Professional Documents
Culture Documents
Evaluacion de Sysmex XE 2100
Uploaded by
willmed0 ratings0% found this document useful (0 votes)
29 views4 pagesHematopoietic stem cells (HSCs) are used for autologous transplantation after high-dose chemotherapy in patients with hematological malignancies. The clinical value of determination of the accurate absolute numbers of CD34+ cells is the most important parameter for evaluating stem and progenitor cell content.
Original Description:
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentHematopoietic stem cells (HSCs) are used for autologous transplantation after high-dose chemotherapy in patients with hematological malignancies. The clinical value of determination of the accurate absolute numbers of CD34+ cells is the most important parameter for evaluating stem and progenitor cell content.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
29 views4 pagesEvaluacion de Sysmex XE 2100
Uploaded by
willmedHematopoietic stem cells (HSCs) are used for autologous transplantation after high-dose chemotherapy in patients with hematological malignancies. The clinical value of determination of the accurate absolute numbers of CD34+ cells is the most important parameter for evaluating stem and progenitor cell content.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 4
test validation
Journal of Appplied Hematology 2010
108
H
ematopoietic stem cells (HSCs) and hemo-
poietic progenitor cells (HPCs) obtained
from either the bone marrow (BM) or mo-
bilized peripheral blood (PB) are used for autologous
transplantation after high-dose chemotherapy in pa-
tients with hematological malignancies. These cells
are characterized by the expression of the adhesion
receptor CD34. The clinical value of determination
of the accurate absolute numbers of CD34+ cells
is the most important parameter for evaluating stem
and progenitor cell content in hematopoietic trans-
plantation. Flow cytometric enumeration of CD34+
cells has become widely accepted as the technique of
choice to quantify HSCs for the clinical management
Hemopoietic Progenitor Cell (HPC)
Enumeration: A Comparitive Study between
Flow Cytometry and Sysmex XE-2100
Hematology Analyzer
Salem H. Khalil, Barbara Geng
From the Hematology Section
Department of Pathology and Laboratory Medicine
King Faisal Specialist Hospital and Research Centre
Riyadh, Saudi Arabia
Corresponding author:
Salem H. Khalil, MD, FRCPA, FCAP
Consultant Hematopathologist
Assistant Professor, Al Faisal University
Department of Pathology and Laboratory Medicine
(MBC-10)
King Faisal Specialist Hospital and Research Centre
P. O. Box 3354, Riyadh 11211
Kingdom of Saudi Arabia
T: +966-1-4424296
F: +966-1-4424280
khalil@kfshrc.edu.sa
The decision to harvest peripheral blood stem cells (PBSCs) is frequently
based on the enumeration of CD34+ cells by flow cytometry by using either
single- or dual-platform methods. The aim of this study at the Department
of Pathology and Laboratory Medicine of King Faisal Specialist Hospital and
Research Centre (General Organization) was to evaluate an alternative meth-
od for circulating hemopoietic stem cell quantification, namely by means of
the hemopoietic progenitor cell (HPC) parameter generated by the Sysmex
XE-2100 automated hematology analyzer available in the Hematology Section.
Two hundred and ninety two (292) samples of peripheral and cord blood were
analyzed using single-platform flow cytometry and the immature myeloid in-
formation (IMI) parameter of the Sysmex XE-2100 hematology analyzer in
parallel runs.
The objective of this study was to minimize the number of CD34 determina-
tions routinely performed by flow cytometry. A strong correlation was found
between HPC and CD34+ in peripheral blood [r
2
(0.692), P value <0.0001]
and cord blood [r
2
(0.555), P value <0.0001]. Therefore, with HPC levels below
the detection limit of 00/mm
3
or count of >30/mm
3
, it is reasonable not to
await the CD34+ results before harvesting. These two situations account for
more than 66% of the CD34+ determinations carried out by flow cytometry
in our laboratory.
