Professional Documents
Culture Documents
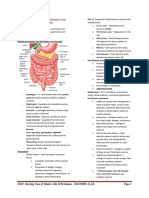
Fluids & Electrolytes
Uploaded by
arvinnnnCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Fluids & Electrolytes
Uploaded by
arvinnnnCopyright:
Available Formats
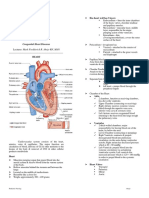
Lecture Notes on Fluids and Electrolytes
Prepared By: Mark Fredderick R Abejo R.N, MAN
STI COLLEGE GLOBAL CITY
College of Nursing
MEDICAL AND SURGICAL NURSING
solute the substance dissolved
solvent substance in which the solute is dissolved
usually water (universal solvent)
molar solution (M) - # of gram-molecular weights of solute
per liter of solution
osmolality concentration of solute per kg of water
normal range = 275-295 mOsm/kg of water
osmolarity concentration of solute per L of solution
* since 1kg=1L, & water is the solvent of the human
body, osmolarity & osmolality are used interchangeably
Fluids and Electrolytes
IV.
Lecturer: Mark Fredderick R. Abejo RN, MAN
________________________________________________
FLUIDS & ELECTROLYTES
I. Fluid Status of Human Body
A. Homeostasis: state of the body when
maintaining a state of balance in the presence
of constantly changing conditions
B. Includes balance of fluid, electrolytes, and acidbase balance
C. Body water intake and output approximately
equal (2500 mL/24 hr.)
Adult body: 40L water, 60% body weight
2/3 intracellular
1/3 extracellular (80% interstitial, 20%
intravascular)
Infant: 70-80% water
Elderly: 40-50% water
II. Body Fluid Composition
A.
Water: 60% of body weight
B.
Electrolytes: substances that become charged
particles in solution
1. Cations: positively charged (e.g. Na+,
K+)
2. Anions: negatively charged (e.g. Cl-)
3. Both are measured in milliequivalents
per liter (mEq/L)
C.
Balance of hydrostatic pressure and osmotic
pressure regulates movement of water
between intravascular and interstitial spaces
III. Body Fluid Distribution:
A.
2 body compartments:
1. Intracellular fluids (ICF): fluids
within cells of body [major
intracellular electrolytes: Potassium
(K+), Magnesium (Mg +2)]
2. Extracellular fluids (ECF): fluid
outside cells; [major extracellular
electrolytes: Sodium (Na+),
Chloride(Cl-)]; this is where
transportation of nutrients, oxygen,
and waste products occurs
B.
Locations of ECF:
1. Interstitial: fluid between most cells
2. Intravascular: fluid within blood
vessels; also called plasma
3. Transcellular: fluids of body
including urine, digestive secretion,
cerebrospinal, pleural, synovial,
intraocular, gonadal, pericardial
MS: Fluids and Electrolyte
Mechanisms of Body Fluid Movement (i.e. movement
of solutes, solvents across different extracellular
locations)
A.
Osmosis: water is mover; water moves from
lower concentration to higher concentration
1. Normal Osmolality of ICF and ECF:
275 295 mOsm/kg
2. Types of solutions according to osmolality
Isotonic: all solutions with osmolality
same as that of plasma .Body cells placed
in isotonic fluid: neither shrink nor swell
Hypertonic: fluid with greater
concentration of solutes than plasma
Cells in hypertonic solution: water in
cells moves to outside to equalize
concentrations: cells will shrink
Hypotonic: fluid with lower concentration
of solutes than plasma Cells in hypotonic
solution: water outside cells moves to
inside of cells: cells will swell and
eventually burst (hemolyze)
3. Different intravenous solutions, used to
correct some abnormal conditions,
categorized according to osmolality:
B.
C.
D.
Diffusion: solute molecules move from higher
concentration to lower concentration
1. Solute, such as electrolytes, is the
mover; not the water
2. Types: simple and facilitated
(movement of large water-soluble
molecules)
Filtration: water and solutes move from area
of higher hydrostatic pressure to lower
hydrostatic pressure
1. Hydrostatic pressure is created by
pumping action of heart and gravity
against capillary wall
2. Usually occurs across capillary
membranes
Active Transport: molecules move across
cell membranes against concentration
gradient; requires energy, e.g. Na K pump
Hydrostatic pressure -pushes fluid out of vessels into tissue
space; higher to lower pressure
due to water volume in vessels; greater in arterial end
swelling: varicose veins, fluid overload, kidney failure
& CHF
Osmotic pressure -pulls fluid into vessels; from weaker
concentration to stronger concentration
- from plasma proteins; greater in venous end
- swelling: liver problems, nephrotic syndrome
Abejo
Lecture Notes on Fluids and Electrolytes
Prepared By: Mark Fredderick R Abejo R.N, MAN
V.
Mechanisms that Regulate Homeostasis:
How the body adapts to fluid and electrolyte changes?
A.
B.
C.
D.
E.
Thirst: primary regulator of water intake
(thirst center in brain)
Kidneys: regulator of volume and
osmolality by controlling excretion of water
and electrolytes
Renin-angiotension-aldosterone
mechanism: response to a drop in blood
pressure; results from vasoconstriction and
sodium regulation by aldosterone
Antidiuretic hormone: hormone to
regulate water excretion; responds to
osmolality and blood volume
Atrial natriuretic factor: hormone from
atrial heart muscle in response to fluid
excess; causes increased urine output by
blocking aldosterone
Fluid Balance Regulation
Thirst reflex triggered by:
1. decreased salivation & dry mouth
2. increased osmotic pressure stimulates
osmoreceptors in the hypothalamus
3. decreased blood volume activates the
renin/angiontensin pathway, which simulates the
thirst center in hypothalamus
ADH produced by hypothalamus, released by posterior
pituitary when osmoreceptor or baroreceptor is
triggered in hypothalamus
Aldosterone produced by adrenal cortex; promotes Na &
water reabsorption
Sensible & Insensible Fluid Loss
Sensible:
Insensible:
urine, vomiting, suctioned secretions
lungs , skin, GI and evaporation
Normal Fluid Intake and Loss in Adults
Intake:
Water in food
Water from oxidation
Water in liquid
Output:
1,000 mls
300 mls
1,200 mls
TOTAL
2,500 mls
Skin
Lungs
Feces
Kidneys
TOTAL
500 mls
300 mls
150 mls
1,500 mls
2,500 mls
Renin-Angiotensin
1. drop in blood volume in kidneys = renin released
2. renin = acts on plasma protein angiotensin
(released by the liver) to form angiotensin I
3. ACE = converts Angiotensin I to Angiotensin II in
the lungs
4. Angiotensin II = vasoconstriction & aldosterone
release
MS: Fluids and Electrolyte
Abejo
Lecture Notes on Fluids and Electrolytes
Prepared By: Mark Fredderick R Abejo R.N, MAN
IV Fluids
Isotonic
Hypotonic
Hypertonic
LR
PNSS (0.9%NSS)
NM
D5W
- isotonic in bag
- dextrose=quickly
metabolized=hypotonic
D2.5W
0.45% NSS
0.3% NSS
0.2% NSS
D50W
D10W
D5NSS
D5LR
3%NSS
Colloids (usually CHONs) & Plasma expanders
Dextran synthetic polysaccharide, glucose solution
increase concentration of blood, improving blood
volume up to 24 hrs
contraindicated: heart failure, pulmonary edema,
cardiogenic shock, and renal failure
Hetastarch like Dextran, but longer-acting
expensive
derived from corn starch
Composition of Fluids
Saline solutions water, Na, Cl
Dextrose solutions water or saline, calories
Lactated Ringers water, Na, Cl, K, Ca, lactate
Plasma expanders albumin, dextran, plasma protein
(plasmanate)
- increases oncotic pressure, pulling fluids
into circulation
Parenteral hyperalimentation fluid, electrolytes, amino
acids, calories
A.
