Professional Documents
Culture Documents
2 Theories in Chemical Kinetics
Uploaded by
AnamarijaKatenovskaCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
2 Theories in Chemical Kinetics
Uploaded by
AnamarijaKatenovskaCopyright:
Available Formats
THEORIES IN CHEMICAL KINETICS
Fundamental aspects
Chemists would like to know the details of how chemical reactions occur on a
microscopic scale. Why do certain reactions take place, but others not? If a single molecule
undergoes some type of rearrangement, how are the old bonds broken and the new bonds
formed? Why are some reactions fast while others are slow and why do reaction rates
depend on temperature? A number of theories have been devised over the years in order to
provide answer to questions such as these. Theories in chemical kinetics are used to
explain:
how chemical reactions take place (on a molecular level)
how temperature and participants concentration affect on the reaction rate
One of these theories is known as collision theory. This theory assumes that, for a
reaction to occur, reactant particles must collide. Collisions are vital for chemical changes.
The rate of a reaction depends on the frequency of collisions between reacting particles.
The more frequent the collisions, the faster the rate of the reaction. For a reaction to occur,
still there is something more than simply collision. For example, in a mixture of H2 and I2 at
ordinary temperature and pressure, each molecule undergoes about 1010 collisions per
second. If every collision between H2 and I2 resulted in the formation of HI, the reaction
would be over in much less than a second. Instead, at room temperature the reaction
proceeds very slow. Only about one in every 1013 collisions produces a reaction.
What keeps the reaction from occurring more rapidly?
The above example tells us that only certain
fraction of the total collisions cause chemical
H
change and is called successful or effective
collisions. In order to be effective, colliding
Br
I
HC
particles must:
- collide with the proper orientation;
H
- possess a certain minimum amount of energy
(activation energy).
ineffective collision (a)
If the orientation of the colliding particles is
wrong, no reaction occurs. In the elementary act,
represented on the left, iodine atom replaces
H
bromine atom in CH3Br molecule (single
displacement). So, only the proper arrangements
HC
Br
I
of the reacting particles lead to the formation of
products. These two particles collide, but are not
H
orientated correctly and then no reaction will take
place (a), the reactants simply "bounce" off each
effective collision (b)
other. Second picture shows correct orientation of
CH3Br + I CH3I + Br
particles; hence reaction takes place (b).
The energy that particles have during collision
is also important factor that keeps the reaction from occurring more rapidly. Not all of the
molecules have the same kinetic energy, as shown in the figure below. This is important
because the kinetic energy molecules carry when they collide is the principal source of the
energy that must be invested in a reaction to get it started. At a given temperature, the
number of particles with very low and with very high energy is very small, but the number of
particles with average kinetic energy is the largest. However, particles must possess energy
above the average in order to interact with each other (they are called active molecule).
This minimum amount of energy required for the
reactant particles react is called activation energy
(EA). Hence, their energy is sufficient to make the
collisions among them effective, so that the existing
chemical bonds between the atoms become labile
and new bonds are formed.
How does collision theory explain the
influence of concentration and temperature on
the reaction rate?
This dependence can be explained in the
following way: at higher concentration of the
reactants, both the number of collisions per unit of
time and that of the effective ones grow. Hence the reaction rate also increases.
However as the temperature increases, not only does the number of collisions per
second increase (and hence the number of collisions in the correct orientation per second)
but also the fraction of fast molecules which have sufficient (kinetic) energy equal to or
greater than Ea, to undergo effective collisions, increases too.
Transition-State Theory (Theory of Activated Complex). Collision theory
explains some features of a reaction, but it does not explain the role of the activation
energy. We said previously that reactant particles must possess a certain minimum amount
of energy in order to react. According to the collision model, this energy comes from the
kinetic energies of the colliding particles and raising the temperature, increases the fraction
of molecules having very high kinetic energies. Theory of activated complex assumes that
reactant particles form an activated complex before are transformed into products.
Activated complex is an unstable atomic group (arrangement) of all particles of the
reactants that are involved in the elementary act of transformation. For example, when the
reactant molecules, A2 and B2 come together with the
proper orientation and sufficient energy an activated
complex is formed in which the old bonds A-A and B-B are
being broken and new bonds A-B are being formed.
Schematically, the process in which reactant particles
undergo effective collisions and formed a high energy
intermediate is described by the scheme given above. The state of the system when the
existing bonds are not entirely broken and the new bonds ones are not completely formed is
termed intermediate or active complex. This group of atoms is very rich in energy and
hence is unstable. In it, atoms are so close so that can exchange electrons. In this phase
electrons are re-group and the electrons in the existing bonds will start to connect the atoms
of the products. Activated complex than, decomposes spontaneously either to form the
reaction products or to go back to the reactants releasing energy.
A reaction profile in collision theory is a graph showing the variation in potential
energy as one reactant molecule approaches another and the products then separate.
(NOTE: Recall that potential energy coming up from the bonds with which atoms are held
together and the interactions between particles (not speed), in this case the separation of
the two reactant molecules as they approach, react, and then separate as products.) The
reaction profile follows energy changes during the course of reaction.