KEYWORDS: HPC, Stem cell, Sysmex XE-2100, Flow cytometry
of stem cell transplantation. Until recently, absolute
CD34+ cell counts were generally derived from the
so-called two-platform analysis in which the percent-
age of CD34+ cells is frst enumerated by fow cy-
tometry against the total CD45+ cells as the denomi-
nator. The white blood cell (WBC) derived from the
hematology analyzer to generate the absolute CD34+
cell count then multiplies this percentage of CD34+
cells. The single-platform method, which utilizes
TruCount absolute-count tubes, reduces variability by
eliminating the need for the cell count from an auto-
mated hematology analyzer.
1-4
The aim of this study
was to validate an alternative method for circulating
HSC quantifcation, namely by means of the HPC pa-
108-111 TV-KHALIL.indd 108 12/18/10 8:57 PM
test validation
HPC ENUMERATION
Journal of Applied Hematology 2010
109
rameter generated by the Sysmex XE-2100 automated
hematology analyzer.
Materials and Methods
Patients and study design
Patients were included in the study provided the HPC
and CD34 enumeration were determined simultane-
ously at least on the presumed day of the harvest
and in some cases, on the days before. A total of 292
samples were analyzed using single-platform fow cy-
tometry and the immature myeloid information (IMI)
parameter of the Sysmex XE-2100 hematology ana-
lyzer. These included 77 peripheral blood samples of
patients with different types of hematological malig-
nancies and 215 samples of cord blood.
Counting Methods
The CD34+ cells were quantifed using a single-plat-
form fow cytometry procedure at the Hematology
Section, Department of Pathology and Laboratory
Medicine, King Faisal Specialist Hospital and Research
Centre (General Organization), Riyadh, Saudi Arabia.
The procedure uses TruCount [Beckton Dickinson
(BD)] kits on a four-color FACScalibur (BD) fow cy-
tometer by a single-platform method (Figures 1 and 2).
The HPCs are identifed in the IMI-HPC area of
the IMI channel of the Sysmex XE-2100 on the basis
of their resistance to the lysing reagent, their volume
(direct current), and their internal structure (radio-
frequency). HPCs, like all immature cells, are resistant
to the lytic reagent used and are located within a spe-
cifc gated area of the scattergram.
Results
All data were entered properly coded into prepared
Microsoft Excel 2007 worksheets. Data were exam-
ined for competencies and accuracy prior to export-
ing to Statistical Package for Social Sciences, Volume
16 (SPSS, Chicago, Illinois, USA) for statistical analy-
sis. Using Spearmans rank test, a strong correlation
was found between HPC and WBC [r
2
(0.395), P val-
ue=0.0004] Figure 3 and between HPC and CD34+
in peripheral blood [r2 (0.692), P value <0.0001] and
cord blood [r
2
(0.555), P value <0.0001] Figures 4
and 5. Up to 45.45% of samples showed HPC levels
of more than 30/mm
3
and 20.78% with HPC levels
below detection limits (00/mm
3
), representing more
than 66% of the samples studied. However, the levels
of HPC ranging between 0/mm
3
and 30/mm
3
have
been detected in up to 33.77% of samples.
Figure 1. Single-platform CD34 enumeration in peripheral
blood.
Figure 2a. Single-platform CD34 enumeration in cord blood
pre-gradient volume reduction protocol (Pre-GVRP).
Figure 2b. Single-platform CD34 enumeration in cord blood
post-gradient volume reduction protocol (Post-GVRP).
108-111 TV-KHALIL.indd 109 12/18/10 8:57 PM
test validation
HPC ENUMERATION
Journal of Applied Hematology 2010
110
Figure 3. Correlation between white blood cell (WBC) and hemopoietic progenitor
cell (HPC) in peripheral blood. r
2
(0.395), P value=0.0004.
Figure 4. Correlation between CD34+ and hemopoietic progenitor cell (HPC) in
peripheral blood. r
2
(0.692), P value <0.0001.
Figure 5. Correlation between CD34+ and hemopoietic progenitor cell (HPC) in
cord blood. r2 (0.555), P value <0.0001.