FLUID VOLUME DEFICIT or HYPOVOLEMIA
Definition: This is the loss of extra cellular fluid
volume that exceeds the intake of fluid. The loss of
water and electrolyte is in equal proportion. It can
be called in various terms- vascular, cellular or
intracellular dehydration. But the preferred term is
hypovolemia.
Dehydration refers to loss of WATER alone, with
increased solutes concentration and sodium
concentration
Pathophysiology of Fluid Volume Deficit
Etiologic conditions include:
a.
Vomiting
b. Diarrhea
c. Prolonged GI suctioning
d. Increased sweating
e. Inability to gain access to fluids
f. Inadequate fluid intake
g. Massive third spacing
MS: Fluids and Electrolyte
Risk factors are the following:
a. Diabetes Insipidus
b. Adrenal insufficiency
c. Osmotic diuresis
d. Hemorrhage
e. Coma
f. Third-spacing conditions like ascites,
pancreatitis and burns
PATHOPHYSIOLOGY:
Risk Factors --- inadequate fluids in the body ---- decreased
blood volume ----- decreased cellular hydration ---- cellular
shrinkage ---- weight loss, decreased turgor, oliguria,
hypotension, weak pulse, etc.
ASSESSMENT:
Physical examination
Weight loss, tented skin turgor, dry mucus membrane
Hypotension
Tachycardia
Cool skin, acute weight loss
Flat neck veins
Decreased CVP
Subjective cues
Thirst
Nausea, anorexia
Muscle weakness and cramps
Change in mental state
Laboratory findings
1.
2.
3.
4.
Elevated BUN due to depletion of fluids or decreased
renal perfusion
Hemoconcentration
Possible Electrolyte imbalances: Hypokalemia,
Hyperkalemia, Hyponatremia, hypernatremia
Urine specific gravity is increased (concentrated
urine) above 1.020
NURSING MANAGEMENT
1.
2.
3.
4.
5.
6.
7.
8.
Assess the ongoing status of the patient by doing an
accurate input and output monitoring
Monitor daily weights. Approximate weight loss 1
kilogram = 1liter!
Monitor Vital signs, skin and tongue turgor, urinary
concentration, mental function and peripheral
circulation
Prevent Fluid Volume Deficit from occurring by
identifying risk patients and implement fluid
replacement therapy as needed promptly
Correct fluid Volume Deficit by offering fluids orally
if tolerated, anti-emetics if with vomiting, and foods
with adequate electrolytes
Maintain skin integrity
Provide frequent oral care
Teach patient to change position slowly to avoid
sudden postural hypotension
Abejo
Lecture Notes on Fluids and Electrolytes
Prepared By: Mark Fredderick R Abejo R.N, MAN
B.
FLUID VOLUME EXCESS: HYPERVOLEMIA
Definition : Refers to the isotonic expansion of
the ECF caused by the abnormal retention of
water and sodium
There is excessive retention of water and
electrolytes in equal proportion. Serum sodium
concentration remains NORMAL
4.
Teach patient about edema, ascites, and fluid therapy.
Advise elevation of the extremities, restriction of
fluids, necessity of paracentesis, dialysis and diuretic
therapy.
Instruct patient to avoid over-the-counter medications
without first checking with the health care provider
because they may contain sodium
5.
Pathophysiology of Fluid Volume Excess
Etiologic conditions and Risks factors
a. Congestive heart failure
b. Renal failure
c. Excessive fluid intake
d. Impaired ability to excrete fluid as in renal
disease
e. Cirrhosis of the liver
f. Consumption of excessive table salts
g. Administration of excessive IVF
h. Abnormal fluid retention
PATHOPHYSIOLOGY
Excessive fluid --- expansion of blood volume ----- edema,
increased neck vein distention, tachycardia, hypertension.
The Nursing Process in Fluid Volume Excess
ASSESSMENT
Physical Examination
Increased weight gain
Increased urine output
Moist crackles in the lungs
Increased CVP
Distended neck veins
Wheezing
Dependent edema
ELECTROLYTES
Sources of electrolytes
IMPLEMENTATION
ASSIST IN MEDICAL INTERVENTION
1.
2.
3.
Administer diuretics as prescribed
Assist in hemodialysis
Provide dietary restriction of sodium and water
NURSING MANAGEMENT
1.
2.
3.
Continually assess the patients condition by
measuring intake and output, daily weight monitoring,
edema assessment and breath sounds
Prevent Fluid Volume Excess by adhering to diet
prescription of low salt- foods.
Detect and Control Fluid Volume Excess by closely
monitoring IVF therapy, administering medications,
providing rest periods, placing in semi-fowlers
position for lung expansion and providing frequent
skin care for the edema
MS: Fluids and Electrolyte
Foods and ingested fluids, medications; IVF and
TPN solutions
Functions of Electrolytes
Maintains fluid balance
Regulates acid-base balance
Needed for enzymatic secretion and activation
Needed for proper metabolism and effective
processes of muscular contraction, nerve
transmission
Types of Electrolytes
CATIONS- positively charged ions; examples are
sodium, potassium, calcium
ANIONS- negatively charged ions; examples are
chloride and phosphates]
The major ICF cation is potassium (K+); the
major ICF anion is Phosphates
The major ECF cation is Sodium (Na+); the major
ECF anion is Chloride (Cl-)
ELECTROLYTE IMBALANCES
Subjective cue/s
Shortness of breath
Change in mental state
Laboratory findings
1. BUN and Creatinine levels are LOW because of
dilution
2. Urine sodium and osmolality decreased (urine
becomes diluted)
3. CXR may show pulmonary congestion
Electrolytes are charged ions capable of conducting
electricity and are solutes found in all body
compartments.
SODIUM
The most abundant cation in the ECF
Normal range in the blood is 135-145 mEq/L
A loss or gain of sodium is usually accompanied by a
loss or gain of water.
Major contributor of the plasma Osmolality
Sources: Diet, medications, IVF. The minimum daily
requirement is 2 grams
Functions:
1.
2.
3.
4.
5.
6.
7.
8.
Participates in the Na-K pump
Assists in maintaining blood volume
Assists in nerve transmission and muscle
contraction
Primary determinant of ECF concentration.
Controls water distribution throughout the body.
Primary regulator of ECF volume.
Sodium also functions in the establishment of the
electrochemical state necessary for muscle
contraction and the transmission of nerve
impulses.
Regulations: skin, GIT, GUT, Aldosterone
increases Na retention in the kidney
Abejo
Lecture Notes on Fluids and Electrolytes
Prepared By: Mark Fredderick R Abejo R.N, MAN
SODIUM DEFICIT: HYPONATREMIA
In summary:
Physical Examination
Altered mental status
Vomiting
Lethargy
Muscle twitching and convulsions (if sodium level is
below 115 mEq/L)
Focal weakness
Definition : Refers to a Sodium serum level of less
than 135 mEq/L. This may result from excessive
sodium loss or excessive water gain.
Pathophysiology
Etiologic Factors
a. Fluid loss such as from Vomiting and nasogastric
suctioning
b. Diarrhea
c. Sweating
d. Use of diuretics
e. Fistula
Other factors
a. Dilutional hyponatremia
Water intoxication, compulsive water
drinking where sodium level is diluted
with increased water intake
b. SIADH
Excessive secretion of ADH causing
water retention and dilutional
hyponatremia
PATHOPHYSIOLOGY
Decrease sodium concentration --- hypotonicity of plasma -- water from the intravascular space will move out and go to
the intracellular compartment with a higher concentration --cell swelling --Water is pulled INTO the cell because of
decreased extracellular sodium level and increased
intracellular concentration
The Nursing Process in HYPONATREMIA
Subjective Cues
Nausea
Cramps
Anorexia
Headache
Laboratory findings
1. Serum sodium level is less than 135 mEq/L
2. Decreased serum osmolality
3. Urine specific gravity is LOW if caused by sodium loss
4. In SIADH, urine sodium is high and specific gravity is
HIGH
IMPLEMENTATION
ASSIST IN MEDICAL INTERVENTION
1. Provide sodium replacement as ordered. Isotonic saline
is usually ordered.. Infuse the solution very cautiously.
