Professional Documents
Culture Documents
Effect of Residence Time on Hydrolysis Reaction
Uploaded by
Elaine PuiOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Effect of Residence Time on Hydrolysis Reaction
Uploaded by
Elaine PuiCopyright:
Available Formats
Module 5:
1.0
BKF2741 Chemical Reaction Engineering Laboratory I
Title of Experiment
Effect Of Residence Time For Hydrolysis Reaction Of Ethyl Acetate In Turbular Flow Reactor
2.0
3.0
Objective
To carry out a hydrolysis reaction of EtAC in a TFR.
To determine the reaction rate constant.
To determine the effect of residence time on the conversion in a TFR.
Introduction
Catalytic Tubular Reactor has been designed for experiments on chemical reactions in liquid phase
under isothermal and adiabatic conditions. The unit comes complete with a jacketed tubular reactor,
reactant feed tank and pump, temperature sensors and conductivity measuring sensors. The reactor will
enable students to conduct the acid-catalysed liquid-phase hydrolysis of ethyl acetate (EtAC) to ethanol
(EtOH) and acetic acid (HAc), using an immobilized anion ion-exchange resin as catalyst:
CH3COOC2H5 + H2O = CH3COOH + C2H5OH
The tubular flow reactor (TFR) (sometimes called plug flow reactor (PFR)) is also commonly used in
industry in addition to the CSTR and batch reactor. It consists of a cylindrical pipe and is normally
operated at steady state. For analysis purposes, the flow in the system is considered to be highly turbulent
and may be modeled by that of plug flow. Thus, there is no radial variation in concentration along the
pipe.In the tubular reactor, the reactants are continually consumed as they flow down the length of the
reactor. In modeling the tubular reactor, the concentration is assumed to vary continuously in the axial
direction through the reactor. Consequently, the reaction rate, which is a function of concentration for all
but zero order reactions, will also vary axially.
__________________________________________________________________________________
FKKSA/BKF2741/Module/Rev01/2/08/10
Module 5:
BKF2741 Chemical Reaction Engineering Laboratory I
TT
02
Q
02
TT
04
Hot Circulator
Glass
Beads
Reactor
Catalyst
Packing
TT
03
V3
FI
01
Q
01
Heat
Exchanger
TT
01
V2
V1
Drain
Peristaltic
Pump
V4
V6
Drain
V5
Drain
Feed Tank
Product Tank
Figure 1: Process flow diagram for the catalytic tubular reactor unit.
FA0
FA
FA(y)
V
FA(y +
y)
Figure 2: Balance on the Tubular flow reactor (TFR)
__________________________________________________________________________________
FKKSA/BKF2741/Module/Rev01/2/08/10
Module 5:
BKF2741 Chemical Reaction Engineering Laboratory I
To develop the TFR design equation, the reactor volume shall be divided into a number of subvolumes
so that within each subvolume V, the reaction may be considered spatially uniform. Assuming that the
subvolume is located a distance y from the entrance of the reactor, then FA(y) is the molar flow rate of A
into volume V and FA(y + y) is the molar flow rate of A out of the volume. In a spatially uniform
subvolume V,
rAdV rA V
(1)
For a tubular reactor at steady state, the general mole balance is reduced to,
dN A
dt
FA ( y ) FA ( y y ) rA V
(2)
In the above expression, rA is an indirect function of y. That is, rA is a function of reactant concentration,
which is a function of the position, y down the reactor. The volume, V is the product of the crosssectional area, A of the reactor and the reactor length, y.
V Ay
(3)
Substituting Equation (3) into Equation (2) yields,
F ( y y ) FA ( y )
A
ArA
y
(4)
Taking the limit as y approaches zero,
F ( y y ) FA ( y ) dFA
lim A
dy ArA
y 0
y
(5)
It is usually most convenient to have the reactor volume, V rather than the reactor length, y as the
independent variable. Accordingly, the variables Ady can be changed to dV to obtain this form of the
design equation for a TFR,
dFA
rA
dV
(6)
Note that for a reactor in which the cross-sectional area, A varies along the length of the reactor, the
design equation remains unchanged. This means that the extent of reaction in a plug flow reactor does
not depend on its shape, but only on its total volume.
