................................................................................................................ [1 mark] (ii) Explain your answer in (b) (i) Jelaskan jawapan anda dalam (b) (i) ................................................................................................................. [1 mark] What is the product formed at the anode in beaker Apakah hasil yang terbentuk di anod dalam bikar A? ............................................................................................................... [1 Mark] Write a half equation for the reaction that occurs at the anode. Tulis setengah persamaan bagi tindakbalas yang berlaku di anod [1 Mark]
(c) (i)
[1 mark]
(ii)
54
MODUL CHEMISTRY 2014
(ii) If the zinc plate is replaced by a copper plate, describe what will happen to the voltmeter reading.Explain your answer. Jika plat zink diganti dengan plat kuprum,nyatakan apa yang berlaku pada bacaan voltmeter.Jelaskan jawapan anda .. [2 marks] TRIAL SPM 2.Diagram 3.1 shows the set-up of apparatus for two types of cell. (Rajah 3.1 menunjukkan susunan radas untuk dua jenis sel.)
Cell P (a) DIAGRAM 3.1 State the energy changes in cell P and cell Q. (Nyatakan perubahan tenaga dalam sel P dan sel Q.) Cell P:...
Cell Q
Cell Q:... [2 marks] (b) Write the half-equation for the reaction that occurs at the magnesium rod in cell P. (Tuliskan setengah persamaan bagi tindak balas yang berlaku di rod magnesium dalam sel P.) [2 marks]
55
MODUL CHEMISTRY 2014
(c) Mark the direction of the flow of electrons in cell P. (Tandakan arah aliran electron dalam Sel P) [1 mark] (d) (i) What happens to the voltmeter reading when the magnesium rod in cell P is replaced by zinc rod? (Apakah yang berlaku kepada bacaan voltmeter apabila rod magnesium dalam sel P digantikan dengan rod zink?) [1 mark]
(ii) Explain your answer in d(i). (Terangkan jawapan anda di d(i).) .. [1 mark] (e) (i) Label the anode in cell Q. (Labelkan anod dalam sel Q) [1 mark] (ii) What is observed at the copper anode in cell Q? (Apakah yang diperhatikan pada anod kuprum dalam sel Q?) . [1 mark] (f) What happens to the intensity of colour of the solution in cell Q? Explain your answer. (Apakah yang berlaku kepada keamatan warna larutan dalam sel Q? Terangkan jawapan anda..) [2 marks]
56
MODUL CHEMISTRY 2014
TRIAL SPM PERLIS 6. Diagram 1 shows the set up of the apparatus to investigate the electrolysis of 1.0 mol dm-3 potassium iodide solution.
Carbon electrode X
Carbon electrode Y Potassium iodide solution
a)
DIAGRAM 1 i) What is meant by anion? [1 mark] ii) Which electrode is cathode ? [1 mark]
b)
i) State all the ions present in potassium iodide solution. [1 mark] ii)State the ion that will be discharged at Electrode X: Electrode Y: .. .. [2 marks]
57
MODUL CHEMISTRY 2014
(c) (i) What is the observation at electrode X? [1 mark] Write the half equation for the reaction at electrode X. [1 mark] (d) (i) Name the gas collected at electrode Y.
(ii)
.... [1 mark] (e) If 0.0001 mol dm-3 potassium iodide solution is used as electrolyte (i) What is produced at electrode X at the beginning of the experiment? .. [1 mark] (ii) Write the half equation in (e)(i) ...................................... [1 mark]
58
MODUL CHEMISTRY 2014
TRIAL SPM MELAKA 5. Diagram 5 shows two types of cells. Rajah 5 menunjukkan dua jenis sel.
Battery Bateri
Voltmeter Volmeter Zinc Plate Kepingan zink
Copper Plate Kepingan kuprum P
Copper Plate Kepingan kuprum
Copper(II) sulphate solution Larutan kuprum(II) sulfat DIAGRAM 5 RAJAH 5
(g) Write the ionic formulae of all cations in copper (II) sulphate solution. Tuliskan formula ion bagi semua kation dalam larutan kuprum(II) sulfat. .............................................................. [1 mark] (h) State the energy changes in cell P and cell Q. Nyatakan perubahan tenaga dalam sel P dan sel Q. Cell P:... Sel P Cell Q : .... Sel Q
[1 mark]
[1 mark]
(i) Write the half equation for the reaction that occurs at the cathode in cell P. Tuliskan setengah persamaan bagi tindak balas yang berlaku pada katod dalam sel P. .. [2 marks]
59
MODUL CHEMISTRY 2014
