Professional Documents
Culture Documents
NSS Chemistry Part 12 Patterns in Chemical World
Uploaded by
Felix YueCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
NSS Chemistry Part 12 Patterns in Chemical World
Uploaded by
Felix YueCopyright:
Available Formats
HKCEE & HKAL Past Paper Questions: Part 12 Patterns in Chemical World
1. HKAL 1988 I
Elements exhibit periodic behaviour as their atomic number increases.
Illustrate graphically the periodicity of one such properties of the elements from hydrogen (Z=1) to argon
(Z=18).
(2 marks)
2. HKAL 1990 II Q4b
Account for the following observation.
SiO2 is a solid with a high melting point, whereas CO2 is a gas at room temperature.
(4 marks)
SiO2: three-dimensional network solid with very strong Si-O bonds. (1M + 1M)
CO2: exists as discrete molecules with only weak intermolecular van der Waals forces. (1M + 1M)
3. HKAL 1991 II Q4a
(i)
Briefly explain what is meant by periodicity in the chemical properties of the elements.
(ii)
Draw diagrams showing the variations in melting point (or boiling point) for the elements from lithium to
argon.
(5 marks)
4.
HKAL 1998 I Q3b
Sketch the trends for the properties below, and account for the trend in each case.
(i)
melting point of the alkali metals, Li, Na and K
(ii)
boiling point of the period 3 elements, Na, Mg and Al.
(4 marks)
5. HKAL 1999 I Q1b
Account for the following:
At 298K and 1 atm pressure, carbon dioxide is a gas whereas silicon dioxide is a solid.
(1 mark)
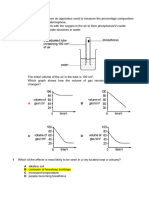
6. HKAL 20022 II Q2c
The graph below
b
showss the variatioons of meltinng points and
d boiling poinnts of the Perriod 3 elemen
nts.
Explain whhy
(i)
silicoon, a metalloiid, has a veryy high meltinng point;
(ii)
the booiling pointss of the metalls are in the order;
o
Al > Mg > Na;
(iii) there is generally a larger diffference betweeen the meltiing point andd the boiling point for meetals than forr
non- metals;
m
(iv) the melting
m
point of sulphur iss the highest among the non-metals.
n
(9 marrks)
7. HKAL 20055 I Q1b
(i)
Sketcch the variatiion in electrical conductivvity of the Period 3 elem
ments from soodium to argo
on at
room
m
tempeerature and atmospheric
a
pressure.
(ii) Explainn the variatioon in (i).
(5 marrks)
8. HKAL 2006 I Q1a
The table below lists the melting points of three oxides of the Period 3 elements:
Oxide
Na2O
Al2O3
SO2
Melting point /
920
2040
75
Account for the large difference in the melting points of the three oxides.
(3 marks)
9. HKAL 2008 II Q1b
Both sodium and chlorine are elements in Period 3 of the Periodic Table.
(i)
At room temperature and atmosphere pressure, Na 2 O is a solid with a very high melting point
whereas Cl 2 O is a gas. Account for this difference in property between Na 2 O and Cl 2 O .
(ii)
Na 2 O 2 is another oxide of sodium. Write the chemical equation for the reaction of Na 2 O with water
and that of Na 2 O 2 with water.
(iii)
Cl 2 O 7 is another oxide of chlorine. Give the products formed when Cl 2 O and Cl 2 O 7 are treated
separately with water.
(6 marks)
10. HKAL 1990 I Q3d
Illustrate, with examples using nickel, two characteristic properties of transition metals.
(2 marks)
Catalytic property: the use of nickel catalyst in the hydrogenation of oils to make margarine.