__________________________________________________________________________________
FKKSA/BKF2741/Module/Rev01/2/08/10
Module 5:
BKF2741 Chemical Reaction Engineering Laboratory I
If FA0 is the molar flow rate of species A fed to a system operated at steady state, the molar flow rate at
which species A is reacting within the entire system will be [FA0X]. The molar feed rate of A to the
system minus the rate of reaction of A within the system equals the molar flow rate of A leaving the
system, FA . This is shown in mathematical form to be,
FA
FA0 FA 0 X
FA0 (1 X )
(7)
The entering molar flow rate FA0 is just the product of the entering concentration C A0 and the entering
volumetric flow rate v 0 ,
FA0
C A 0v 0
(8)
Combining Equation [4-7] and Equation [4-6] yields the design equation with a conversion term for the
TFR,
FA0
dX
rA
dV
(9)
Rearranging and integrating Equation 9 with the limit V = 0 when X = 0, we obtain the plug-flow
reactor volume necessary to achieve a specified conversion X,
V FA 0
X
0
dX
rA
(10)
__________________________________________________________________________________
FKKSA/BKF2741/Module/Rev01/2/08/10
Module 5:
BKF2741 Chemical Reaction Engineering Laboratory I
4.0
Experimental Procedure
4.1
Equipment
4.3
4.4
1. TFR
2. Beakers
3. Measuring Cylinders
5. Ethyl Acetate
6. Deionised water
4. Stop Watch
General Start-Up Procedures
1.
Ensure that all valves are initially closed.
2.
Prepare a 20 liter of ethyl acetate solution by adding 800mL of ethyl acetate into 19.2 L of
deionised water.
3.
Turn on the power for the contol panel.
4.
Connect the water de-ionizer to the laboratory water supply. Fill up feed tank B1 with the
19.2 L of de-ionized water. Then add in 800mL of ethyl acetate to prepare the feed solution.
5.
The unit is now ready for experiment.
General Shut-Down Procedures
1.
Switch off the pump.
2.
Switch off the heater.
3.
Keep the cooling water circulating through the reactor while the stirrer motor is running to
allow the water jacket to cool down to room temperature.
4.
If the equipment is not going to be used for long period of time, drain all liquid from the unit.
Rinse the feed tanks with clean water.
5.
4.5
Turn off the power for the control panel.
Experiment Procedures
1.
Perform the general start-up procedures.
2.
Open valves V1 and V4.
3.
Allow EtAC solution to enter the tubular reactor.
4.
Adjust the flowrate controller to give a constant flow rate of 40 ml/min.
__________________________________________________________________________________
FKKSA/BKF2741/Module/Rev01/2/08/10
Module 5:
5.
BKF2741 Chemical Reaction Engineering Laboratory I
Start monitoring the inlet (QI-01) and outlet (QI-02) conductivity values until they do not
change over time. This is to ensure that the reactor has reached steady state.
6.
Record both inlet and outlet steady state conductivity values. Find the concentration of HAc
exiting the reactor and extent of conversion from the calibration curve.
7.
Repeat the experiment (steps 4 to 6) for different residence times by changing the feed flow
rates of EtAC to 30, 50 and 60 mL/min.
5.0
Data Collection
1.
2.
Record data using the Table 1
Plot a graph of conversion vs. residence time. The reactors residence time is defined as the
reactor volume divided by the total feed flow rates.
Residence time,
3.
VTFR
v0
For a first order reaction with initial reactant concentration C A0 , the rate law is shown to
be:
rA kCA kCA0 (1 X )
rA FA0
VTFR
where: X
v0
k
dX
dX
v 0C A 0
dV
dV
X
0
v0
dX
ln(1 X )
(1 X )
k
= extent of conversion
C A = exit concentration of reactant EtAC [M]
v0
= inlet flow rate of solution [mL/min]
C A0 = inlet concentration of reactant in the reactor [M]
Reactor volume
= _____ L
Concentration of EtAC in feed vessel
_____ M
__________________________________________________________________________________
FKKSA/BKF2741/Module/Rev01/2/08/10
Module 5:
BKF2741 Chemical Reaction Engineering Laboratory I
No.
Flow rate of
Residence
EtAC
time, (min)
(mL/min)
Inlet
conductivity
(mS/cm)
Outlet
conductivity
(mS/cm)
Exit
concentration of
HAc (M)
Conversion,
X (%)
1
2
3
4
5
6
7
8
9
10
4. For constant TFR volume, flow rate and initial concentrations, the reaction rate
constant is,
v0
ln(1 x)
VTFR C A0
5. For different reactor residence times, calculate the value of the reaction rate constant, k
and the rate of reaction, r A .