Discussion
HSCs can be mobilized from the bone marrow to the
peripheral blood by chemotherapy, by a combination
of chemotherapy and hematopoietic growth factors,
as well as by hematopoietic growth factors alone.
Today, mobilization is performed with chemotherapy
in combination with growth factors, since it provides
for the most effcient stem cell mobilization, in addi-
tion to the eradication of malignant cells.
The stem cell yield may be increased up to 100
times with the application of a growth factor [e.g.,
granulocyte colony-stimulating factor (G-CSF), gran-
ulocyte macrophage colony-stimulating factor (GM-
CSF)]. The administration of a growth factor alone,
without pre-chemotherapy, e.g., with healthy alloge-
neic donors, has the advantage of exactly planning
the time of apheresis.
An accurate determination of the available amount
of stem cells before transplantation will be required.
This will be done routinely by the number of CD34+
cells measured by different methods of fow cytom-
etry.
5-6
A special parameter of HPC cell quantifcation per
microliter of blood was developed in the IMI chan-
nel of the Sysmex XE-2100 hematology analyzer.
The objective was to provide an alternative method
of the daily CD34+ monitoring by fow cytometry,
which was not only costly but also entailed great ex-
penditures in time and personnel. The IMI channel
of the Sysmex system lyses mature and erythropoietic
cells by means of a special lyse reagent. The remain-
ing immature cells will be two-dimensionally analyzed
by volume and intracellular structure. Cells in the area
of low volume and a reduced plasma/nucleus relation
are detected as HPC cells and analyzed using a special
HPC software program.
7
Since the introduction of the HPC software in
1997, many studies of HPC determination versus
CD34+ cell enumeration have been performed and
reported with different levels of correlation.8-14
From this study, HPC determination has been shown
to be helpful in two situations. First, during the fol-
low-up of patients after mobilization, the CD34+
count does not need to be assessed before HPCs are
detectable. Second, if the HPC count is >30/mm
3
, it
is reasonable not to await the CD34+ results before
harvesting.
These two situations account for more than 66%
of the CD34+ determinations carried out by fow cy-
tometry in our daily practice.
108-111 TV-KHALIL.indd 110 12/18/10 8:57 PM
test validation
HPC ENUMERATION
Journal of Applied Hematology 2010
111
1. Gajkowska1 A, Oldak1 T, Jastrzewska M, Machaj EK, Walewski J, Kraszewska
E and Pojda Z. Flow cytometric enumeration of CD34+ hematopoietic stem
and progenitor cells in leukapheresis product and bone marrow for clinical
transplantation: A comparison of three methods. Folia Histochemica, ETCyto-
biologica. 2006;44(1):53-60.
2. Brocklebank AM, Sparrow RL. Enumeration of CD34+ cells in cord blood: A
variation on a single-platform flow cytometric method based on the ISHAGE
gating strategy. Cytometry. 2001;46:254-261.
3. Fornas O, Garcia J, Petritz J. Flow cytometry of CD34+ cells in whole blood.
Nature Med. 2000;6:833-836.
4. Fritsch G, Printz D, Stimpfl M. Quantification of CD34+ cells: Comparison of
methods. Transfusion. 1997;37:775-784.
5. Sutherland DR. The ISHAG Guidelines for CD34+ cell determination by flow
cytometry. ISHAG Guidelines 1996. J Hematother.1996, 5:213-226.
6. Brocklenad AM. Enumeration of CD34+ Cells in Cord Blood: A variation on
a single-platform flow cytometric method based on the ISHAGE gating strategy.
Cytometry (Communication in Clinical Cytometry). 1996,46:254-261.
7. Cymbalista F, Letestu R. Haemopoietic progenitor cell (HPC) counts on the
Sysmex XE-2100: A new toll for peripheral blood stem cell (PBSC) harvest
monitoring. Sysmex J Int. 2005;15(1):21-26.
8. Yamane T et al. Possibility of identification of hematopoietic stem cells using a
conventional blood cell counter. Eur J Haematol. 1995;55:207208.