Variable oxidation states: Ni2O3, NiO, NiO2 etc
Form coloured ions: Ni2+(aq) is green
You might also like
- NSS Chemistry Part 2 Structural Questions and AnswersDocument22 pagesNSS Chemistry Part 2 Structural Questions and AnswersFelix YueNo ratings yet
- NSS Chemistry Part 7 Redox Reactions Chemical Cells and Electrolysis - LQDocument38 pagesNSS Chemistry Part 7 Redox Reactions Chemical Cells and Electrolysis - LQFelix YueNo ratings yet
- Periodic Table SQDocument17 pagesPeriodic Table SQNg Swee Loong StevenNo ratings yet
- NSS Chemistry Part 11 Chemistry of Carbon CompoundsDocument47 pagesNSS Chemistry Part 11 Chemistry of Carbon CompoundsFelix YueNo ratings yet
- NSS Chemistry Part 9 Rate of ReactionsDocument26 pagesNSS Chemistry Part 9 Rate of ReactionsFelix YueNo ratings yet
- NSS Chemistry Part 13 Industrial Chemistry - IIDocument21 pagesNSS Chemistry Part 13 Industrial Chemistry - IIFelix YueNo ratings yet
- NSS Chemistry Part 4 Acids and Bases - MCDocument35 pagesNSS Chemistry Part 4 Acids and Bases - MCFelix YueNo ratings yet
- NSS Chemistry Part 4 Acids and Bases - LQDocument40 pagesNSS Chemistry Part 4 Acids and Bases - LQFelix YueNo ratings yet
- NSS Chemistry Part 3 Metals - MCDocument20 pagesNSS Chemistry Part 3 Metals - MCFelix YueNo ratings yet
- Redox Reactions, Chemical Cells and Electrolysis (Less than 40 chars: 35 charsDocument38 pagesRedox Reactions, Chemical Cells and Electrolysis (Less than 40 chars: 35 charsミーチェルNo ratings yet
- NSS Chemistry Part 2 Microscopic World I - MCDocument32 pagesNSS Chemistry Part 2 Microscopic World I - MCFelix YueNo ratings yet
- MSS 1718MockPaper1ADocument11 pagesMSS 1718MockPaper1AKelvin Chow100% (1)
- NSS Chemistry Part 7 Redox Reactions Chemical Cells and Electrolysis - MCDocument37 pagesNSS Chemistry Part 7 Redox Reactions Chemical Cells and Electrolysis - MCFelix YueNo ratings yet
- 2015 F6 Mock P2 PDFDocument7 pages2015 F6 Mock P2 PDFKaylie WongNo ratings yet
- Strength of Acids and AlkalisDocument16 pagesStrength of Acids and AlkalisRyanNo ratings yet
- Afterschool Mole Calculation Exercise Ans.Document32 pagesAfterschool Mole Calculation Exercise Ans.J TNo ratings yet
- Hkdse Chemistry - A Modern View: (Second Edition) (Reprinted With Minor Amendments 2019)Document25 pagesHkdse Chemistry - A Modern View: (Second Edition) (Reprinted With Minor Amendments 2019)Tat LNo ratings yet
- QB 1B Ch07e FDocument40 pagesQB 1B Ch07e FRyan KwokNo ratings yet
- Suggested Answers To Practical Workbook: CH 1 Introducing BiologyDocument33 pagesSuggested Answers To Practical Workbook: CH 1 Introducing BiologyCharmine Holmes100% (1)
- Chemical Bonding: Ionic Bonding and Metallic Bonding: Learning GoalDocument36 pagesChemical Bonding: Ionic Bonding and Metallic Bonding: Learning GoalRyanNo ratings yet
- Chem 18 (2nd) PDFDocument36 pagesChem 18 (2nd) PDFRyan100% (1)
- Chapter 1 Temperature and ThermometersDocument4 pagesChapter 1 Temperature and ThermometersZhu JiankunNo ratings yet
- F3 1st ExamDocument10 pagesF3 1st ExamSharon WongNo ratings yet
- NSS Chemistry Part 15 Analytical Chemistry - LQDocument42 pagesNSS Chemistry Part 15 Analytical Chemistry - LQFelix Yue100% (1)