__________________________________________________________________________________
FKKSA/BKF2741/Module/Rev01/2/08/10
Module 5:
BKF2741 Chemical Reaction Engineering Laboratory I
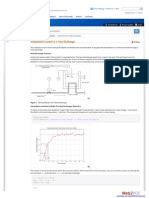
Appendix A : SAMPLE CALIBRATION CURVE AT 30C
Calibration Curve at 30C
2.50
Conductivity (mS/cm)
2.00
1.50
y = -0.0003x6 + 0.0101x5 - 0.1161x4 + 0.6709x3 - 2.0634x2 + 3.2862x
R = 0.9635
1.00
0.50
0.00
0.00
1.00
2.00
3.00
4.00
5.00
6.00
7.00
8.00
9.00
10.00
Concentration (mol/liter)
__________________________________________________________________________________
FKKSA/BKF2741/Module/Rev01/2/08/10
You might also like
- Module 05 Catalytic Tubular Reactor (TFR)Document8 pagesModule 05 Catalytic Tubular Reactor (TFR)Farizal RahmatNo ratings yet
- CSTR Saponification Reaction AnalysisDocument26 pagesCSTR Saponification Reaction AnalysisHazieqahNo ratings yet
- Lab Report CSTR 40LDocument26 pagesLab Report CSTR 40LAnonymous NyvKBW33% (3)
- CSTR 40 LDocument28 pagesCSTR 40 LCik Tiem Ngagiman50% (2)
- Lab CSTRDocument24 pagesLab CSTRNurtasha Atikah50% (2)
- Exp5 Teo ResultsanddiscussionDocument21 pagesExp5 Teo ResultsanddiscussionGökhan CesurNo ratings yet
- Module 04 Continuous Stirred Tank Reactor (CSTR)Document6 pagesModule 04 Continuous Stirred Tank Reactor (CSTR)Farah -HNo ratings yet
- Plug Flow ReactorDocument15 pagesPlug Flow ReactorSeiji Kyousei91% (11)
- Lab RPRT (CSTR)Document21 pagesLab RPRT (CSTR)Black White80% (5)
- PFR Lab ReportDocument16 pagesPFR Lab Reportcog0812No ratings yet
- CSTR 40LDocument16 pagesCSTR 40LhishamNo ratings yet
- Continuous Stirred Tank Reactor (CSTR)Document6 pagesContinuous Stirred Tank Reactor (CSTR)Elaine PuiNo ratings yet
- Plug Flow Reactor ExperimentDocument16 pagesPlug Flow Reactor ExperimentN Afiqah RazakNo ratings yet
- 1.0 Title of Experiment: BKF2741 Chemical Reaction Engineering Laboratory IDocument4 pages1.0 Title of Experiment: BKF2741 Chemical Reaction Engineering Laboratory IAs Eleyana100% (2)
- UTM Chemical Engineering Lab Report OptimizationDocument15 pagesUTM Chemical Engineering Lab Report OptimizationNabilah SyaheeraNo ratings yet
- Module 07 Batch ReactorDocument4 pagesModule 07 Batch ReactorFarah -HNo ratings yet
- CSTR Reaction Order and Rate ConstantDocument21 pagesCSTR Reaction Order and Rate Constantirfan hilmanNo ratings yet
- CSTR Saponification Reaction StudyDocument18 pagesCSTR Saponification Reaction StudyEmily Swan50% (4)
- CSTR 40L LAB EXPERIMENTDocument18 pagesCSTR 40L LAB EXPERIMENTSaber Minato Azrul100% (2)
- L2 Plug Flow Reactor Cover PageDocument23 pagesL2 Plug Flow Reactor Cover PageShahrizatSmailKassim100% (1)
- Module 06 Plug Flow Reactor (PFR)Document4 pagesModule 06 Plug Flow Reactor (PFR)Farah -HNo ratings yet
- Effect of Temperature on CSTR ReactionDocument22 pagesEffect of Temperature on CSTR Reactionafnan_lion94No ratings yet
- Batch ReactorDocument4 pagesBatch ReactorFoo Xiao BingNo ratings yet
- CRE Lab ManualDocument19 pagesCRE Lab ManualMayursinh Solanki100% (1)
- Lab ManualDocument24 pagesLab ManualAasia FarrukhNo ratings yet
- Experiment No: 3: ObjectivesDocument14 pagesExperiment No: 3: ObjectivesAlimah Azeli100% (1)
- CSTR ExperimentDocument5 pagesCSTR ExperimentValentinoDullSatinNo ratings yet
- Understanding Continuous Stirred Tank Reactors (CSTRs) for Chemical Reactions (38 charactersDocument12 pagesUnderstanding Continuous Stirred Tank Reactors (CSTRs) for Chemical Reactions (38 charactersAisyah Addia AzizanNo ratings yet