The serum sodium must NOT be increased by greater
than 12 mEq/L because of the danger of pontine
osmotic demyelination
2. Administer lithium and demeclocycline in SIADH
3. Provide water restriction if with excess volume
NURSING MANAGEMENT
1.
ASSESSMENT
Sodium Deficit (Hyponatremia)
Clinical Manifestations
Clinical manifestations of hyponatremia depend on the
cause, magnitude, and rapidity of onset.
Although nausea and abdominal cramping occur, most
of the symptoms are neuropsychiatric and are probably
related to the cellular swelling and cerebral edema
associated with hyponatremia.
As the extracellular sodium level decreases, the
cellular fluid becomes relatively more concentrated and
pulls water into the cells.
In general, those patients having acute decline in serum
sodium levels have more severe symptoms and higher
mortality rates than do those with more slowly
developing hyponatremia.
Features of hyponatremia associated with sodium loss
and water gain include anorexia, muscle cramps, and a
feeling of exhaustion.
When the serum sodium level drops below 115 mEq/L
(SI: 115 mmol/L), thee ff signs of increasing
intracranial pressure occurs:
lethargy
Confusion
muscular twitching
focal weakness
hemiparesis
papilledema
convulsions
MS: Fluids and Electrolyte
2.
3.
Provide continuous assessment by doing an accurate
intake and output, daily weights, mental status
examination, urinary sodium levels and GI
manifestations. Maintain seizure precaution
Detect and control Hyponatremia by encouraging food
intake with high sodium content, monitoring patients
on lithium therapy, monitoring input of fluids like IVF,
parenteral medication and feedings.
Return the Sodium level to Normal by restricting water
intake if the primary problem is water retention.
Administer sodium to normovolemic patient and
elevate the sodium slowly by using sodium chloride
solution
SODIUM EXCESS: HYPERNATREMIA
Serum Sodium level is higher than 145 mEq/L
There is a gain of sodium in excess of water or a
loss of water in excess of sodium.
Pathophysiology:
Etiologic factors
a. Fluid deprivation
b. Water loss from Watery diarrhea, fever, and
hyperventilation
c. Administration of hypertonic solution
d. Increased insensible water loss
e. Inadequate water replacement, inability to swallow
f. Seawater ingestion or excessive oral ingestion of salts
Abejo
Lecture Notes on Fluids and Electrolytes
Prepared By: Mark Fredderick R Abejo R.N, MAN
Other factors
a. Diabetes insipidus
b. Heat stroke
c. Near drowning in ocean
d. Malfunction of dialysis
IMPLEMENTATION
ASSIST IN THE MEDICAL INTERVENTION
1.
2.
PATHOPHYSIOLOGY
Increased sodium concentration --- hypertonic plasma ---water will move out form the cell outside to the interstitial
space ----- CELLULAR SHRINKAGE ----- then to the
blood ---- Water pulled from cells because of increased
extracellular sodium level and decreased cellular fluid
concentration
The Nursing Process in HYPERNATREMIA
3.
NURSING MANAGEMENT
1.
2.
3.
Sodium Excess (Hypernatremia)
Clinical Manifestations
primarily neurologic
Presumably the consequence of cellular dehydration.
Hypernatremia results in a relatively concentrated ECF,
causing water to be pulled from the cells.
Clinically, these changes may be manifested by:
o restlessness and weakness in moderate
hypernatremia
o disorientation, delusions, and
hallucinations in severe hypernatremia.
Dehydration (hypernatremia) is often overlooked as the
primary reason for behavioral changes in the elderly.
If hypernatremia is severe, permanent brain damage
can occur (especially in children). Brain damage is
apparently due to subarachnoid hemorrhages that result
from brain contraction.
A primary characteristic of hypernatremia is thirst.
Thirst is so strong a defender of serum sodium levels in
normal people that hypernatremia never occurs unless the
person is unconscious or is denied access to water;
unfortunately, ill people may have an impaired thirst
mechanism. Other signs include dry, swollen tongue and
sticky mucous membranes. A mild elevation in body
temperature may occur, but on correction of the
hypernatremia the body temperature should return to
normal.
ASSESSMENT
Physical Examination
Restlessness, elevated body temperature
Disorientation
Dry, swollen tongue and sticky mucous membrane,
tented skin turgor
Flushed skin, postural hypotension
Increased muscle tone and deep reflexes
Peripheral and pulmonary edema
4.
5.
6.
Laboratory findings
1. Serum sodium level exceeds 145 mEq/L
2. Serum osmolality exceeds 295 mOsm/kg
3. Urine specific gravity and osmolality INCREASED
or elevated
MS: Fluids and Electrolyte
Continuously monitor the patient by assessing
abnormal loses of water, noting for the thirst and
elevated body temperature and behavioral changes
Prevent hypernatremia by offering fluids regularly
and plan with the physician alternative routes if oral
route is not possible. Ensure adequate water for
patients with DI. Administer IVF therapy cautiously
Correct the Hypernatremia by monitoring the
patients response to the IVF replacement. Administer
the hypotonic solution very slowly to prevent sudden
cerebral edema.
Monitor serum sodium level.
Reposition client regularly, keep side-rails up, the bed
in low position and the call bell/light within reach.
Provide teaching to avoid over-the counter
medications without consultation as they may contain
sodium
POTASSIUM
The most abundant cation in the ICF
Potassium is the major intracellular electrolyte; in fact,
98% of the bodys potassium is inside the cells.
The remaining 2% is in the ECF; it is this 2% that is
all-important in neuromuscular function.
Potassium is constantly moving in and out of cells
according to the bodys needs, under the influence of
the sodium-potassium pump.
Normal range in the blood is 3.5-5 mEq/L
Normal renal function is necessary for maintenance of
potassium balance, because 80-90% of the potassium is
excreted daily from the body by way of the kidneys.
The other less than 20% is lost through the bowel and
sweat glands.
Major electrolyte maintaining ICF balance
Sources- Diet, vegetables, fruits, IVF, medications
Functions:
1.
2.
3.
4.
5.
Subjective Cues
Delusions and hallucinations
Extreme thirst
Behavioral changes
Administer hypotonic electrolyte solution slowly as
ordered
Administer diuretics as ordered
Loop diuretics (thiazides ok)
Desmopressin is prescribed for diabetes insipidus
6.
Maintains ICF Osmolality
Important for nerve conduction and muscle
contraction
Maintains acid-base balance
Needed for metabolism of carbohydrates, fats and
proteins
Potassium influences both skeletal and cardiac muscle
activity.
( For example, alterations in its concentration change
myocardial irritability and rhythm )
Regulations: renal secretion and excretion,
* Aldosterone promotes renal excretion
* Acidosis promotes K exchange for hydrogen
Abejo
Lecture Notes on Fluids and Electrolytes
Prepared By: Mark Fredderick R Abejo R.N, MAN
POTASSIUM DEFICIT: HYPOKALEMIA
Condition when the serum concentration of potassium
is less than 3.5 mEq/L
Pathophysiology
Etiologic Factors
a. Gastro-intestinal loss of potassium such as
diarrhea and fistula
b. Vomiting and gastric suctioning
c. Metabolic alkalosis
d. Diaphoresis and renal disorders
e. Ileostomy
Other factor/s
a. Hyperaldosteronism
b. Heart failure
c. Nephrotic syndrome
d. Use of potassium-losing diuretics
e. Insulin therapy
f. Starvation
g. Alcoholics and elderly
PATHOPHYSIOLOGY
Decreased potassium in the body impaired nerve
excitation and transmission signs/symptoms such as
weakness, cardiac dysrhythmias etc..
IMPLEMENTATION
ASSIST IN THE MEDICAL INTERVENTION
1.
2.