9. Mougi H et al. Determination of peripheral blood stem cells using automated
hematology analyzer, SE-9000 IMI channel. Sysmex J Int. 1997;7:6370.
10. Yamane T et al. Determination of hematopoietic stem cells in peripheral
blood by automated hematology analyzer, SE-9000. Sysmex J Int. 1997;7:57
62.
11. Saigo K et al. Optimum timing of peripheral blood stem cell harvest using
se9000 IMI channel in the case of patients with breast cancer. Sysmex J Int.
1997;7:7181.
12. Pollard Y et al. Use of the haemopoietic progenitor cell count of the Sysmex
SE-9500 to refine apheresis timing of peripheral blood stem cells. Br J Haematol.
1999;106:538544.
13. Wang FS, Rowan RM, Creer M, Hay A, Dorfner M, et. al. Detecting Human
CD34+ and CD34- Hematopoietic Stem and Progenitor Cells Using a Sysmex
Automated Hematology Analyzer. Lab Hematol. 2004; 10:200-205.
14. Wang et al. Monitoring hematopoietic stem and progenitor cells with Sys-
mex XE-2100 and SE-9500 analyzers. Lab Haematol. 2002;8(3):119125.
References
Acknowledgments
The authors would like to thank the Flow cytometry
staff (Mr. Faisal Rawas and Ms. Ameena Saifaldeen) at
King Faisal Specialist Hospital and Research Centre,
Riyadh for their technical support and also Dr.
Mohamed Shoukri, PhD for statistical analysis.
108-111 TV-KHALIL.indd 111 12/18/10 8:57 PM
You might also like
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (119)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (265)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (399)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (587)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5794)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2219)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (344)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (894)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (73)
- Mesenquinal Stem Cells Basics and ClinicsDocument198 pagesMesenquinal Stem Cells Basics and ClinicsXavier100% (1)
- Structure and Function of HaemoglobinDocument21 pagesStructure and Function of HaemoglobinEmily HoNo ratings yet
- Mechanical Properties of Red Blood Cell Membranes: Yun Ge Supervisor: Dr. H.Jerry QiDocument9 pagesMechanical Properties of Red Blood Cell Membranes: Yun Ge Supervisor: Dr. H.Jerry QiwillmedNo ratings yet
- Blood and HemopoiesisDocument30 pagesBlood and HemopoiesiswillmedNo ratings yet
- Structure and Function of HaemoglobinDocument21 pagesStructure and Function of HaemoglobinEmily HoNo ratings yet
- Multilevel, Subdivision-Based, Thin Shell Finite Elements: Development and Application To Red Blood Cell Modeling Seth GreenDocument66 pagesMultilevel, Subdivision-Based, Thin Shell Finite Elements: Development and Application To Red Blood Cell Modeling Seth GreenwillmedNo ratings yet
- Lecture 5 Red Cell Defects FinalDocument28 pagesLecture 5 Red Cell Defects FinalwillmedNo ratings yet
- Red Cell Disorders GuideDocument49 pagesRed Cell Disorders GuideGeraldine AgpesNo ratings yet
- Desordenes de Membrana InfDocument3 pagesDesordenes de Membrana InfwillmedNo ratings yet
- Bses Moleculares PDFDocument5 pagesBses Moleculares PDFwillmedNo ratings yet
- Parametros Hematologicos SepsisDocument28 pagesParametros Hematologicos SepsiswillmedNo ratings yet