(j) (i) Is there any colour change of the copper(II) sulphate solution in cell P? Adakah terdapat perubahan warna pada larutan kuprum(II) sulfat dalam sel P? ........................................................... (ii) Explain your answer in (d)(i). Terangkan jawapan anda dalam (d)(i) ............................................................... ............................................................. [2 marks] (k) State the negative terminal in cell Q. Nyatakan terminal negatif dalam sel Q. ... [1 mark] (l) If the zinc plate is replaced with a magnesium plate, what will happen to the voltmeter reading in cell Q? Jika kepingan zink digantikan dengan kepingan magnesium, apakah yang berlaku kepada bacaan voltmeter dalam sel Q? ........................................................ [1 mark] (ii) Explain your answer in (d)(i). Terangkan jawapan anda dalam (d)(i) ...................................................... ..................................................... [2 marks] (m) State the negative terminal in cell Q. Nyatakan terminal negatif dalam sel Q. .. [1 mark] (n) If the zinc plate is replaced with a magnesium plate, what will happen to the voltmeter reading in cell Q? Jika kepingan zink digantikan dengan kepingan magnesium, apakah yang berlaku kepada bacaan voltmeter dalam sel Q? ...................................................... [1 mark]
60
MODUL CHEMISTRY 2014
SET 4 ACID AND BASE
61
MODUL CHEMISTRY 2014
CHAPTER 7 Favourite question: i. What is meant by strong acid? Strong acid is acid that ionized completely in water to produced a high concentration of hydrogen ions ii. Give the examples of strong acid Hydrochloric acid (HCl), Nitric acid (HNO3) and Sulphuric acid (H2SO4) iii. What is meant by weak acid? Acid that ionized partially in water to produced low concentration of hydrogen ion iv. Give the examples of weak acid Ethanoic acid V. What is meant by strong alkali? Strong alkali ia alkali that ionized completely in water to produced a high concentration of hydroxide ion vi. Give examples of strong alkali Sodium Hydroxide (NaOH), Potassium Hydroxide (KOH) vii. What is meant by weak alkali Alkali that ionized partially in water to produced low hydroxide ion viii. Give examples of weak alkali Ammonia
EXPERIMENT INVOLVING NEUTRALISATION PROCESS Favourite question:
v. Name the reaction that take places in experiment Neutralisation process vi. Name the technique/ method using in this experiment Titration method vii.State the colour change of the phenolphthalein in the conical flask at the end pink to colourless viii. Name the another substance that can be used to replace phenolphthalein methyl orange x. What is meant by end point is the point in the titration at which the indicator changes colour point
62
MODUL CHEMISTRY 2014
hydrochloric acid 0.1mol dm-3
25.00 cm3 sodium hydroxide
Calculation : Exp I Volume acid used = Final reading initial reading = 39.70- 17.30 cm3 = 22.40 cm3 Exp II Volume acid used = 39.20 17.00 cm3 = 22.20 cm3 Exp III Volume acid used = 39.70 17.70 cm3 = 22.00 cm3 xi. Calculate the average volume of sulphuric acid used in this reaction 22.40 + 22.20 + 22.00 cm3 3 = 22.20 cm3
xiii. Write the chemical equation 1HCl + 1NaOH NaCl + H2O xiv. Calculate the Molarity (M) of Sodium Hydroxide MacidVacid MalkaliValkali (0.1) (22.20) Malkali(25.00) = 1 1 = 1 1
Molarity/ Malkali (25.00) (1) = (0.1) (22.20) Molarity / Malkali = 2.22 = 0.0888 mol dm-3 25.00
63
MODUL CHEMISTRY 2014
xv. The experiment is repeated with 0.1 mol dm-3 sulphuric acid to replaced hydrocholoric acid.Predict the volume of sulphuric acid needed to neutralized 25.00 cm3 sodium hydroxide Volume = volume HCl acid = 22.20 = 11.10 cm3 2 2 xvi. Explain your answer in xv Sulphuric acid is diprotic acid and Hydrochloric acid is monoprotic acid. So sulphuric acid has twice the number of hydrogen ions compared to HCl acid CONCEPT OF pH ACID AND ALKALI Favorite question Hydrochloric acid A B C D Concentration of Hydrcholoric acid Mol dm-3 0.001 0.01 0.1 1 pH 3 2 1 0
i. Based on table state the relationship between the concentration of hydrochloric acid and pH value The higher concentration of hydrochloric acid the lower pH value ii. Explain your answer in a (i) -The pH value is used to measure the concentration of H+ ions - the higher the number of H+ ion per unit volume solution , the lower the pH value iii. Arrange the hydrocholoric acid according to increasing order A,B,C,D
Alkali Sodium Hydroxide Ammonia Solution
Concentration mol dm-3 0.1 0.1
pH 13 10
iv. Explain why the two alkalis have different pH values? - Sodium hydroxide is a strong alkali - Ammonia is a weak alkali - Sodium hydroxide ionized completely in water to produced higher concentration of ion OH- Ammonia ionized partially in water to produced low concentration of OH- The concentration of ion OH- in sodium Hydroxide in higher than Ammonia - When the concentration of OH- ion is higher the pH value is higher.