The Nursing Process in Hypokalemia
Clinical Manifestations
Potassium deficiency can result in widespread
derangements in physiologic functions and especially
nerve conduction.
Most important, severe hypokalemia can result in death
through cardiac or respiratory arrest.
Clinical signs rarely develop before the serum
potassium level has fallen below 3 mEq/L (51: 3
mmol/L) unless the rate of fall has been rapid.
Manifestations of hypokalemia include fatigue,
anorexia, nausea, vomiting, muscle weakness,
decreased bowel motility, paresthesias, dysrhythmias,
and increased sensitivity to digitalis.
If prolonged, hypokalemia can lead to impaired renal
concentrating ability, causing dilute urine, polyuria,
nocturia, and polydipsia
ASSESSMENT
Physical examination
Muscle weakness
Decreased bowel motility and abdominal distention
Paresthesias
Dysrhythmias
Increased sensitivity to digitalis
Subjective cues
Nausea , anorexia and vomiting
Fatigue, muscles cramps
Excessive thirst, if severe
Laboratory findings
1. Serum potassium is less than 3.5 mEq/L
2. ECG: FLAT T waves, or inverted T waves,
depressed ST segment and presence of the U wave
and prolonged PR interval.
3. Metabolic alkalosis
MS: Fluids and Electrolyte
3.
Provide oral or IV replacement of potassium
Infuse parenteral potassium supplement. Always dilute
the K in the IVF solution and administer with a pump.
IVF with potassium should be given no faster than 1020-mEq/ hour!
NEVER administer K by IV bolus or IM
NURSING MANAGEMENT
1.
2.
3.
4.
5.
Continuously monitor the patient by assessing the
cardiac status, ECG monitoring, and digitalis
precaution
Prevent hypokalemia by encouraging the patient to eat
potassium rich foods like orange juice, bananas,
cantaloupe, peaches, potatoes, dates and apricots.
Correct hypokalemia by administering prescribed IV
potassium replacement. The nurse must ensure that the
kidney is functioning properly!
Administer IV potassium no faster than 20 mEq/hour
and hook the patient on a cardiac monitor. To
EMPHASIZE: Potassium should NEVER be given IV
bolus or IM!!
A concentration greater than 60 mEq/L is not advisable
for peripheral veins.
POTASSIUM EXCESS: HYPERKALEMIA
Serum potassium greater than 5.5 mEq/L
Pathophysiology
Etiologic factors
a. Iatrogenic, excessive intake of potassium
b. Renal failure- decreased renal excretion of
potassium
c. Hypoaldosteronism and Addisons disease
d. Improper use of potassium supplements
Other factors
1. Pseudohyperkalemia- tight tourniquet and
hemolysis of blood sample, marked leukocytosis
Abejo
Lecture Notes on Fluids and Electrolytes
Prepared By: Mark Fredderick R Abejo R.N, MAN
2.
3.
4.
Transfusion of old banked blood
Acidosis
Severe tissue trauma
PATHOPHYSIOLOGY
Increased potassium in the body ---- Causing irritability of
the cardiac cells --- Possible arrhythmias!!
The Nursing Process in Hyperkalemia
Clinical Manifestations
By far the most clinically important effect of
hyperkalemia is its effect on the myocardium.
Cardiac effects of an elevated serum potassium level
are usually not significant below a concentration of 7
mEq/L (SI: 7 mmol/L), but they are almost always
present when the level is 8 mEq/L (SI: 8 mmol/L) or
greater.
As the plasma potassium concentration is increased,
disturbances in cardiac conduction occur.
The earliest changes, often occurring at a serum
potassium level greater than 6 mEq/ L (SI: 6
mmol/L), are peaked narrow T waves and a shortened
QT interval.
If the serum potassium level continues to rise, the PR
interval becomes prolonged and is followed by
disappearance of the P waves.
Finally, there is decomposition and prolongation of
the QRS complex. Ventricular dysrhythmias and
cardiac arrest may occur at any point in this
progression.
Note that in Severe hyperkalemia causes muscle
weakness and even paralysis, related to a
depolarization block in muscle.
Similarly, ventricular conduction is slowed.
Although hyperkalemia has marked effects on the
peripheral neuromuscular system, it has little effect on
the central nervous system.
Rapidly ascending muscular weakness leading to
flaccid quadriplegia has been reported in patients with
very high serum potassium levels.
Paralysis of respiratory muscles and those required for
phonation can also occur.
Gastrointestinal manifestations, such as nausea,
intermit tent intestinal colic, and diarrhea, may occur
in hyperkalemic patients.
ASSESSMENT
Physical Examination
Diarrhea
Skeletal muscle weakness
Abnormal cardiac rate
Subjective Cues
Nausea
Intestinal pain/colic
Palpitations
Laboratory Findings
1. Peaked and narrow T waves
2. ST segment depression and shortened QT interval
3. Prolonged PR interval
4. Prolonged QRS complex
5. Disappearance of P wave
6. Serum potassium is higher than 5.5 mEq/L
7. Acidosis
MS: Fluids and Electrolyte
IMPLEMENTATION
ASSIST IN MEDICAL INTERVENTION
1. Monitor the patients cardiac status with cardiac
machine
2. Institute emergency therapy to lower potassium level
by:
a. Administering IV calcium gluconateantagonizes action of K on cardiac conduction
b. Administering Insulin with dextrose-causes
temporary shift of K into cells
c. Administering sodium bicarbonate-alkalinizes
plasma to cause temporary shift
d. Administering Beta-agonists
e. Administering Kayexalate (cation-exchange
resin)-draws K+ into the bowel
NURSING MANAGEMENT
1.
2.
3.
4.
5.
6.
7.
8.
Provide continuous monitoring of cardiac status,
dysrhythmias, and potassium levels.
Assess for signs of muscular weakness, paresthesias,
nausea
Evaluate and verify all HIGH serum K levels
Prevent hyperkalemia by encouraging high risk patient
to adhere to proper potassium restriction
Correct hyperkalemia by administering carefully
prescribed drugs. Nurses must ensure that clients
receiving IVF with potassium must be always
monitored and that the potassium supplement is given
correctly
Assist in hemodialysis if hyperkalemia cannot be
corrected.
Provide client teaching. Advise patients at risk to avoid
eating potassium rich foods, and to use potassium salts
sparingly.
Monitor patients for hypokalemia who are receiving
potassium-sparing diuretic
CALCIUM
Majority of calcium is in the bones and teeth
Small amount may be found in the ECF and ICF
Normal serum range is 8.5 10.5 mg/dL
Sources: milk and milk products; diet; IVF and
medications
Abejo
Lecture Notes on Fluids and Electrolytes
Prepared By: Mark Fredderick R Abejo R.N, MAN
Functions:
1.
2.
3.
4.
5.
Needed for formation of bones and teeth
For muscular contraction and relaxation
For neuronal and cardiac function
For enzymatic activation
For normal blood clotting
HYPOCALCEMIA
Low levels of calcium in the blood
Risk Factors
a. Hypoparathyroidism (idiopathic or postsurgical)
b. Alkalosis (Ca binds to albumin)
c. Corticosteroids (antagonize Vit D)
d. Hyperphosphatemia
e. Vit D deficiency
f. Renal failure (vit D deficiency)
Clinical Manifestation
Decreased cardiac contractility
Arrhythmia
ECG: prolonged QT interval, lengthened ST
segment
Trousseaus sign (inflate BP cuff 20mm above
systole for 3 min = carpopedal spasm)
Regulations:
1.
2.
3.
GIT- absorbs Ca+ in the intestine; Vitamin D helps to
increase absorption
Renal regulation- Ca+ is filtered in the glomerulus
and reabsorbed in the tubules:
Endocrine regulation:
Parathyroid hormone from the parathyroid glands
is released when Ca+ level is low. PTH causes
release of calcium from bones and increased
retention of calcium by the kidney but PO4 is
excreted
Calcitonin from the thyroid gland is released when
the calcium level is high. This causes excretion of
both calcium and PO4 in the kidney and promoted
deposition of calcium in the bones.