- Exp 1 Packed Column DistillationDocument12 pagesExp 1 Packed Column DistillationLuqman WasirNo ratings yet
- PFR ReactorDocument19 pagesPFR Reactorkhairi100% (1)
- CSTR 40LDocument11 pagesCSTR 40LSeiji Kyousei100% (1)
- Understanding CSTR Dynamics with a Step Change Input ExperimentDocument16 pagesUnderstanding CSTR Dynamics with a Step Change Input ExperimentAhmadAzriMohamad50% (2)
- Objective Theory Apparatus Procedure Result Sample of Calculation Discussion Conclusion Recommendation Reference AppendicesDocument19 pagesObjective Theory Apparatus Procedure Result Sample of Calculation Discussion Conclusion Recommendation Reference Appendicesahmad pidotNo ratings yet
- Chemical Reaction Engineering 40 L CSTR SaponificationDocument21 pagesChemical Reaction Engineering 40 L CSTR SaponificationMohamad Turmizi Jaafar67% (3)
- CSTR in SeriesDocument12 pagesCSTR in Seriesmnizamarzuki0% (2)
- Continuous Distillation Column Lab Report (40Document27 pagesContinuous Distillation Column Lab Report (40JimNo ratings yet
- Lab 3 Plug FlowDocument29 pagesLab 3 Plug FlowHikaru MokaNo ratings yet
- Continuous Stirred Tank Reactor (40 L)Document16 pagesContinuous Stirred Tank Reactor (40 L)Mohd Zhariff75% (4)
- Plug Flow ReactorDocument28 pagesPlug Flow ReactorNurul AinNo ratings yet
- Lab Report Effect of Residence Time On TDocument26 pagesLab Report Effect of Residence Time On TMuhammad IqmalNo ratings yet
- Chme 401 Chemical Engineering Laboratory Ii Experiment 401-4 Chemical Reactors ObjectiveDocument4 pagesChme 401 Chemical Engineering Laboratory Ii Experiment 401-4 Chemical Reactors ObjectiveDyako D TaherNo ratings yet
- Modelling and Simulation of Saponification Reaction in Different Type of ReactorDocument9 pagesModelling and Simulation of Saponification Reaction in Different Type of ReactorrahulNo ratings yet
- Effect of Residence Time on Saponification Reaction in a Plug Flow ReactorDocument21 pagesEffect of Residence Time on Saponification Reaction in a Plug Flow ReactorValentinoDullSatin100% (1)
- 4 - (PFR BP101)Document15 pages4 - (PFR BP101)Aisyah Addia AzizanNo ratings yet
- Table of ContentsDOCUMENTTOC: CSTR ExperimentDocument19 pagesTable of ContentsDOCUMENTTOC: CSTR ExperimentAmir Al-AimanNo ratings yet
- Plug Flow Reactor (PFR)Document4 pagesPlug Flow Reactor (PFR)Elaine PuiNo ratings yet
- CSTR 40 LDocument20 pagesCSTR 40 LMuhammad NasrulNo ratings yet
- Continuous Stirred Tank Reactor CSTR in Series PDFDocument15 pagesContinuous Stirred Tank Reactor CSTR in Series PDFMuhamad Hafifi AjwadNo ratings yet
- Chapter 2 - 3Document33 pagesChapter 2 - 3Shalini KrishnanNo ratings yet
- PFRDocument19 pagesPFRNur AqilahNo ratings yet
- CSTR in SeriesDocument17 pagesCSTR in SeriesDhiyyah MardhiyyahNo ratings yet
- Exp - 2 Bubble Cap Distillation ColumnDocument13 pagesExp - 2 Bubble Cap Distillation ColumnAdawiyah Al-jufri100% (1)
- Analysis and DiscussionDocument13 pagesAnalysis and Discussiondelwi8788% (8)
- CSTRDocument15 pagesCSTRbilisfreak100% (3)
- TRK1 2013 Chapt 2Document14 pagesTRK1 2013 Chapt 2Putri JulietaNo ratings yet
- Multiphase Catalytic Reactors: Theory, Design, Manufacturing, and ApplicationsFrom EverandMultiphase Catalytic Reactors: Theory, Design, Manufacturing, and ApplicationsNo ratings yet