- SSGS 17-18 F.6 Final Exam 1 and 2 CHEMDocument37 pagesSSGS 17-18 F.6 Final Exam 1 and 2 CHEMKelvin ChowNo ratings yet
- Calculation Exercise Answer Key Provides Detailed SolutionsDocument6 pagesCalculation Exercise Answer Key Provides Detailed SolutionsBelladonna Lee100% (1)
- Past Paper - Microscopic WorldDocument7 pagesPast Paper - Microscopic Worldapi-3739994100% (1)
- Baptist Lui Ming Choi Secondary School First Term Examination (2012-2013) Form 3 ChemistryDocument12 pagesBaptist Lui Ming Choi Secondary School First Term Examination (2012-2013) Form 3 ChemistryyuNo ratings yet
- Microscopic World I - Atomic Structure and PropertiesDocument40 pagesMicroscopic World I - Atomic Structure and PropertiesRyanNo ratings yet
- Movement of Substances Across Cell Membrane: Multiple-Choice QuestionsDocument51 pagesMovement of Substances Across Cell Membrane: Multiple-Choice QuestionsRyanNo ratings yet
- Air and AtmosphereDocument12 pagesAir and Atmospherebob leowNo ratings yet
- Gas Exchange in Humans: Multiple-Choice QuestionsDocument56 pagesGas Exchange in Humans: Multiple-Choice QuestionsZ s2021 4C Ma Ka Ki Margaret 4C16No ratings yet
- 4 Transfer Processes: Practice 4.1 (p.109)Document6 pages4 Transfer Processes: Practice 4.1 (p.109)Oscar TSANGNo ratings yet
- Acid and AlkaliDocument9 pagesAcid and Alkali云吸仓鼠吉尼斯保持者No ratings yet
- Structured Questions: HKDSE Chemistry A Modern View Part VIII Chemical Reactions and EnergyDocument21 pagesStructured Questions: HKDSE Chemistry A Modern View Part VIII Chemical Reactions and EnergyNg Swee Loong StevenNo ratings yet
- Atomicstructurequestions PDFDocument42 pagesAtomicstructurequestions PDFNfor KlinsmanNo ratings yet
- Ch5 Food and Humans: Multiple-Choice QuestionsDocument5 pagesCh5 Food and Humans: Multiple-Choice QuestionsYing Hei NgNo ratings yet
- MSS 1718MockPaper2Document8 pagesMSS 1718MockPaper2Kelvin ChowNo ratings yet
- Chapter 6 Work, Energy and PowerDocument12 pagesChapter 6 Work, Energy and PowerZhu Jiankun100% (1)
- HKDSE Biology S5 Exam (2020)Document28 pagesHKDSE Biology S5 Exam (2020)FuchingAuntie100% (1)
- Concentration of SolutionsDocument20 pagesConcentration of SolutionsRyanNo ratings yet
- Chap 2Document72 pagesChap 2miguel0angel0ramos-1100% (6)
- NSS Chemistry Part 2 The Microscopic World HKCEE Past Paper Question The Microscopic World I Ns - Multiple Choice QuestionsDocument32 pagesNSS Chemistry Part 2 The Microscopic World HKCEE Past Paper Question The Microscopic World I Ns - Multiple Choice QuestionsミーチェルNo ratings yet
- MATHEMATICS Compulsory Part Mock Exam 1 Paper 1 Question-Answer BookDocument10 pagesMATHEMATICS Compulsory Part Mock Exam 1 Paper 1 Question-Answer Bookyanai taiNo ratings yet
- 2RM Cb4Aans eDocument76 pages2RM Cb4Aans eTat LNo ratings yet
- Chem 2 (2nd) PDFDocument28 pagesChem 2 (2nd) PDFRyanNo ratings yet
- Reacting MassesDocument2 pagesReacting MassesTsz Wai WONGNo ratings yet
- Chem 6 (2nd) PDFDocument28 pagesChem 6 (2nd) PDFRyanNo ratings yet
- Plants absorb minerals and exchange gasesDocument8 pagesPlants absorb minerals and exchange gasessafbadNo ratings yet
- The Ultimate Question Bank: Dse Chem MasteryDocument48 pagesThe Ultimate Question Bank: Dse Chem MasteryYip AvaNo ratings yet