Chvosteks sign (tap facial nerve anterior to the
ear = ipsilateral muscle twitching)
Tetany
Hyperreflexia, seizures
Laryngeal spasms/stridor
Diarrhea, hyperactive bowel sounds
Bleeding
Collaborative Management
1. Calcium gluconate 10% IV
2. Calcium chloride 10% IV
3. both usually given by Dr, very slowly; venous irritant;
cardiac probs
4. Oral: calcium citrate, lactate, carbonate; Vit D
supplements
5. Diet: high calcium
6. Watch out for tetany, seizures, laryngospasm, resp &
cardiac arrest
7. Seizure precautions
Sources:
milk, yogurt, cheese, sardines, broccoli, tofu, green leafy
vegetables
MS: Fluids and Electrolyte
Abejo
Lecture Notes on Fluids and Electrolytes
Prepared By: Mark Fredderick R Abejo R.N, MAN
HYPERCALCEMIA
HYPOMAGNESEMIA
is an elevated calcium level in the blood
usually from bone resorption
is an electrolyte disturbance in which there is an
abnormally low level of magnesium in the blood.
Risk Factors / Causes
a. Hyperparathyroidism (eg adenoma)
b. Metastatic cancer (bone resorption as tumors
ectopic PTH effect) eg. Multiple myeloma
c. Thiazide diuretics (potentiate PTH effect)
d. Immobility
e. Milk-alkali syndrome (too much milk or antacids
in aegs with peptic ulcer)
Clinical Manifestation
groans (constipation)
moans (psychotic noise)
bones (bone pain, especially if PTH is elevated)
stones (kidney stones)
psychiatric overtones (including depression and
confusion)
Risk Factors and Cause
a. Chronic alcoholism (most common), Alcohol
stimulates renal excretion of magnesium,
b. Inflammatory bowel disease
c. Small bowel resection
d. GI cancer
e. chronic pancreatitis (poor absorption)
f. Loop and thiazide diuretic use (the most common
cause of hypomagnesemia)
g. Antibiotics (i.e. aminoglycoside, amphotericin,
pentamidine, gentamicin, tobramycin, viomycin)
block resorption in the loop of Henle.
h. Excess calcium
i. Excess saturated fats
j. Excess coffee or tea intake
k. Excess phosphoric or carbonic acids (soda pop)
l. Insufficient water consumption
m. Excess salt or sugar intake
n. Insufficient selenium,vitamin D, sunlight
exposure or vitamin B6
o. Increased levels of stress
Clinical Manifestation
Weakness
muscle cramps
cardiac arrhythmia
increased irritability of the nervous system with
tremors, athetosis, jerking, nystagmus and an
extensor plantar reflex. Confusion
disorientation
hallucinations
depression
epileptic fits
hypertension, tachycardia and tetany.
Arrhythmia
ECG: shortened QT interval, decreased ST
segment
Hyporeflexia, lethargy, coma
Collaborative Management
1. If parathyroid tumor = surgery
2. Diet: low Ca, stop taking Ca Carbonate antacids,
increase fluids
3. IV flushing (usually NaCl)
4. Loop diuretics
5. Corticosteroids
6. Biphosphonates, like etidronate (Calcitonin) &
alendronate (Fosamax)
7. Plicamycin (Mithracin) inhibits bone resorption
8. Calcitonin IM or intranasal
9. Dialysis (severe case)
10. Watch out for digitalis toxicity
11. Prevent fractures, handle gently
MAGNESIUM
2nd most abundant intracellular cation
50% found in bone, 45% is intracellular
ATP (adenosine triphosphate), the main source of
energy in cells, must be bound to a magnesium ion in
order to be biologically active.
competes with Ca & P absorption in the GI
inhibits PTH
Normal value : 1.5-2.5 mEq/L
Functions:
1. important in maintaining intracellular activity
2. affects muscle contraction, & especially relaxation
3. maintains normal heart rhythm
4. promotes vasodilation of peripheral arterioles
Sources:
green leafy vegetables, nuts, legumes, seafood, whole
grains, bananas, oranges, cocoa, chocolate
MS: Fluids and Electrolyte
* Like hypocalcemia, hypokalemia
Potentiates digitalis toxicity
Collaborative Management
1. Magnesium sulfate IV, IM (make sure renal
function is ok) may cause flushing
2. Oral: Magnesium oxide 300mg/day,
3. Mg-containing antacids (SE diarrhea)
4. Diet: high magnesium (fruits,green vegetables,
whole grains cereals, milk, meat, nuts and sea
foods )
5. Promotion of safety, protect from injury
HYPERMAGNESEMIA
Etiologic Factors
a. Magnesium treatment for pre-eclampsia
b. Renal failure
c. Diabetic Ketoacidosis
d. Excessive use of Mg antacids/laxatives
PATHOPHYSIOLOGY
Increase Mg. ----- Blocks acetylcholine release ---- decrease
excitability of muscle
Abejo
Lecture Notes on Fluids and Electrolytes
Prepared By: Mark Fredderick R Abejo R.N, MAN
Clinical Manifestation
Hyporeflexia
Hypotension, bradycardia, arrhythmia
Flushing
Weakness, lethargy, coma
Decreased RR & respiratory paralysis
Loss of DTRs
HYPERPHOSPHATEMIA
Risk Factors
a. Acidosis (Ph moves out of cell)
b. Cytotoxic agents/chemotherapy in cancer
c. Renal failure
d. Hypocalcemia
e. Massive BT (P leaks out of cells during storage of
blood)
f. Hyperthyroidism
Clinical Manifestation
Calcification of kidney, cornea, heart
Muscle spasms, tetany, hyperreflexia
*like hypercalcemia
Collaborative Management
1. Diuretics
2. Stop Mg-containing antacids & enemas
3. IV fluids rehydration
4. Calcium gluconate (antidote, antagonizes
cardiac & respiratory effects of Mg)
5. Dialysis if RF
PHOSPHORUS
primary intracellular anion
part of ATP energy
85% bound with Ca in teeth/bones, skeletal muscle
reciprocal balance with Ca
absorption affected by Vit D, regulation affected by
PTH (lowers P level)
Normal value : 2.5-4.5 mg/dL
Functions:
1. bone/teeth formation & strength
2. phospholipids (make up cell membrane integrity)
3. part of ATP
4. affects metabolism, Ca levels
Sources:
red & organ meats (brain, liver, kidney), poultry, fish, eggs,
milk, legumes, whole grains, nuts, carbonated drinks
HYPOPHOSPHATEMIA
Risk Factors
a. Decreased Vit D absorption, sunlight exposure
b. Hyperparathyroidism (increased PTH)
c. Aluminum & Mg-containing antacids (bind P)
d. Severe vomiting & diarrhea
Clinical Manifestation
Anemia, bruising (weak blood cell membrane)
Seizures, coma
Muscle weakness, paresthesias
Constipation, hypoactive bowel sounds
*like hypocalcemia
Collaborative ManagementM
1. Aluminum antacids as phosphate binders: Al
carbonate (Basaljel), Al hydroxide (Amphojel)
2. Ca carbonate for hypocalcemia
3. Avoid phosphate laxatives/enemas
4. Increase fluid intake
5. Diet: low Phos, no carbonated drinks
CHLORIDE
Functions:
1. helps regulate BP, serum osmolarity
2. part of HCl
3. acid/base balance (exchanges with HCO3)
Sources:
salt, canned food, cheese, milk, eggs, crab, olives
HYPOCHLOREMIA
Risk Factors
a. Diuresis
b. Metabolic alkalosis
c. Hyponatremia, prolonged D5W IV
d. Addisons
Clinical Manifestation
Slow, shallow respirations (met. Alkalosis)
Hypotension (Na & water loss)
*Like hypercalcemia
Collaborative Management