- Recent Advances in the Science and Technology of Zeolites and Related Materials: Proceedings of the 14th International Zeolite Conference, Cape Town, South Africa, 25-30th April 2004From EverandRecent Advances in the Science and Technology of Zeolites and Related Materials: Proceedings of the 14th International Zeolite Conference, Cape Town, South Africa, 25-30th April 2004No ratings yet
- 1ACii - IS EngDocument2 pages1ACii - IS EngElaine PuiNo ratings yet
- Instrument Security Procedures: ModelDocument1 pageInstrument Security Procedures: ModelElaine PuiNo ratings yet
- 013526216X PDFDocument926 pages013526216X PDFElaine Pui100% (1)
- Burn in Procedure PDFDocument1 pageBurn in Procedure PDFElaine PuiNo ratings yet
- Installing The Software: Dpc/TrackDocument4 pagesInstalling The Software: Dpc/TrackElaine PuiNo ratings yet
- Understanding and Using Ir PDFDocument6 pagesUnderstanding and Using Ir PDFElaine PuiNo ratings yet
- Sigma Paco Meng 0000Document154 pagesSigma Paco Meng 0000matzoneNo ratings yet
- Met/Track Software: Users ManualDocument50 pagesMet/Track Software: Users ManualElaine PuiNo ratings yet
- Especificaciones Tecnicas de Tuberias Hpde MineraDocument34 pagesEspecificaciones Tecnicas de Tuberias Hpde MineraReynaldo SanchezNo ratings yet
- 67max - Fluke ManualDocument22 pages67max - Fluke ManualElaine PuiNo ratings yet
- 2019 Book InternetOfThingsFromHypeToReal PDFDocument393 pages2019 Book InternetOfThingsFromHypeToReal PDFTawab Ullas100% (2)
- Illustrator For Beginners Tastytuts PDFDocument25 pagesIllustrator For Beginners Tastytuts PDFBejenaru GeorgeNo ratings yet
- Headers Footers Microsoft WordDocument4 pagesHeaders Footers Microsoft WordElaine PuiNo ratings yet
- Thermolyne Oven: Operation Manual and Parts ListDocument52 pagesThermolyne Oven: Operation Manual and Parts ListElaine PuiNo ratings yet
- ASTM D2000 Standard Test MethodsDocument6 pagesASTM D2000 Standard Test MethodsHazel EbordeNo ratings yet
- Solar Simulator Safety TipsDocument7 pagesSolar Simulator Safety TipsElaine PuiNo ratings yet
- New - AIAG - VDA - FMEA - Whitepaper - 1Document5 pagesNew - AIAG - VDA - FMEA - Whitepaper - 1Ulysses CarrascoNo ratings yet
- ISPM 15 ED en 2017-02-10Document47 pagesISPM 15 ED en 2017-02-10Utpal ChakabortyNo ratings yet
- SCAC 2019 FMEA Handbook SG Final 13 Sept 2019 PDFDocument20 pagesSCAC 2019 FMEA Handbook SG Final 13 Sept 2019 PDFSilverio Acuña100% (1)
- AIAG & VDA FMEA Handbook Errata SheetDocument8 pagesAIAG & VDA FMEA Handbook Errata Sheetr aNo ratings yet
- Hpa Ammonia Toxicological Overview v2Document13 pagesHpa Ammonia Toxicological Overview v2Elaine PuiNo ratings yet
- Result Citrex 2015 - StudentDocument15 pagesResult Citrex 2015 - StudentElaine PuiNo ratings yet
- Determination of Head Loss in Fixed & Fluidised BedDocument6 pagesDetermination of Head Loss in Fixed & Fluidised BedElaine PuiNo ratings yet
- Result Citrex 2015 - StaffDocument9 pagesResult Citrex 2015 - StaffElaine PuiNo ratings yet
- Multiple ReactionsDocument8 pagesMultiple ReactionsElaine PuiNo ratings yet
- Effect of Pressure, Mixing and Temperature on Acetic Acid-Baking Soda ReactionDocument11 pagesEffect of Pressure, Mixing and Temperature on Acetic Acid-Baking Soda ReactionElaine PuiNo ratings yet
- Signal TransductionDocument17 pagesSignal TransductionElaine PuiNo ratings yet
- Autothermal Reforming of Methane To Synthesis Gas Modeling and Simulation 2009 International Journal of Hydrogen EnergyDocument9 pagesAutothermal Reforming of Methane To Synthesis Gas Modeling and Simulation 2009 International Journal of Hydrogen EnergyElaine PuiNo ratings yet