- CH 3Document15 pagesCH 3Choi MingNo ratings yet
- Exam Paper - Core (By Topic)Document15 pagesExam Paper - Core (By Topic)Henry Leung100% (1)
- Suggested Answers To In-Text Activities and Unit-End Exercises Topic 3 Unit 12Document21 pagesSuggested Answers To In-Text Activities and Unit-End Exercises Topic 3 Unit 12ミーチェルNo ratings yet
- 2006 Form 3 Physics Half-Yearly Exam (Dec 2006)Document4 pages2006 Form 3 Physics Half-Yearly Exam (Dec 2006)Raistlin Chan Ching KitNo ratings yet
- F3 Physics Sample Assignment QuestionsDocument5 pagesF3 Physics Sample Assignment QuestionsAngus AnizNo ratings yet
- Unit 3: Inorganic Chemistry: Recommended Prior KnowledgeDocument4 pagesUnit 3: Inorganic Chemistry: Recommended Prior KnowledgeHubbak KhanNo ratings yet
- 1 BondingDocument50 pages1 BondingSherey FathimathNo ratings yet
- Chem S6Document4 pagesChem S6sugi mugiNo ratings yet
- ASSIGNMENT D & F Block ElementDocument6 pagesASSIGNMENT D & F Block ElementAKVanugrahNo ratings yet
- Section: Site 2Document5 pagesSection: Site 2Hubbak KhanNo ratings yet
- NSS Chemistry Part 15 Analytical Chemistry - LQDocument42 pagesNSS Chemistry Part 15 Analytical Chemistry - LQFelix Yue100% (1)
- NSS Chemistry Part 7 Redox Reactions Chemical Cells and Electrolysis - MCDocument37 pagesNSS Chemistry Part 7 Redox Reactions Chemical Cells and Electrolysis - MCFelix YueNo ratings yet
- NSS Chemistry Part 15 Analytical Chemistry - MCDocument27 pagesNSS Chemistry Part 15 Analytical Chemistry - MCFelix YueNo ratings yet
- NSS Chemistry Part 13 Industrial Chemistry - IIDocument21 pagesNSS Chemistry Part 13 Industrial Chemistry - IIFelix YueNo ratings yet
- NSS Chemistry Part 6 Microscopic World IIDocument20 pagesNSS Chemistry Part 6 Microscopic World IISabrina WongNo ratings yet
- NSS Chemistry Part 13 Industrial Chemistry - IDocument36 pagesNSS Chemistry Part 13 Industrial Chemistry - Izwindows123456789No ratings yet
- NSS Chemistry Part 10 Chemical EquilibriumDocument16 pagesNSS Chemistry Part 10 Chemical EquilibriumFelix YueNo ratings yet
- NSS Chemistry Part 8 Chemical Reactions and EnergyDocument17 pagesNSS Chemistry Part 8 Chemical Reactions and EnergyFelix YueNo ratings yet
- NSS Chemistry Part 3 Metals - LQDocument25 pagesNSS Chemistry Part 3 Metals - LQNicole ChanNo ratings yet
- HKCEE & HKAL Past Paper Questions on Fossil Fuels, Carbon Compounds, and Organic ChemistryDocument84 pagesHKCEE & HKAL Past Paper Questions on Fossil Fuels, Carbon Compounds, and Organic ChemistryFelix YueNo ratings yet
- NSS Chemistry Part 4 Acids and Bases - MCDocument35 pagesNSS Chemistry Part 4 Acids and Bases - MCFelix YueNo ratings yet
- NSS Chemistry Part 4 Acids and Bases - LQDocument40 pagesNSS Chemistry Part 4 Acids and Bases - LQFelix YueNo ratings yet
- NSS Chemistry Part 3 Metals - MCDocument20 pagesNSS Chemistry Part 3 Metals - MCFelix YueNo ratings yet
- NSS Chemistry Part 2 Microscopic World I - MCDocument32 pagesNSS Chemistry Part 2 Microscopic World I - MCFelix YueNo ratings yet
- MT 06Document2 pagesMT 06Glen Rey BrunNo ratings yet
- Assessment - The Bridge Between Teaching and Learning (VFTM 2013)Document6 pagesAssessment - The Bridge Between Teaching and Learning (VFTM 2013)Luis CYNo ratings yet