64
MODUL CHEMISTRY 2014
TRIAL KEDAH SPM 2009 1. An experiment was carried out to investigate neutralisation reaction between 1.0 mol dm-3 sulphuric acid and 20.00 cm3 of sodium hydroxide solution. 3 drops of phenolphthalein are added to the solution. The acid is slowly added to the sodium hydroxide solution until end point. Table below shows the result of the experiment. Satu eksperimen telah dijalankan untuk mengkaji tindak balas peneutralan di antara 1.0 moldm-3 asid sulfurik dan 20.00 cm3 larutan natrium hidroksida. 3 titis fenolftalein telah ditambahkan ke dalam larutan tersebut. Larutan asid ditambahkan sedikit demi sedikit sehingga mencapai takat akhir. Jadual di bawah menunjukkan keputusan eksperimen tersebut. Set of experiment Initial burette reading, cm3. Bacaan awal buret, cm3 Final burette reading, cm3. Bacaan akhir buret, cm3 Volume of acid used, cm3 Isipadu asid digunakan, cm3 Table 1 What is the meaning of end point? Apakah yang dimaksudkan dengan takat akhir? ......................................................................... [1 mark] State the colour change of phenolphthalein when it reaches end point of titration. Nyatakan perubahan warna fenolftalein apabila mencapai takat akhir pentitratan. ............................................................... [1 mark] [1 markah] c) Write the chemical equation for the reaction. Tuliskan persamaan kimia bagi tindak balas di atas. ................................................................. [1 mark] [1 markah] d) (i) Find the average volume of sulphuric acid used? Cari isipadu purata asid sulfurik asid yang digunakan? ................................. [1 mark] [1 markah] 1 0.50 20.60 20.10 2 20.60 40.60 20.00 3 1.20 20.10 19.90
a)
b)
65
MODUL CHEMISTRY 2014
Calculate the molarity of sodium hydroxide solution used in this experiment. Kira kepekatan larutan natrium hidroksida yang digunakan dalam eksperimen ini.
(ii)
[2 marks] [2 markah] e) Draw labelled diagram of set up apparatus to carry out the experiment. Lukis gambarajah berlabel susunan radas untuk menjalankan eksperimen ini.
f)
[2 marks] [2 markah] If sulphuric acid is replaced with 1.0 moldm-3 hydrochloric acid to neutralise 20.00cm3 sodium hydroxide solution of the same concentration. It is shown that the volume of the acid needed is double the volume of sulphuric acid. Explain why? Jika asid sulfurik digantikan dengan 1.0 moldm-3 asid hidroklorik untuk meneutralkan 20.00cm3 larutan natrium hidroksida dengan kepekatan yang sama. Didapati isipadu asid yang diperlukan ialah dua kali ganda daripada isipadu asid sulfurik. Terangkan mengapa? ................................................... .................................................. .................................................... [2 marks] [2 markah]
66
MODUL CHEMISTRY 2014
FINAL EXAM JOHOR 2011 1. Diagram 1 shows the apparatus set-up for the neutralisation reaction between a strong acid and a strong alkali. Rajah 1 menunjukkan susunan radas untuk tindak balas peneutralan antara satu asid kuat dan satu alkali kuat.
Hydrochloric acid, 0.1 mol dm-3 Asid hidroklorik, 0.1 mol dm-3
25 cm3 sodium hydroxide solution + phenolphthalein 25 cm3 larutan natrium hidroksida + fenolftalein DIAGRAM 1 3 25.0 cm of sodium hydroxide solution is poured into a conical flask. A few drops of phenolphthalein are added into the solution. The solution in the conical flask is titrated -3 with 0.1 mol dm hydrochloric acid . 25.0 cm3 larutan natrium hidroksida dimasukkan ke dalam sebuah kelalang kon. Beberapa titik fenolftalein ditambah ke dalam larutan itu. Larutan dalam kelalang kon dititratkan dengan 0.1 mol dm-3 asid hidroklorik. (a) Hydrochloric acid is a strong acid. What is meant by a strong acid? Asid hidroklorik adalah asid kuat. Apakah yang dimaksudkan dengan asid kuat? .. [2 marks] (b) Suggest an apparatus that can be used to measure 25.0 cm3 of sodium hydroxide solution accurately. Cadangkan satu radas yang dapat digunakan untuk menyukat 25.0 cm3 larutan natrium hidroksida dengan tepat. . [1 mark]
67
MODUL CHEMISTRY 2014
(c) State the colour change of the solution in the conical flask at the end point. Nyatakan perubahan warna larutan dalam kelalang kon pada takat akhir. [1 mark] (d) (i) Write a chemical equation for the above reaction. Tulis persamaan kimia bagi tindak balas di atas. . [1 mark] (ii) In this experiment, 20.0 cm3 hydrochloric acid is needed to neutralise 25.0 cm3 of sodium hydroxide solution. Calculate the molarity of the sodium hydroxide solution. Dalam eksperimen ini, 20.0 cm3 asid hidroklorik diperlukan untuk meneutralkan 25.0 cm3 larutan natrium hidroksida. Hitungkan kemolaran larutan natrium hidroksida itu.
[2 marks] (e) (i) The experiment is repeated with 0.1 mol dm-3 sulphuric acid to replace hydrochloric acid. Predict the volume of sulphuric acid needed to neutralise 25.0 cm3 sodium hydroxide solution. Eksperimen itu diulang dengan menggunakan 0.1 mol dm-3 asid sulfurik bagi menggantikan asid hidroklorik. Ramalkan isipadu asid sulfurik yang diperlukan untuk meneutralkan 25.0 cm3 larutan natrium hidroksida. [1 mark] Explain your answer in (e) (i). Terangkan jawapan anda dalam (e)(i). . [2 marks]
(ii)
68
MODUL CHEMISTRY 2014
TRIAL JOHOR SPM 2010
3 Table 3 shows pH values of different concentration of hydrochloric acid, HCl solutions. Jadual 3 menunjukkan nilai-nilai pH bagi larutan asid hidroklorik, HCl yang berbeza kepekatan.