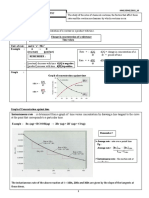
Now look at the reaction between carbon monoxide (CO) gas and nitrogen dioxide
(NO2) gas at a temperature above 500 K. Reaction is described by the following equation:
CO(g) + NO2(g) CO2(g) + NO(g)
The graph depicts schematically the changing potential energy of the two molecules that
approach, collide, and then go on to form products. The activation energy is the height of
the barrier above the potential energy of the reactants.
Potential energy, kJ
Reaction path (reaction coordinate or reaction progress)
Vertical axis in the graph represents the potential energy of the two reactant molecules that
are far apart from each other (in the beginning) while the horizontal coordinate shows the
reaction path. In the course of this reaction, an oxygen atom is transferred from nitrogen
dioxide to carbon monoxide. In order for the reaction to occur, the oxygen atom in NO2 must
collide with the carbon atom in CO. Reaction won't occur if the oxygen end of the NO2
molecule collides with the same atom on CO. Nor will it occur if nitrogen atom on NO2
collides with the carbon atom on CO (figure).
Figure. The collisions in a and b do not result in a reaction because the
molecules are not in position to form bonds. The molecules in are in c the
correct orientation when they collide, and a reaction occurs. The molecules in
d are also in the correct positions on collision, but insufficient energy at the
point of collision prevents a chemical reaction.
As the C end of a CO molecule approaches one of the oxygens in NO2, the electron clouds
surrounding the molecules begin to repel each other and the energy of the system rises
(see the above curve). If the two colliding molecules do not have at least 134 kJ mol1
between them, the O atom cannot be forced close enough to the C atom to transfer its
allegiance from the N atom. The process of breaking bonds thus requires energy, so nearly
all reactions require some input of energy, called activation energy. Increasing the
temperature increases the number of molecules with enough
kinetic energy to climb the activation energy barrier and to start to
react. The NO bond, then, begins to break; so that a short-lived,
activated complex is formed. Its structure is shown on the right
figure (the dashed lines indicate bonds which are just beginning to
form or are in the process
of breaking). As the reaction
occurs, the NO bond gets
longer while the OC
distance continually gets
shorter. Consequently the
difference between the
NO bond length and the
CO bond length becomes
larger and larger during the
reaction.
An
activated
complex is a temporary,
unstable arrangement of
atoms that may form
products or may break apart
to re-form the reactants.
Because
the
activated
complex is as likely to form reactants as it is to form products, it is sometimes referred to as
the transition state. An activated complex is the first step leading to the resulting chemical
reaction. Transition states have extremely short lifetime (less than about 10-13 seconds), so
they are extremely difficult to detect. We therefore have very little experimental knowledge
about them.
The potential energy reaches a peak when the CO and NO2 molecules are highly
distorted. Then it starts to decrease as the new bonds are formed. At separations, the
potential energy rapidly falls to a low value as the product molecules separate from one to
each other.
In addition to the energies of the reactants and products, this diagram shows the
activation energy of the reaction. Activation energy can be thought of as a barrier the
reactants must overcome in order to form the products. In this case, the CO and NO2
molecules collide with enough energy to overcome the barrier, and the products formed lie
at a lower energy level. Because heat energy is given out to the surroundings this process
is exothermic. For many reactions, the process from reactants to products is reversible.
Formation of gaseous products is reversible, endothermic reaction. In this reverse
reaction, the reactants, which are the molecules that were formed in the exothermic forward
reaction, lie at a low
energy level and must
overcome
significant
activation energy to
reform CO and NO2.
This requires an input of
energy. If this reverse
reaction is achieved, CO
and NO2 again lie at a
high energy level.
(Figure;
In
the
reverse endothermic reaction, the reactant molecules
lying at a low energy level
must absorb energy to
overcome
the
activation
energy barrier and form highenergy products.)
Activated Complex Theory Summary
1. This theory suggests that as reactant molecules
approach each other closely they are momentarily
in a less stable state than either the reactants or the
products. In this less stable state, the atoms rearrange themselves, original bonds are weakened
and new bonds are partially formed.
2. The potential energy of the system increases at this
point because:
- the approaching reactant molecules must overcome
the mutual repulsive forces between the outer shell
electrons of their constituent atoms;
- atoms must be separated from each other as bonds
are broken.
3. This increase in potential energy corresponds to an
energy barrier over which the reactant molecules
must pass if the reaction is to proceed. The
arrangement of atoms at the maximum of this
energy barrier is called activated complex or
transition state and is a transitory intermediate state
between reactant and product.
4. The energy difference between the reactants and
the potential energy maximum is referred to as the
activation energy.