- BBFH107 - Business Statistics II Assignment IIDocument2 pagesBBFH107 - Business Statistics II Assignment IIPeter TomboNo ratings yet
- KNAW Proceedings from 1919-1920Document481 pagesKNAW Proceedings from 1919-1920Eduardo Salgado EnríquezNo ratings yet
- Filterlist FanboyDocument257 pagesFilterlist FanboyAjitesh MehtaNo ratings yet
- Sale DeedDocument3 pagesSale DeedGaurav Babbar0% (1)
- Eight Principles of Ethical Leadership in EducationDocument2 pagesEight Principles of Ethical Leadership in EducationKimberly Rose Nativo100% (1)
- Bolomeya Model For Normal Concrete Mix DesignDocument6 pagesBolomeya Model For Normal Concrete Mix DesignprincessrandotaNo ratings yet
- English - Method BobathDocument4 pagesEnglish - Method Bobathje_corektNo ratings yet
- All Glory Book-1Document187 pagesAll Glory Book-1fredkayf100% (1)
- Vodafone - Market Research ProposalDocument4 pagesVodafone - Market Research ProposalNikita Sanghvi100% (2)
- Holy Trinity University: Puerto Princesa City Basic Education DepartmentDocument2 pagesHoly Trinity University: Puerto Princesa City Basic Education DepartmentBrian Reyes GangcaNo ratings yet
- B1 Mod 01 MathsDocument152 pagesB1 Mod 01 MathsTharrmaselan VmanimaranNo ratings yet
- Minimizing Dose Is DR PDFDocument4 pagesMinimizing Dose Is DR PDFYamuna GovindarajNo ratings yet
- Yu-Gi-Oh Nightmare Troubadour InfoDocument12 pagesYu-Gi-Oh Nightmare Troubadour InfoBrandon Bradley0% (1)
- Casalla vs. PeopleDocument2 pagesCasalla vs. PeopleJoan Eunise FernandezNo ratings yet
- Affidavit of DesistanceDocument6 pagesAffidavit of Desistancesalasvictor319No ratings yet
- TGT EnglishDocument3 pagesTGT EnglishKatta SrinivasNo ratings yet
- Radio Journalism GuideDocument13 pagesRadio Journalism GuideMark June B. GonzagaNo ratings yet
- Assessing The NeckDocument3 pagesAssessing The NeckAnne Joyce Lara AlbiosNo ratings yet
- Fraud under Indian Contract Act - Essentials and Exceptions to Mere SilenceDocument6 pagesFraud under Indian Contract Act - Essentials and Exceptions to Mere SilenceabhilashNo ratings yet
- Learning Disabilities in The ClassroomDocument5 pagesLearning Disabilities in The ClassroomekielaszekNo ratings yet
- Dilo SpaDocument2 pagesDilo SpajosefNo ratings yet
- Fundamentals of Human Neuropsychology 7th Edition Kolb Test BankDocument26 pagesFundamentals of Human Neuropsychology 7th Edition Kolb Test BankDanielYoungabrn100% (60)
- CBSE Class 10 Maths Sample Paper 2019-20Document33 pagesCBSE Class 10 Maths Sample Paper 2019-20omvrat singhNo ratings yet
- Case - 1 Marketing and Distribution of MushroomDocument2 pagesCase - 1 Marketing and Distribution of MushroomKetan BohraNo ratings yet
- Con Law I - Case Cheat SheetDocument22 pagesCon Law I - Case Cheat SheetPriscilla Quansah100% (1)
- Clinical Mental Health Counseling in Community and Agency Settings 5Th Edition Full ChapterDocument41 pagesClinical Mental Health Counseling in Community and Agency Settings 5Th Edition Full Chapterwilliam.kellar832100% (24)
- Sources of Coherent RadiationDocument7 pagesSources of Coherent RadiationSubhash DhungelNo ratings yet
- EDMU 520 Phonics Lesson ObservationDocument6 pagesEDMU 520 Phonics Lesson ObservationElisa FloresNo ratings yet