Type of solution Jenis larutan Concentration Kepekatan pH value Nilai pH
Solution I Larutan I 0.1 mol dm-3 1
Solution II Larutan II 0.01 mol dm-3 2
Solution III Larutan III 0.001 mol dm-3 3
(a)
(i)
Table 3 Which of the solutions in Table 3 has the highest concentration of hydrogen ions? Larutan yang manakah dalam Jadual 3 mempunyai kepekatan ion hidrogen yang paling tinggi? ........................................................................................................................ [1 mark] [1 markah]
(ii)
Calculate the number of hydrogen ions in 25 cm3 of Solution II. [Avogadro Constant = 6 x 1023 mol-1 ] Hitungkan bilangan ion hidrogen dalam 25 cm3 Larutan II. [Nombor Avogadro = 6 x 1023 mol -1 ]
[2 marks] [2 markah] (iii) Name an indicator that can be used to measure the pH values of the solutions I, II and III. Namakan satu penunjuk yang boleh digunakan untuk mengukur nilai pH larutan-larutan I, II dan III. ... [1 mark] [1 markah]
69
MODUL CHEMISTRY 2014
(b) Solution I is used to neutralise 25 cm3 of 0.1 mol dm-3 of sodium hydroxide, NaOH solution. Larutan I digunakan untuk meneutralkan 25 cm3 larutan natrium hidroksida, NaOH 0.1 mol dm-3. (i) Calculate the number of moles of sodium hydroxide, NaOH in the solution. Hitungkan bilangan mol natrium hidroksida, NaOH dalam larutan.
[1 mark] [1 markah] (ii) Calculate the volume of Solution I used to neutralise sodium hydroxide, NaOH solution in cm3. Hitungkan isipadu Larutan I yang digunakan untuk meneutralkan larutan natrium hidroksida, NaOH dalam cm3.
(iii)
[3 marks] [3 markah] Draw an apparatus set-up for the titration of sodium hydroxide solution with solution I. Lukis susunan radas untuk pentitratan larutan natrium hidroksida dengan Larutan I
[2 marks] [2 markah]
70
MODUL CHEMISTRY 2014
SET 5
MANUFACTURED SUBSTANCE IN INDUSTRY
71
MODUL CHEMISTRY 2014
TRIAL JOHOR 2009 1 Diagram 1.1 shows the arrangement of atoms in substance X and Y. Rajah 1.1 di bawah menunjukkan susunan atom-atom untuk bahan X dan Y
Substance X Bahan X (a)
Diagram 1.1 Rajah 1.1
Substance Y Bahan Y
Based on Diagram 1.1, answer the following questions: Berdasarkan Rajah 1.1, jawab soalan-soalan berikut : (i) Identify which of the substance in Diagram 1.1 is a pure metal or alloy. Tentukan bahan yang manakah dalam Rajah 1.1 adalah logam tulen atau aloi. Pure metal : .................................................................................................... Logam Tulen [1 mark] [1 markah] Alloy : ................................................................................................................. Aloi [1 mark] [1 markah] (ii) Explain your answers in (a) (i) Terangkan jawapan anda dalam (a) (i). ............................................................................................................................ .......................................................................................................................... [2 marks] (iii) Which substance is harder, X or Y? Give a reason for your answer. Di antara bahan X dan Y, yang manakah lebih keras? Berikan satu sebab bagi jawapan anda. ....................................................................................................................... ......................................................................................................................
[ 2marks]
72
MODUL CHEMISTRY 2014
(b) Diagram 1.2 below shows the structural formula of a polymer. Rajah 1.2 di bawah menunjukkan formula struktur bagi suatu polimer.
H C H
H C
H C
H C Cl
H C H
H C Cl n
Cl H
Diagram 1.2 Rajah 1.2 Based on Diagram 1.2, answer the following questions: Berdasarkan Rajah 1.2, jawab soalan-soalan berikut: (i) State the meaning of polymer. Nyatakan maksud polimer. ........................................................................................................................... ........................................................................................................................... [1 mark] [1 markah] (ii) Draw and name the structural formula of its monomer. Lukiskan dan namakan formula struktur bagi monomernya.
[2 marks] (iii) State one use of the polymer in Diagram 1.2 in our daily life. Nyatakan satu kegunaan polimer pada Rajah 1.2 dalam kehidupan seharian kita. ............................................................................................................ [1 mark]
73
MODUL CHEMISTRY 2014
TRIAL KEDAH SPM 2 Diagram 5 shows the flow chart for the formation of salt A from the reaction between ammonia and . sulphuric acid. Rajah 5 menunjukkan carta alir pembentukan garam A daripada tindak balas antara ammonia dan asid sulfurik.
Sulphur Sulfur
Sulphur dioxide Sulfur dioksida
Sulphur trioxide Sulfur trioksida
Nitrogen Nitrogen
Hydrogen Hidrogen Process Y Proses Y
Ammonia Ammonia
Sulphuric acid Asid sulfurik
Salt A Garam A Diagram 5 Rajah 5 (a) Nitrogen reacts with hydrogen in Process Y to produce ammonia. Nitrogen bertindak balas dengan hidrogen dalam Proses Y untuk menghasilkan ammonia. (i) Name Process Y. Namakan Proses Y. .................................................................................................................................. [1 mark] [1 markah] (ii) Write the chemical equation for the reaction that occurs in a(i). Tuliskan persamaan kimia bagi tindak balas yang berlaku dalam a(i). .................................................................................................................................. [1 marks] State the catalyst and the optimum temperature needed in the manufacture of ammonia. Nyatakan mangkin dan suhu optimum yang diperlukan dalam penghasilan ammonia. Catalyst : Temperature : oC Mangkin Suhu
(iii)
[2 marks]
74
MODUL CHEMISTRY 2014
TRIAL KELANTAN SPM
2.(a)
Polymers are long chained molecules made by joining together thousands of smaller molecules called monomers . Polimer adalah molekul berantai panjang yang terhasil daripada cantuman beribu-ribu molekul kecil yang dipanggil monomer.