1. Sodium phosphate or potassium phosphate IV
(give slowly, no faster than 10 mEq/hr)
2. Sodium & potassium phosphate orally (NeutraPhos, K-Phos) give with meals to prevent gastric
irritation
3. Avoid Phos-binding antacids
4. Diet: high Mg, milk
5. Monitor joint stiffness, arthralgia, fractures,
bleeding
MS: Fluids and Electrolyte
extracellular anion, part of salt
binds with Na, H (also K, Ca, etc)
exchanges with HCO3 in the kidneys (& in RBCs)
Normal value: 95 -108 mEq/L
Collaborative Management
1. Administer IV or Oral : KCl, NaCl
2. Diet: high Cl (& usually Na)
HYPERCHLOREMIA
Risk Factors / Cause
a. Metabolic acidosis
b. Usually noted in hyperNa, hyperK
Abejo
Lecture Notes on Fluids and Electrolytes
Prepared By: Mark Fredderick R Abejo R.N, MAN
Clinical Manifestation
Deep, rapid respirations (met. Acidosis)
hyperK, hyperNa S/S
Increased Cl sweat levels in cystic fibrosis
Collaborative Management
1. Diuretics
2. Hypotonic solutions, D5W to restore balance
3. Diet: low Cl (& usually Na)
4. Treat acidosis
Acid-Base Balance Mechanisms
Buffer - prevents major changes in ECF by releasing or
accepting H ions
Buffer mechanism: first line (takes seconds)
1. combine with very strong acids or bases to
convert them into weaker acids or bases
2. Bicarbonate Buffer System
- most important
- uses HCO3 & carbonic acid/H2CO3 - (20:1)
- closely linked with respiratory & renal
mechanisms
3. Phosphate Buffer System
- more important in intracellular fluids, where
concentration is higher
- similar to bicarbonate buffer system, only uses
phosphate
4. Protein Buffer System
- hemoglobin, a protein buffer, promotes
movement of chloride across RBC membrane in
exchange for HCO3
Respiratory mechanism: 2nd line (takes minutes)
1. increased respirations liberates more CO2 =
increase pH
2. decreased respirations conserve more CO2 =
decrease pH
carbonic acid (H2CO3) = CO2 + water
Renal mechanism: 3rd line (takes hours-days)
1. kidneys secrete H ions & reabsorb bicarbonate ions =
increase blood pH
2. kidneys form ammonia that combines with H ions to
form ammonium ions, which are excreted in the urine
in exchange for sodium ions
Review: Acid-Base Imbalance
pH 7.35-7.45
pCO2 measurement of the CO2 pressure that is being
exerted on the plasma
35-45mmHg
PaO2- amount of pressure exerted by O2 on the plasma
80-100mmHg
SaO2- percent of hemoglobin saturated with O2
Base excess amount of HCO3 available in the ECF
-3 to +3
MS: Fluids and Electrolyte
Interpretation Arterial Blood Gases
Step 1
Step 2
Step 3
If acidosis the pH is down
If alkalosis the pH is up
The respiratory function indicator is the PCO2
The metabolic function indicator is the HCO3
Look at the pH
Is it up or down?
If it is up - it reflects alkalosis
If it is down - it reflects acidosis
Look at the PCO2
Is it up or down?
If it reflects an opposite response as the pH,
then you know that the condition is a
respiratory imbalance
If it does not reflect an opposite response as the
pH - move to step III
Look at the HCO3
Does the HCO3 reflect a corresponding
response with the pH
If it does then the condition is a metabolic
imbalance
FACTORS AFFECTING BODY FLUIDS,
ELECTROLYTES AND ACID-BASE BALANCE
AGE
Infants have higher proportion of body water than
adults
Water content of the body decreases with age
Infants have higher fluid turn-over due to immature
kidney and rapid respiratory rate
GENDER AND BODY SIZE
Women have higher body fat content but lesser
water content
Lean body has higher water content
ENVIRONMENT AND TEMPERATURE
Climate and heat and humidity affect fluid balance
DIET AND LIFESTYLE
Anorexia nervosa will lead to nutritional depletion
Stressful situations will increase metabolism,
increase ADH causing water retention and
increased blood volume
Chronic Alcohol consumption causes malnutrition
ILLNESS
Trauma and burns release K+ in the blood
Cardiac dysfunction will lead to edema and
congestion
MEDICAL TREATMENT, MEDICATIONS AND
SURGERY
Suctioning, diuretics and laxatives may cause
imbalances
Abejo
Lecture Notes on Fluids and Electrolytes
Prepared By: Mark Fredderick R Abejo R.N, MAN
ACID-BASE BALANCE PROBLEMS
RESPIRATORY ACIDOSIS
pH < 7.35
pCO2 > 45 mm Hg (excess carbon dioxide in the
blood)
Respiratory system impaired and retaining CO2;
causing acidosis
Common Stimuli
a. Acute respiratory failure from airway obstruction
b. Over-sedation from anesthesia or narcotics
c. Some neuromuscular diseases that affect ability to
use chest muscles
d. Chronic respiratory problems, such as Chronic
Obstructive Lung Disease
Signs and Symptoms
Compensation: kidneys respond by generating and
reabsorbing bicarbonate ions, so HCO3 >26 mm Hg
Respiratory: hypoventilation, slow or shallow
respirations
Neuro: headache, blurred vision, irritability,
confusion
Respiratory collapse leads to unconsciousness and
cardiovascular collapse
Collaborative Management
1. Early recognition of respiratory status and treat
cause
2. Restore ventilation and gas exchange; CPR for
respiratory failure with oxygen supplementation;
intubation and ventilator support if indicated
3. Treatment of respiratory infections with
bronchodilators, antibiotic therapy
4. Reverse excess anesthetics and narcotics with
medications such as naloxone (Narcan)
5. Chronic respiratory conditions
Breathe in response to low oxygen levels
Adjusted to high carbon dioxide level
through metabolic compensation (therefore,
high CO2 not a breathing trigger)
Cannot receive high levels of oxygen, or will
have no trigger to breathe; will develop
carbon dioxide narcosis
Treat with no higher than 2 liters O2 per
cannula
6. Continue respiratory assessments, monitor further
arterial blood gas results
RESPIRATORY ALKALOSIS
pH < 7.35
pCO2 < 35 mm Hg.
Carbon dioxide deficit, secondary to
hyperventilation
Common Stimuli
a. Hyperventilation with anxiety from uncontrolled
fear, pain, stress (e.g. women in labor, trauma
victims)
b. High fever
c. Mechanical ventilation, during anesthesia
MS: Fluids and Electrolyte
Signs and Symptoms
Compensation: kidneys compensate by
eliminating bicarbonate ions; decrease in
bicarbonate HCO3 < 22 mm Hg.
Respiratory: hyperventilating: shallow, rapid
breathing
Neuro: panicked, light-headed, tremors, may
develop tetany, numb hands and feet (related to
symptoms of hypocalcemia; with elevated pH
more Ca ions are bound to serum albumin and less
ionized active calcium available for nerve and
muscle conduction)
May progress to seizures, loss of consciousness
(when normal breathing pattern returns)
Cardiac: palpitations, sensation of chest tightness
Collaborative Management
1.
2.
3.
4.