- Desordenes de Membrana InfDocument3 pagesDesordenes de Membrana InfwillmedNo ratings yet
- Membrane The Molecular Basis of Disorders of The Red Cell: J. Clin. PatholDocument5 pagesMembrane The Molecular Basis of Disorders of The Red Cell: J. Clin. PatholwillmedNo ratings yet
- Red Cell Disorders GuideDocument49 pagesRed Cell Disorders GuideGeraldine AgpesNo ratings yet
- Structure and Function of HaemoglobinDocument21 pagesStructure and Function of HaemoglobinEmily HoNo ratings yet
- Desordenes de Membrana InfDocument3 pagesDesordenes de Membrana InfwillmedNo ratings yet
- Lecture 5 Red Cell Defects FinalDocument28 pagesLecture 5 Red Cell Defects FinalwillmedNo ratings yet
- Membrane The Molecular Basis of Disorders of The Red Cell: J. Clin. PatholDocument5 pagesMembrane The Molecular Basis of Disorders of The Red Cell: J. Clin. PatholwillmedNo ratings yet
- Estandarizacion de Muestra de Medula OseaDocument16 pagesEstandarizacion de Muestra de Medula OseawillmedNo ratings yet
- Evaluacion de La Leucocitosis PDFDocument23 pagesEvaluacion de La Leucocitosis PDFwillmedNo ratings yet
- Roussel Et Al-2012-Cytometry Part A PDFDocument10 pagesRoussel Et Al-2012-Cytometry Part A PDFwillmedNo ratings yet
- Velocidad de Sedimentacion PDFDocument8 pagesVelocidad de Sedimentacion PDFwillmedNo ratings yet
- Estudio de Casos Xe Vs XN PDFDocument29 pagesEstudio de Casos Xe Vs XN PDFwillmedNo ratings yet
- Estudio de Casos Xe Vs XNDocument29 pagesEstudio de Casos Xe Vs XNwillmedNo ratings yet
- EsquistocitosDocument10 pagesEsquistocitoswillmedNo ratings yet
- EsquistocitosDocument10 pagesEsquistocitoswillmedNo ratings yet
- JApplHematol44137-8138261 223622Document8 pagesJApplHematol44137-8138261 223622willmedNo ratings yet
- XN 2000Document20 pagesXN 2000willmedNo ratings yet
- Organizacion Mayor de La Membrana EritrocitoDocument6 pagesOrganizacion Mayor de La Membrana EritrocitowillmedNo ratings yet
- Physio Chapter 11 30Document30 pagesPhysio Chapter 11 30willmedNo ratings yet
- 497.full Evaluacion Del Celldyn 4000Document9 pages497.full Evaluacion Del Celldyn 4000willmedNo ratings yet
- ADAM - CD34 Brochure NanoEntekDocument2 pagesADAM - CD34 Brochure NanoEntekbchaikerhamidNo ratings yet
- Vet Pathol 2011 2011 ACVP Annual Meeting E1 E51Document52 pagesVet Pathol 2011 2011 ACVP Annual Meeting E1 E51Haroon RashidNo ratings yet
- Immunohistochemical MarkersDocument12 pagesImmunohistochemical MarkersAsmara SyedNo ratings yet
- EmCyte BMC Vs PureBMC Comparison Data (3) - 2Document5 pagesEmCyte BMC Vs PureBMC Comparison Data (3) - 2Joel SuraciNo ratings yet
- Base Editing of Hematopoietic Stem Cells Rescues Sickle Cell Disease in MiceDocument42 pagesBase Editing of Hematopoietic Stem Cells Rescues Sickle Cell Disease in MiceOsvaldo VillarNo ratings yet
- 2004-10 NF Vs Schwannoma v1Document2 pages2004-10 NF Vs Schwannoma v1Mario QuinteroNo ratings yet
- (12204749 - Romanian Journal of Internal Medicine) Multiple Histological Subtypes of Dermatofibrosarcoma Protuberans Occurring in The Same TumorDocument10 pages(12204749 - Romanian Journal of Internal Medicine) Multiple Histological Subtypes of Dermatofibrosarcoma Protuberans Occurring in The Same TumordrelvNo ratings yet