Polypropene and polyvinyl chloride are examples of polymers. State the name of their monomers. Polipropena dan polivinil klorida adalah contoh polimer. Nyatakan nama monomer mereka. Polypropene: ... Polipropena Polyvinyl chloride : Polivinil klorida [2 marks] b. Plastic bag used in our daily life is a type of synthetic polymer. State one advantage and one disadvantage of plastic bag. Beg plastik yang digunakan dalam kehidupan seharian ialah sejenis polimer sintetik. Nyatakan satu kebaikan dan satu keburukan beg plastik. Advantage: Kebaikan: .................................................... Disadvantage: Keburukan: .............................................................. [2 marks] c. Bronze is an alloy formed from copper and tin. Gangsa adalah suatu aloi yang terbentuk daripada kuprum dan timah (i)State one use of bronze. Nyatakan satu kegunaan gangsa. ................................................................... [1 mark] (ii)State one aim of alloying . Nyatakan satu tujuan pengaloian. ................................................................. [1 mark ]
75
MODUL CHEMISTRY 2014
(iii) Draw the arrangement of particles in bronze. Lukiskan susunan zarah-zarah dalam gangsa.
(iv) Bronze is harder than copper. Explain why. Gangsa adalah lebih keras daripada kuprum. Terangkan mengapa. ........................................................................................................................................... .......................................................................................................................................... .......................................................................................................................................... [3 marks] TRIAL CHEMISTRY SPM 2008
Alloy Bronze Aloi Gangsa
Diagram 2 Rajah 2 The Digram 2 shows the formation of an alloy bronze. Rajah di atas menunjukkan pembentukkan aloi gangsa. (a) (i) Name the components of bronze . Namakan komponen-komponen dalam gangsa .
............................................................................................ [1 mark]
76
MODUL CHEMISTRY 2014
(ii) State which is harder by comparing the alloy and its pure metal. Nyatakan yang manakah lebih keras dengan membandingkan aloi dengan logam tulennya,
[ 1 mark] (iii) Explain your answer in (a)(ii) Terangkan jawapan anda dalam (a)(ii) ....................................................................................................................... ...................................................................................................................... [2 marks]. (iv) State one uses of bronze. Nyatakan satu kegunaan gangsa .................................................................................................................................. [1 mark] TRIAL PERLIS SPM 2010 1.Table 1 shows the particulars of four different types of manufactured substances in industry. Rajah 1 menunjukkan butiran tentang empat jenis bahan buatan dalam industri.
Type of manufactured substance Jenis bahan buatan Glass Kaca Polymer Polimer Alloy Aloi
Example Contoh Borosilicate Borosilikat Polythene Politena Brass Loyang Brick Bata
Component Komponen Silicon dioxide, sodium oxide, calcium oxide and Y. Silikon dioksida, natrium oksida, kalsium oksida dan Y. Ethene Etena Copper and zinc Kuprum dan zink Silicon dioxide, aluminium oxide, iron (III) oxide. Silikon dioksida, aluminium oksida, ferum(III) oksida. Table 1
Jadual 1
77
MODUL CHEMISTRY 2014
(a) Name the type of manufactured substance Z. Namakan jenis bahan buatan Z. . [1 mark]
(b)(i) Brass is harder than its pure copper. Explain. Loyang lebih keras daripada logam kuprum tulennya.Terangkan. ..... ................................................................................................................................... ................................................................................................................................... [2 marks]
(ii) Draw a labelled diagram that shows the arrangement of particles in brass. Lukis gambar rajah berlabel yang menunjukkan susunan zarah-zarah dalam loyang.
[2 marks]
(c) Substance Y is part of components of borosilicate glass. Bahan Y adalah sebahagian daripada kaca borosilikat. (i) Name the substance Y. Namakan bahan Y. ....... [1 mark] (ii) State one characteristic of borosilicate glass compared to the other types of glasses. Nyatakan satu sifat kaca borosilikat berbanding dengan kaca jenis yang lain. ....... [1 mark]
78
MODUL CHEMISTRY 2014
(d) Diagram 1 shows the structural formula of polythene. Rajah 1 menunjukkan formula struktur bagi politena.
H H H H | | | | CC CC | | | | H H H H
Diagram 1
Rajah 1
(i)
Draw the structural formula of monomer of polythene. Lukis formula struktur monomer bagi politena.