Treatment: encourage client to breathe slowly in a
paper bag to rebreathe CO2
Breathe with the patient; provide emotional
support and reassurance, anti-anxiety agents,
sedation
On ventilator, adjustment of ventilation settings
(decrease rate and tidal volume)
Prevention: pre-procedure teaching, preventative
emotional support, monitor blood gases as
indicated
METABOLIC ACIDOSIS
pH <7.35
Deficit of bicarbonate in the blood NaHCO3 <22
mEq/L
Caused by an excess of acid, or loss of
bicarbonate from the body
Common Stimuli
a. Acute lactic acidosis from tissue hypoxia (lactic
acid produced from anaerobic metabolism with
shock, cardiac arrest)
b. Ketoacidosis (fatty acids are released and
converted to ketones when fat is used to supply
glucose needs as in uncontrolled Type 1 diabetes
or starvation)
c. Acute or chronic renal failure (kidneys unable to
regulate electrolytes)
d. Excessive bicarbonate loss (severe diarrhea,
intestinal suction, bowel fistulas)
e. Usually results from some other disease and is
often accompanied by electrolyte and fluid
imbalances
f. Hyperkalemia often occurs as the hydrogen ions
enter cells to lower the pH displacing the
intracellular potassium; hypercalcemia and
hypomagnesemia may occur
Signs and Symptoms
Compensation: respiratory system begins to
compensate by increasing the depth and rate of
respiration in an effort to lower the CO2 in the blood;
this causes a decreased level of carbon dioxide: pCO2
<35 mm HG.
Neuro changes: headache, weakness, fatigue
progressing to confusion, stupor, and coma
Cardiac: dysrhythmias and possibly cardiac arrest from
hyperkalemia
GI: anorexia, nausea, vomiting
Skin: warm and flushed
Abejo
Lecture Notes on Fluids and Electrolytes
Prepared By: Mark Fredderick R Abejo R.N, MAN
Respiratory: tries to compensate by hyperventilation:
deep and rapid respirations known as Kussmauls
respirations
Diagnostic test findings:
1. ABG: pH < 7.35, HCO3 < 22
2. Electrolytes: Serum K+ >5.0 mEq/L
3. Serum Ca+2 > 10.0 mg/dL
4. Serum Mg+2 < 1.6 mg/dL
Collaborative Management
1. Medications: Correcting underlying cause will
often improve acidosis
2. Restore fluid balance, prevent dehydration with
IV fluids
3. Correct electrolyte imbalances
4. Administer Sodium Bicarbonate IV, if acidosis is
severe and does not respond rapidly enough to
treatment of primary cause. (Oral bicarbonate is
sometimes given to clients with chronic metabolic
acidosis) Be careful not to overtreat and put client
into alkalosis
5. As acidosis improves, hydrogen ions shift out of
cells and potassium moves intracellularly.
Hyperkalemia may become hypokalemia and
potassium replacement will be needed.
6. Assessment
Vital signs
Intake and output
Neuro, GI, and respiratory status;
Cardiac monitoring
Reassess repeated arterial blood gases and
electrolytes
METABOLIC ALKALOSIS
pH >7.45
HCO3 > 26 mEq/L
Caused by a bicarbonate excess, due to loss of
acid, or a bicarbonate excess in the body
1.
2.
3.
4.
Selected Water and Electrolyte
Solutions
Isotonic Solutions
A. 0.9% NaCl (isotonic, also called NSS)
Na+ 154 mEq/L
Cl- 154 mEq/L
(308 mOsm/L)
Also available with varying concentrations of dextrose (the
most frequent used is a 5% dextrose concentration
Common Stimuli
a. Loss of hydrogen and chloride ions through
excessive vomiting, gastric suctioning, or
excessive diuretic therapy Response to
hypokalemia
b. Excess ingestion of bicarbonate rich antacids or
excessive treatment of acidosis with Sodium
Bicarbonate
Signs and Symptoms
Compensation: Lungs respond by decreasing the
depth and rate of respiration in effort to retain carbon
dioxide and lower pH
Neuro: altered mental status, numbness and tingling
around mouth, fingers, toes, dizziness, muscle spasms
(similar to hypocalcemia due to less ionized calcium
levels)
Respiratory: shallow, slow breathing
Diagnostic test findings
1. ABGs: pH> 7.45, HCO3 >26
2. Electrolytes: Serum K+ < 3.5 mEq/L
3. Electrocardiogram: as with hypokalemia
MS: Fluids and Electrolyte
An isotonic solution that expands the ECF
volume, used in hypovolemic states, resuscitative
efforts, shock, diabetic ketoacidosis, metabolic
alkalosis, hypercalcemia, mild Na deficit
Supplies an excess of Na and Cl; can cause fluid
volume excess and hyperchloremic acidosis if
used in excessive volumes, particularly in patients
with compromised renal function, heart failure or
edema
Not desirable as a routine maintenance solution,
as it provides only Na and Cl (and these are
provided in excessive amounts)
When mixed with 5% dextrose, the resulting
solution becomes hypertonic in relation to plasma,
and in addition to the above described
electrolytes, provides 170cal/L
Only solution that may be administered with
blood products
B. Lactated Ringers solution (Hartmanns solution)
Na+ 130 mEq/L
K+ 4 mEq/L
Ca++ 3 mEq/L
Cl- 109 mEq/L
Lactate (metabolized to bicarbonate) 28 mEq/L (274
mOsm/L)
Also available with varying concentration of dextrose (the
most common is 5% dextrose)
Collaborative Management
Correcting underlying cause will often improve
alkalosis
Restore fluid volume and correct electrolyte
imbalances (usually IV NaCl with KCL).
With severe cases, acidifying solution may be
administered.
Assessment
Vital signs
Neuro, cardiac, respiratory assessment
Repeat arterial blood gases and electrolytes
An isotonic solution that contains multiple
electrolytes in roughly the same concentration as
found in plasma (note that solution is lacking in
Mg++) provides 9 cal/L
Used in the tx of hypovolemia, burns, fluid lost as
bile or diarrhea, and for acute blood loss
replacement
Lactate is rapidly metabolized into HCO3- in the
body. Lactated Ringers solution should not be
used in lactic acidosis because the ability to
convert lactate into HCO3- is impaired in this
disorder.
Abejo
Lecture Notes on Fluids and Electrolytes
Prepared By: Mark Fredderick R Abejo R.N, MAN
Not to be given with a pH > 7.5 because
bicarbonates is formed as lactate breaks down
causing alkalosis
Should not be used in renal failure because it
contains potassium and can cause hyperkalemia
Similar to plasma
C. 5% Dextrose in Water (D5W)
No electrolytes
50 g of glucose
An isotonic solution that supplies 170 cal/L and
free water to aid in renal excretion of solutes
Used in treatment of hypernatremia, fluid loss and
dehydration
Should not be used in excessive volumes in the
early post-op period (when ADH secretion is
increased due to stress reaction)
Should not be used solely in tx of fluid volume
deficit, because it dilutes plasma electrolyte
concentrations
Contraindicated in head injury because it may
cause increased intracranial pressure
Should not be used for fluid resuscitation because
it can cause hyperglycemia
Should be used with caution in patients with renal
or cardiac dse because of risk of fluid overload
Electrolyte-free solutions may cause peripheral
circulatory collapse, anuria in pt. with sodium
deficiency and increased body fluid loss
Converts to hypotonic solution as dextrose is
metabolized by body. Overtime D5W without
NaCl can cause water intoxication (ICF vol.