- Plug Flow Reactor (PFR)Document4 pagesPlug Flow Reactor (PFR)Elaine PuiNo ratings yet
- Heat of Chemical ReactionsDocument9 pagesHeat of Chemical ReactionsWeng KhimNo ratings yet
- JavaDocument20 pagesJavaSivasakthi MarimuthuNo ratings yet
- Bar BendingDocument142 pagesBar Bendingjaffna100% (4)
- Battery ModelingDocument6 pagesBattery ModelingmusabnajeebNo ratings yet
- Dell Emc Data Domain Dd3300 Faq: Frequently Asked QuestionsDocument14 pagesDell Emc Data Domain Dd3300 Faq: Frequently Asked QuestionseriquewNo ratings yet
- V Single Vane Pumps: 1 - "F3" 2 - Pump TypeDocument14 pagesV Single Vane Pumps: 1 - "F3" 2 - Pump TypeJose Leandro Neves FerreiraNo ratings yet
- Diebold ManualDocument2 pagesDiebold Manualjoseperezlopes069No ratings yet
- SAT-101 User Manual: Document No: MAN-0013 Issue No: 4 Dated: 24 Aug 2004Document24 pagesSAT-101 User Manual: Document No: MAN-0013 Issue No: 4 Dated: 24 Aug 2004VM ServicesNo ratings yet
- Temperarura4 PDFDocument371 pagesTemperarura4 PDFmario yanezNo ratings yet
- Student's Error Analysis in Finishing Mathematic Word Problem Based Newman AnalysisDocument11 pagesStudent's Error Analysis in Finishing Mathematic Word Problem Based Newman AnalysisguanyitorNo ratings yet
- Course Notes For GeophysicsDocument30 pagesCourse Notes For GeophysicsShivendra Pratap SinghNo ratings yet
- Practice Paper 1 Biology: WWW - Oxfordsecodary.co - UkDocument11 pagesPractice Paper 1 Biology: WWW - Oxfordsecodary.co - UkOHNo ratings yet
- 5367227Document2 pages5367227aliha100% (1)
- Technology: ControlsDocument32 pagesTechnology: ControlsAli Hossain AdnanNo ratings yet
- Quantum Garage Door OpenerDocument32 pagesQuantum Garage Door OpenerNguiBDNo ratings yet
- Computer Science Project On Management SystemDocument26 pagesComputer Science Project On Management Systemsajid aliNo ratings yet
- Fontargen AJ PDFDocument282 pagesFontargen AJ PDFantonyNo ratings yet
- Discoverer Installation and Integration With EBSDocument25 pagesDiscoverer Installation and Integration With EBSRajendra PrasadNo ratings yet
- Effects of Osteoarthritis and Fatigue On Proprioception of The Knee JointDocument5 pagesEffects of Osteoarthritis and Fatigue On Proprioception of The Knee JointRosaneLacerdaNo ratings yet
- Tomescu PDFDocument353 pagesTomescu PDFLuís Farias100% (3)
- Jurnal PterygiumDocument5 pagesJurnal PterygiummirafitrNo ratings yet
- Thrust Bearing CalculationsDocument162 pagesThrust Bearing Calculationslahiru1983100% (1)
- Qual Exam 2005Document31 pagesQual Exam 2005Kevin Lius BongNo ratings yet
- WWW Mathworks inDocument7 pagesWWW Mathworks inRagini SharmaNo ratings yet
- A Stated-Preference Study of The Willingness-To-Pay To Reduce Traffic Risk in Urban vs. Rural RoadsDocument13 pagesA Stated-Preference Study of The Willingness-To-Pay To Reduce Traffic Risk in Urban vs. Rural RoadsSantanu KumarNo ratings yet
- A510m 06Document7 pagesA510m 06psewag100% (1)
- Lecture 5 (River As A Geological Agent)Document20 pagesLecture 5 (River As A Geological Agent)Boos yousufNo ratings yet
- Astm D2899Document6 pagesAstm D2899btwilliNo ratings yet
- Exercise 1: Pressing Cheeses: Cheese ProductionDocument8 pagesExercise 1: Pressing Cheeses: Cheese ProductionAhmed HusseinNo ratings yet
- Seagate Part1 PDFDocument113 pagesSeagate Part1 PDFwaranhaNo ratings yet
- Chapter 5 Lennard Jones PotentialDocument6 pagesChapter 5 Lennard Jones PotentialMuhamad RayhanNo ratings yet