[1 mark]
(ii)
State one use of polythene. Nyatakan satu kegunaan politena. ................................................................................................................. [1 mark]
79
MODUL CHEMISTRY 2014
80
MODUL CHEMISTRY 2014
81
MODUL CHEMISTRY 2014
82
You might also like
- Pahang 2008 STPM Chem - p2 QuestDocument8 pagesPahang 2008 STPM Chem - p2 QuestLooi Chui YeanNo ratings yet
- f4 StructureDocument39 pagesf4 StructureM Ξ R Λ NNo ratings yet
- 0620 w14 QP 33Document12 pages0620 w14 QP 33Haider AliNo ratings yet
- University of Cambridge International Examinations General Certificate of Education Ordinary LevelDocument20 pagesUniversity of Cambridge International Examinations General Certificate of Education Ordinary Levelmstudy123456No ratings yet
- Structure of The Atom and The Periodic TableDocument28 pagesStructure of The Atom and The Periodic TableMimakos CyberNo ratings yet
- 9701 s17 QP 42 RemovedDocument18 pages9701 s17 QP 42 RemovedSherise EeNo ratings yet
- 9701 s08 QP 2Document12 pages9701 s08 QP 2Ahmed SideegNo ratings yet
- 6CH01 01 QueDocument24 pages6CH01 01 QueMathloob DexterNo ratings yet
- Thermal Physics Assignment 2013Document10 pagesThermal Physics Assignment 2013asdsadNo ratings yet
- 9701 s06 QP 2Document14 pages9701 s06 QP 2G M Ali KawsarNo ratings yet
- 9701 s08 QP 2 PDFDocument12 pages9701 s08 QP 2 PDFtess_15No ratings yet
- Cambridge International AS & A Level: CHEMISTRY 9701/51Document12 pagesCambridge International AS & A Level: CHEMISTRY 9701/51Lyric GuptaNo ratings yet
- 1.1 - Atoms Amount Equations Reactions Acid-Base Redox QP A-Level Ocr Chemistry PDFDocument16 pages1.1 - Atoms Amount Equations Reactions Acid-Base Redox QP A-Level Ocr Chemistry PDFAnonymous Don't wanna sayNo ratings yet
- Edexcel January 2014 - Question Paper - Chemistry U1Document24 pagesEdexcel January 2014 - Question Paper - Chemistry U1Kabir KhanNo ratings yet
- Rate of Reaction 2 QP (Tomek)Document9 pagesRate of Reaction 2 QP (Tomek)Tomasz OstrowskiNo ratings yet
- Pahang STPM Trial 2011 Chemistry Paper 2 (W Ans)Document21 pagesPahang STPM Trial 2011 Chemistry Paper 2 (W Ans)plouffle100% (1)
- Cambridge IGCSE: CHEMISTRY 0620/42Document16 pagesCambridge IGCSE: CHEMISTRY 0620/42Manya PunjabiNo ratings yet
- Chemical Energetics 2 QPDocument11 pagesChemical Energetics 2 QPWilliam TsuiNo ratings yet
- 525 1 Eot 3 2022Document17 pages525 1 Eot 3 2022OTTO OLIMANo ratings yet
- Read These Instructions First: 7 Printed PagesDocument7 pagesRead These Instructions First: 7 Printed PagesZainab ShigriNo ratings yet
- Buku Latihan Kimia f5Document50 pagesBuku Latihan Kimia f5Hajar Norasyikin Abu BakarNo ratings yet
- Energetics Exam QsDocument5 pagesEnergetics Exam Qszadinova.tereza16No ratings yet
- Cambridge International General Certificate of Secondary EducationDocument12 pagesCambridge International General Certificate of Secondary EducationJaden HalkNo ratings yet
- Accounting QuestionDocument25 pagesAccounting Questionmay thansinNo ratings yet
- 0620 w10 QP 63Document12 pages0620 w10 QP 63Haider AliNo ratings yet
- AQA Chemistry GCSE Unit 1 Question Paper June 12Document20 pagesAQA Chemistry GCSE Unit 1 Question Paper June 12Fred MateNo ratings yet
- Chemistry End of Year IncompleteDocument5 pagesChemistry End of Year IncompleteSebastian ZhangNo ratings yet
- Cambridge International General Certifi Cate of Secondary EducationDocument12 pagesCambridge International General Certifi Cate of Secondary EducationDark GreenNo ratings yet
- Ocr PP 07 Legacy Gce 2824 JanDocument20 pagesOcr PP 07 Legacy Gce 2824 JanshahidchemistNo ratings yet
- 1 Hydrogen Peroxide Decomposes To Form Water and Oxygen. This Reaction Is Catalysed byDocument9 pages1 Hydrogen Peroxide Decomposes To Form Water and Oxygen. This Reaction Is Catalysed byKelvin DenhereNo ratings yet
- 0620 m15 QP 32Document12 pages0620 m15 QP 32Jean LeeNo ratings yet
- Chemistry: University of Cambridge International Examinations General Certificate of Education Ordinary LevelDocument16 pagesChemistry: University of Cambridge International Examinations General Certificate of Education Ordinary Levelmstudy123456No ratings yet