- SMCLK SJMC 08 2022Document132 pagesSMCLK SJMC 08 2022Maja TešićNo ratings yet
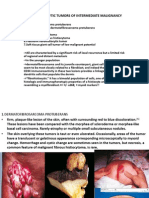
- Fibrohistiocytic Tumors of Intermediate MalignancyDocument39 pagesFibrohistiocytic Tumors of Intermediate MalignancyOana BarbuNo ratings yet
- 2015-Journal of Thrombosis and Haemostasis PDFDocument997 pages2015-Journal of Thrombosis and Haemostasis PDFnikospapaioannouNo ratings yet
- DS CD34 MicroBead Kit HDocument4 pagesDS CD34 MicroBead Kit Hsankalpa3No ratings yet
- Seminars in Diagnostic Pathology Volume 30 Issue 1 2013 (Doi 10.1053 - J.semdp.2012.01.003) Requena, Luis Kutzner, Heinz - HemangioendotheliomaDocument16 pagesSeminars in Diagnostic Pathology Volume 30 Issue 1 2013 (Doi 10.1053 - J.semdp.2012.01.003) Requena, Luis Kutzner, Heinz - HemangioendotheliomazaimmuhtarNo ratings yet
- 2007 Cancer Stem Cells-Biol 610 No AnimDocument49 pages2007 Cancer Stem Cells-Biol 610 No Animapi-3746517No ratings yet
- Human Bone Marrow Organoids For Disease Modeling, Discovery, and Validation of Therapeutic Targets in Hematologic MalignanciesDocument22 pagesHuman Bone Marrow Organoids For Disease Modeling, Discovery, and Validation of Therapeutic Targets in Hematologic MalignanciesSourav DasNo ratings yet
- Apr 11 PDFDocument6 pagesApr 11 PDFBimalKrishnaNo ratings yet
- Stem Cell Lineage and Identity: ZygoteDocument2 pagesStem Cell Lineage and Identity: ZygoteAkanksha RaiNo ratings yet
- 2018 November Magazine For WebsiteDocument40 pages2018 November Magazine For WebsiteCBRNo ratings yet
- Article Text 75576 1 10 20210527Document10 pagesArticle Text 75576 1 10 20210527Nimbus MultiversesNo ratings yet
- R&D Systems Hematology Products: Controls Calibrators Linearity Materials Quality Control ProgramDocument20 pagesR&D Systems Hematology Products: Controls Calibrators Linearity Materials Quality Control Programa sangNo ratings yet
- Archives of Pathology & Laboratory Medicine Online - Acute Leukemia Immunohistochemistry - A Systematic Diagnostic ApproachDocument13 pagesArchives of Pathology & Laboratory Medicine Online - Acute Leukemia Immunohistochemistry - A Systematic Diagnostic Approachpooo80No ratings yet
- IHC - InterpretareDocument185 pagesIHC - InterpretareAnca NeaguNo ratings yet
- Mesenchymal Stem CellsDocument13 pagesMesenchymal Stem CellsArya NugrahaNo ratings yet
- Differentiation of Human Induced Pluripotent Stem Cells Into Erythroid CellsDocument13 pagesDifferentiation of Human Induced Pluripotent Stem Cells Into Erythroid CellsMauricio ChamorroNo ratings yet
- Mouse Cell Surface MarkersDocument9 pagesMouse Cell Surface Markersavalon784No ratings yet
- Spindle Cell TumorsDocument138 pagesSpindle Cell TumorsMadhura ShekatkarNo ratings yet
- C42135AA Beckman Coulter ClearLLab 10C Casebook PDFDocument586 pagesC42135AA Beckman Coulter ClearLLab 10C Casebook PDFHam Bone100% (1)
- Ludy Dobrila, PH.D., Phil Coelho, Pablo Rubinstein, M.D. New York Blood Center & Thermogenesis CorpDocument1 pageLudy Dobrila, PH.D., Phil Coelho, Pablo Rubinstein, M.D. New York Blood Center & Thermogenesis CorpCampaign MediaNo ratings yet
- 2018 - 05 IssuDocument188 pages2018 - 05 IssuEduardo RomeroNo ratings yet
- Ishage ProtocolDocument9 pagesIshage ProtocolEfrain E. Cabrera RojasNo ratings yet