Water synthesis reaction profile
You might also like
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5795)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (588)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (895)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (400)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (345)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1091)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (121)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- Synthesis of Propylene Glycol Methyl Ether Catalyzed by Imidazole Polymer Catalyst: Performance Evaluation, Kinetics Study, and Process SimulationDocument10 pagesSynthesis of Propylene Glycol Methyl Ether Catalyzed by Imidazole Polymer Catalyst: Performance Evaluation, Kinetics Study, and Process SimulationPaola OlveraNo ratings yet
- Kinetics PDFDocument1 pageKinetics PDFAlizaNo ratings yet
- Problem As Cat Al Is Is 2018Document10 pagesProblem As Cat Al Is Is 2018Victor Daniel Martínez CarreteroNo ratings yet
- Protein Extraction From Lupin Seeds-A Mathematical ModelDocument11 pagesProtein Extraction From Lupin Seeds-A Mathematical Modeltbbdocument1s0% (1)
- Hydrogen Reduction Cobalt-Chromium Spinel Oxides. Stoichiometric Cobalt ChromiteDocument6 pagesHydrogen Reduction Cobalt-Chromium Spinel Oxides. Stoichiometric Cobalt ChromiteDuongNo ratings yet
- Cantera12 f90Document120 pagesCantera12 f90Siva Ramakrishna ValluriNo ratings yet
- Reaction Rates: Section 3 Reaction Rate LawsDocument3 pagesReaction Rates: Section 3 Reaction Rate LawsRicki HanNo ratings yet
- V VI+Sem+Chemical+Syllabus+2020 2021Document67 pagesV VI+Sem+Chemical+Syllabus+2020 2021Voora GowthamNo ratings yet
- Materi Kuliah Biokimia Stmi Ke 11 (Enzyme Kinetics)Document72 pagesMateri Kuliah Biokimia Stmi Ke 11 (Enzyme Kinetics)soni_abdullahNo ratings yet
- 98 Hydration Kinetics Modeling of Cement PasteDocument11 pages98 Hydration Kinetics Modeling of Cement PasteDavid J. CruzNo ratings yet
- Reactor DesignDocument16 pagesReactor DesignHeather EllaineNo ratings yet
- Manfred Baerns Catalyst Deactivation 110121 PDFDocument38 pagesManfred Baerns Catalyst Deactivation 110121 PDFRaghunath VenkatramananNo ratings yet
- I Sem B.pharmacy SyllabusDocument14 pagesI Sem B.pharmacy SyllabusSUMIT KUMAR PANDITNo ratings yet
- Enzymes PDFDocument50 pagesEnzymes PDFMuhammad Luqman NisaNo ratings yet
- 5 Enzyme KineticsDocument39 pages5 Enzyme KineticsEbook Download100% (1)
- XII Chemistry QuestionBank Hathim HssliveDocument9 pagesXII Chemistry QuestionBank Hathim HsslivekeerthyNo ratings yet
- Factors Affecting Enzyme ActivityDocument32 pagesFactors Affecting Enzyme ActivityHus Arif100% (1)
- Etoos Aiets Planner 2Document2 pagesEtoos Aiets Planner 2FATHIMANo ratings yet
- Biology Chapter 3 EnzymesDocument4 pagesBiology Chapter 3 EnzymesAhmedNo ratings yet
- Advancedchemistry-Lecture Slides-Kinetics Lessons Student VersionDocument26 pagesAdvancedchemistry-Lecture Slides-Kinetics Lessons Student VersionJavier Blanco AlvarezNo ratings yet
- General Chemistry 2 PDFDocument202 pagesGeneral Chemistry 2 PDFBruce Edward WilliamsNo ratings yet
- Instantaneous Rate: Is Determined From A Graph of Time Versus Concentration by Drawing A Line Tangent To The CurveDocument13 pagesInstantaneous Rate: Is Determined From A Graph of Time Versus Concentration by Drawing A Line Tangent To The CurveSupia NazmaNo ratings yet
- Hatta NumberDocument17 pagesHatta NumberShyamPanthavoorNo ratings yet
- Biological Oxidation: Dr. Hariharan. V Assistant Professor Karpagam Faculty of Medical Sciences CoimbatoreDocument6 pagesBiological Oxidation: Dr. Hariharan. V Assistant Professor Karpagam Faculty of Medical Sciences CoimbatoreGorav SharmaNo ratings yet
- Fe (II) - Activated Persulfate Oxidation Effectively Degrades Iodoform in Water: in Uential Factors and Kinetics AnalysisDocument9 pagesFe (II) - Activated Persulfate Oxidation Effectively Degrades Iodoform in Water: in Uential Factors and Kinetics AnalysissalwaNo ratings yet
- I. Multiple Choice. Write The Best Answer From The Following ChoicesDocument5 pagesI. Multiple Choice. Write The Best Answer From The Following ChoicesDoom Refuge100% (1)
- Introducción A Los Bioreactores - Reactor CSTR PDFDocument20 pagesIntroducción A Los Bioreactores - Reactor CSTR PDFmaikNo ratings yet
- Enzyme Action: Learning Objective: To Describe Different Models ForDocument28 pagesEnzyme Action: Learning Objective: To Describe Different Models ForMatt BarhamNo ratings yet
- 03 Chemical KineticsDocument39 pages03 Chemical KineticsEman GamalNo ratings yet
- Week 2 Chemical Kinetics Analysis of Rate EquationDocument31 pagesWeek 2 Chemical Kinetics Analysis of Rate EquationPeachYpeachasNo ratings yet