- Liquid Out, Temperature 25.5 °C Tube: M/gs P / WDocument7 pagesLiquid Out, Temperature 25.5 °C Tube: M/gs P / WGianra RadityaNo ratings yet
- Topic 3 Test - Paper 2Document2 pagesTopic 3 Test - Paper 2IB BaddiesNo ratings yet
- As-Level Paper 1 pp9Document15 pagesAs-Level Paper 1 pp9ConorNo ratings yet
- Kinetics IIDocument46 pagesKinetics IIHannah IqbalNo ratings yet
- Pahang STPM Trial 2011 Chemistry Paper 2 (W Ans)Document21 pagesPahang STPM Trial 2011 Chemistry Paper 2 (W Ans)lawrenceNo ratings yet
- Toddle MytestDocument11 pagesToddle MytestashwithanumandlaNo ratings yet
- 9701 w17 QP 42 RemovedDocument18 pages9701 w17 QP 42 RemovedSherise EeNo ratings yet
- Cambridge International AS & A Level: CHEMISTRY 9701/23Document16 pagesCambridge International AS & A Level: CHEMISTRY 9701/23jamalNo ratings yet
- Review Pack 1st Term (23-24)Document21 pagesReview Pack 1st Term (23-24)Teck TieNo ratings yet
- 9701 s10 QP 21Document24 pages9701 s10 QP 21G M Ali KawsarNo ratings yet
- 4 Ideal Gas0001Document18 pages4 Ideal Gas0001Pooja MehraNo ratings yet
- November 2020 (v1) QP - Paper 5 CIE Chemistry A-Level Najat BteichDocument12 pagesNovember 2020 (v1) QP - Paper 5 CIE Chemistry A-Level Najat BteichNajat Naja BteichNo ratings yet
- Mixed Topic Revision 1 DiffusionDocument23 pagesMixed Topic Revision 1 DiffusionYaakkwNo ratings yet
- WCH06 01 Que 20140514Document12 pagesWCH06 01 Que 20140514Paras AgarwalNo ratings yet
- Chemistry: University of Cambridge International Examinations General Certificate of Education Ordinary LevelDocument16 pagesChemistry: University of Cambridge International Examinations General Certificate of Education Ordinary Levelmstudy123456No ratings yet
- Grade 9 Chemistry PT 2 Paper 2Document7 pagesGrade 9 Chemistry PT 2 Paper 2Naisha ParekhNo ratings yet
- Energy Changes, Rate, Reversible, NH3, H2SO4Document5 pagesEnergy Changes, Rate, Reversible, NH3, H2SO4HudaNo ratings yet
- Topic 9 TestDocument15 pagesTopic 9 TestTyler SurveyNo ratings yet
- Separate Chemistry: Higher Tier in BoldDocument16 pagesSeparate Chemistry: Higher Tier in BoldzipperNo ratings yet
- 9701 s16 QP 42 RemovedDocument19 pages9701 s16 QP 42 RemovedSherise EeNo ratings yet
- Kimia P3 SBP Mid Year SPM 2008Document30 pagesKimia P3 SBP Mid Year SPM 2008adeebsparkNo ratings yet
- F334 Jun 09Document20 pagesF334 Jun 09ExamStuffNo ratings yet
- 9701 s07 QP 4Document16 pages9701 s07 QP 4Krishna GangaramNo ratings yet
- Advanced Synthesis of Gold and Zirconia Nanoparticles and their CharacterizationFrom EverandAdvanced Synthesis of Gold and Zirconia Nanoparticles and their CharacterizationNo ratings yet
- CHAPTER 6 +chapter 7Document5 pagesCHAPTER 6 +chapter 7Juni FarhanaNo ratings yet
- Modul Ulangkaji Form 4Document69 pagesModul Ulangkaji Form 4KHARTHIKA78% (9)
- Chemistry Trial Documents 2017Document38 pagesChemistry Trial Documents 2017Juni FarhanaNo ratings yet
- Graf Fungsi 5k2 GalusDocument2 pagesGraf Fungsi 5k2 GalusJuni FarhanaNo ratings yet
- Pecutan Sebatian Karbon SPM 2016Document8 pagesPecutan Sebatian Karbon SPM 2016Juni FarhanaNo ratings yet
- Paper 3 SPM 2011 Mastery PracticesDocument31 pagesPaper 3 SPM 2011 Mastery PracticesJuni FarhanaNo ratings yet
- CHAPTER 6 +chapter 7Document5 pagesCHAPTER 6 +chapter 7Juni FarhanaNo ratings yet
- Latihan Gabungan Alkana N AlkenaDocument6 pagesLatihan Gabungan Alkana N AlkenaJuni FarhanaNo ratings yet
- Chemistry Trial Documents 2017Document38 pagesChemistry Trial Documents 2017Juni FarhanaNo ratings yet
- ANALISIS PAPER 3 CHEMISTRY - Docx 10'4Document14 pagesANALISIS PAPER 3 CHEMISTRY - Docx 10'4Juni FarhanaNo ratings yet
- An Experiment Is Conducted To Determine The Rate of Reaction Between 25 CMDocument3 pagesAn Experiment Is Conducted To Determine The Rate of Reaction Between 25 CMJuni FarhanaNo ratings yet
- Chapter 7 Acids N BasesDocument6 pagesChapter 7 Acids N BasesJuni FarhanaNo ratings yet
- ANALISIS PAPER 3 CHEMISTRY - Docx 10'4Document14 pagesANALISIS PAPER 3 CHEMISTRY - Docx 10'4Juni FarhanaNo ratings yet
- Modul Amali Kertas 3 F4 2015Document9 pagesModul Amali Kertas 3 F4 2015acik5596No ratings yet
- Esei F5 Trial SPM 2015Document34 pagesEsei F5 Trial SPM 2015Juni FarhanaNo ratings yet
- ElectroplatingDocument3 pagesElectroplatingJuni FarhanaNo ratings yet
- Quiz MathematicsDocument2 pagesQuiz MathematicsJuni FarhanaNo ratings yet
- Preparation and Purification of Soluble SaltsDocument12 pagesPreparation and Purification of Soluble SaltsJuni FarhanaNo ratings yet
- Chap 05Document4 pagesChap 05Jamaliah Daud100% (5)
- Chapter 8 SaltDocument2 pagesChapter 8 SaltJuni FarhanaNo ratings yet
- Chapter 8Document7 pagesChapter 8Juni FarhanaNo ratings yet
- Notes Updates Salts2Document33 pagesNotes Updates Salts2Juni FarhanaNo ratings yet
- Chapter 8Document7 pagesChapter 8Juni FarhanaNo ratings yet
- ISSF Stainless Steel For DesignersDocument23 pagesISSF Stainless Steel For DesignersnargissuhailNo ratings yet
- 02 February 1990Document108 pages02 February 1990Monitoring TimesNo ratings yet
- Borax Vpn3449 00 - enDocument1 pageBorax Vpn3449 00 - enManu Galdeano RuizNo ratings yet
- 1814 - KMBD-200-2700 Installation Operation Maintenance Repair Manual - Mechanical Vacuum BoostersDocument34 pages1814 - KMBD-200-2700 Installation Operation Maintenance Repair Manual - Mechanical Vacuum BoostersahmedNo ratings yet
- Woodwork: Cambridge International Examinations General Certificate of Education Ordinary LevelDocument12 pagesWoodwork: Cambridge International Examinations General Certificate of Education Ordinary Levelmstudy123456No ratings yet
- Project Documentation: Mickey Mouse ProgramDocument24 pagesProject Documentation: Mickey Mouse ProgramVaishali PatialNo ratings yet
- Sangean ATS-803a - Manual & SchematicDocument16 pagesSangean ATS-803a - Manual & SchematicBenjamin Dover100% (1)
- Electric Boiler BrochureDocument4 pagesElectric Boiler BrochuredeztruktorNo ratings yet
- L190 #3624 VmeDocument590 pagesL190 #3624 Vmethierrylindo100% (2)
- PI-50 Ball Pump: Operation and Service ManualDocument23 pagesPI-50 Ball Pump: Operation and Service Manualsebastian Zapata QuinteroNo ratings yet
- Chem 1221 Module 6 PDFDocument49 pagesChem 1221 Module 6 PDFMaries San PedroNo ratings yet
- TCM IO520 OpsInstallManualDocument78 pagesTCM IO520 OpsInstallManualleather_nunNo ratings yet
- Selecting ERP Consulting PartnerDocument7 pagesSelecting ERP Consulting PartnerAyushmn SikkaNo ratings yet
- Devoirs AnglaisDocument2 pagesDevoirs Anglaisyounes bahiNo ratings yet
- Water Proofing Toilets WorksDocument63 pagesWater Proofing Toilets WorksGhaffar Laghari100% (1)
- BrutalismDocument9 pagesBrutalismQamar AtheerNo ratings yet
- Finite Element Modeling of Reinforced Concrete Beams Strengthened With FRP Laminates: A ReviewDocument8 pagesFinite Element Modeling of Reinforced Concrete Beams Strengthened With FRP Laminates: A ReviewAfia.S.HameedNo ratings yet
- Brother 5370DW Config v2Document15 pagesBrother 5370DW Config v2zebitoexpostoNo ratings yet
- 5 Bears - Ignition CircuitsDocument2 pages5 Bears - Ignition CircuitsAlonso CoradoNo ratings yet
- Karta Danych - enDocument5 pagesKarta Danych - enwiruNo ratings yet
- The Properties of Roof TilesDocument9 pagesThe Properties of Roof TilesSingh is KingNo ratings yet
- ECOPUNK Crew SheetDocument1 pageECOPUNK Crew SheetECO SpeedsNo ratings yet
- STS304 Datasheet: Chemical, Physical & Mechanical PropertiesDocument2 pagesSTS304 Datasheet: Chemical, Physical & Mechanical PropertiesThanakit Srisaeng100% (1)
- Ibm Appscan Source DatasheetDocument12 pagesIbm Appscan Source DatasheetAnonymous G1iPoNOKNo ratings yet
- ASTM D 1337 - 96 Storage Life of Adhesives by Consistency and BondDocument2 pagesASTM D 1337 - 96 Storage Life of Adhesives by Consistency and Bondalin20050% (1)
- KR-G11 User Guider AlarmDocument21 pagesKR-G11 User Guider AlarmJuan DuqueNo ratings yet
- Lecture5 IntroMapReduceDocument57 pagesLecture5 IntroMapReduceClaudio Canales VicencioNo ratings yet
- Refcon RMM Modem Leaflet en GB 3667694Document2 pagesRefcon RMM Modem Leaflet en GB 3667694FatsahNo ratings yet
- Thermodynamics V0thermofluids 1 School Ofmechanical Engineering University of LeedsDocument45 pagesThermodynamics V0thermofluids 1 School Ofmechanical Engineering University of LeedsLaith GammohNo ratings yet
- Design PatternsDocument4 pagesDesign PatternsHemalatha GanireddyNo ratings yet