excess bec. solution is hypotonic)
Hypotonic Solutions
Hypertonic Solutions
E. 3% NaCl (hypertonic saline)
Na+ 513 mEq/L
Cl- 513 mEq/L
(1026 mOsm/L)
F. 5% NaCl (hypertonic solution)
Na+ 855 mEq/L
Cl- 855 mEq/L
(1710 mOsm/L)
Provides Na, Cl and free water
Free water is desirable to aid the kidneys in
elimination of solute
Lacking in electrolytes other than Na and Cl
When mixed with 5% dextrose, the solution
becomes slightly hypertonic to plasma and in
addition to the above-described electrolytes
provides 170 cal/L
Used in the tx of hypertonic dehydration, Na and
Cl depletion and gastric fluid loss
Not indicated for third-space fluid shifts or
increased intracranial pressure
Administer cautiously, because it can cause fluid
shifts from vascular system into cells, resulting in
cardiovascular collapse and increased intracranial
pressure
MS: Fluids and Electrolyte
Highly hypertonic solution used to treat
symptomatic hyponatremia
Administered slowly and cautiously, because it
can cause intravascular volume overload and
pulmonary edema
Supplies no calories
Colloid Solutions
G. Dextran in NS or 5% D5W
Available in low-molecular-weight (Dextran 40) and highmolecular-weight (Dextran 70) forms
D. 0.45% NaCl
half-strength saline)
Na+ 77 mEq/L
Cl- 77 mEq/L
(154 mOsm/L)
Also available with varying concentration of dextrose (the
most common is 5% dextrose)
Used to increase ECF volume, decrease cellular
swelling
Highly hypertonic solution used only in critical
situations to treat hyponatremia
Must be administered slowly and cautiously,
because it can cause intravascular volume
overload and pulmonary edema
Supplies no calories
Assists in removing ICF excess
Colloid solution used as volume/plasma expander
for intravascular part of ECF
Affects clotting by coating platelets and
decreasing ability to clot
Remains in circulatory system up to 24 hours
Used to treat hypovolemia in early shock to
increase pulse pressure, CO, and arterial BP
Improves microcirculation by decreasing RBC
aggregation
Contraindicated in hemorrhage,
thrombocytopenia, renal dse and severe
dehydration
Not a substitute for blood or blood products
Abejo
You might also like
- A Simple Guide to Hypovolemia, Diagnosis, Treatment and Related ConditionsFrom EverandA Simple Guide to Hypovolemia, Diagnosis, Treatment and Related ConditionsNo ratings yet
- Medicationpart1 110202192115 Phpapp02Document10 pagesMedicationpart1 110202192115 Phpapp02Jessamine Rochelle Reyes Esberto100% (1)
- Medical Surgical NursingDocument24 pagesMedical Surgical NursingWilmaBongotanPadawilNo ratings yet
- NCLEX Exam Cardiovascular Surgery CareDocument5 pagesNCLEX Exam Cardiovascular Surgery CareHeather ClemonsNo ratings yet
- Fluids and Electrolytes - RATIODocument16 pagesFluids and Electrolytes - RATIOLouie Bello100% (3)
- Cardiovascular Med Surg Memory Notebook of NursingDocument8 pagesCardiovascular Med Surg Memory Notebook of NursingdmsapostolNo ratings yet
- Acute Renal Failure-Student-Rapid ReasoningDocument4 pagesAcute Renal Failure-Student-Rapid Reasoningapi-268403950No ratings yet
- Review Questions Fluid and ElectrolytesDocument2 pagesReview Questions Fluid and Electrolytesmarie100% (13)
- Care of Adults 25 Cardiovascular ManagementDocument39 pagesCare of Adults 25 Cardiovascular ManagementGaras AnnaBerniceNo ratings yet
- Med-Surg LEWIS 47 Final Foofed UpDocument10 pagesMed-Surg LEWIS 47 Final Foofed Uphariniliaankhen100% (1)
- Med-Surg Nusing BulletsDocument65 pagesMed-Surg Nusing BulletsHarley C. Tan100% (1)
- Pharmacology Questions CardiacDocument5 pagesPharmacology Questions CardiacAmanda SimpsonNo ratings yet
- NCLEX StrokeSeizureDocument9 pagesNCLEX StrokeSeizureArthur Christopher CorpuzNo ratings yet
- GI NursingDocument14 pagesGI NursingKarl Bayonito100% (5)
- Handout Medical-Surgical Nursing Fluid and ElectrolyteDocument13 pagesHandout Medical-Surgical Nursing Fluid and ElectrolytePaul Christian P. Santos, RN100% (10)
- Notes-Maternal Health NursingDocument22 pagesNotes-Maternal Health NursingRachael Crossgrove100% (4)
- Hematologic DiseasesDocument11 pagesHematologic DiseasesPerrilyn Perey100% (2)
- Chapter 14 Fluid and Electrolytes ChartsDocument7 pagesChapter 14 Fluid and Electrolytes ChartsBNA_RN100% (3)
- Medical Surgical Nursing With MnemonicsDocument111 pagesMedical Surgical Nursing With MnemonicsChernobyle Tolentino BattadNo ratings yet
- Med SurgDocument98 pagesMed SurgKimsha Concepcion88% (8)
- Med Surg Study GuideDocument24 pagesMed Surg Study Guidekylie100% (4)
- COMPREHENSIVE NURSING ACHIEVEMENT TEST (RN): Passbooks Study GuideFrom EverandCOMPREHENSIVE NURSING ACHIEVEMENT TEST (RN): Passbooks Study GuideNo ratings yet
- Janeth Cardiovascular ExamDocument10 pagesJaneth Cardiovascular ExammatthewandmaverickjeNo ratings yet
- F&E ExamDocument3 pagesF&E ExamDino PringNo ratings yet
- MEDICAL - SURGICAL by John RicafortDocument9 pagesMEDICAL - SURGICAL by John RicafortTazneem Apostol Esmael100% (2)
- Fluids and Electrolytes HandoutsDocument15 pagesFluids and Electrolytes HandoutsSiv Carlaisle100% (3)
- Mabes Fluid and Electrolyte ImbalancesDocument15 pagesMabes Fluid and Electrolyte ImbalancesMabesNo ratings yet
- Medical Surgical Nursing Pre-Test 2 RATIONALEDocument8 pagesMedical Surgical Nursing Pre-Test 2 RATIONALEBlaine ManiegoNo ratings yet
- Endocrine Disorders (Study Notes)Document11 pagesEndocrine Disorders (Study Notes)Danelle Harrison, RN90% (10)
- Fluids and Electrolytes ExamDocument3 pagesFluids and Electrolytes Exammonmon100% (3)
- Nursing Skills: Blood Transfusion/ Iv TherapyDocument6 pagesNursing Skills: Blood Transfusion/ Iv TherapyVince John SevillaNo ratings yet
- Fluid and ElectrolytesDocument13 pagesFluid and ElectrolytesHenry Philip93% (15)
- Nursing Quiz AnticoagulantsDocument3 pagesNursing Quiz AnticoagulantsChieChay Dub100% (4)
- Med Surg Exam 1 Test BankDocument18 pagesMed Surg Exam 1 Test BankBrian Bilecky100% (2)
- NCLEX - Acid Base BalanceDocument1 pageNCLEX - Acid Base BalanceNathalee WalkerNo ratings yet
- Medical Surgical QandADocument63 pagesMedical Surgical QandAAlona Lumidao100% (8)
- Physiologic Changes of Aging: System AlterationDocument1 pagePhysiologic Changes of Aging: System Alterationshenric16100% (1)
- Nursing Pharmacology HandoutsDocument9 pagesNursing Pharmacology HandoutsVince LeonidaNo ratings yet
- CARDIODocument17 pagesCARDIORayana Ubas100% (1)
- NCM 118Document8 pagesNCM 118Zanie CruzNo ratings yet
- Fluid & ElectrolytesDocument6 pagesFluid & ElectrolyteskauragiousNo ratings yet
- Medical and Surgical NursingDocument4 pagesMedical and Surgical NursingCrystal Ann Monsale TadiamonNo ratings yet
- Fluid ElectrolytesDocument6 pagesFluid ElectrolytesKareem Mae Pines RaborNo ratings yet
- Fluid and ElectrolytesDocument13 pagesFluid and ElectrolytesMarcus, RN100% (7)
- Medical Surgical NursingDocument59 pagesMedical Surgical NursingAhmari Zamora Julkarnain100% (8)
- Exam 4 MedsurgDocument9 pagesExam 4 MedsurgTori RolandNo ratings yet
- Congenital Heart DiseasesDocument6 pagesCongenital Heart DiseasesMox SwanNo ratings yet
- Precautions NclexDocument1 pagePrecautions NclexAngelo Karzovic100% (1)
- Compressive Ati PDFDocument9 pagesCompressive Ati PDFsarahjaimeeNo ratings yet
- PerfusionDocument9 pagesPerfusionAmanda Brittain100% (2)
- DI - Diabetes InsipidusDocument1 pageDI - Diabetes InsipidusTori Roland100% (1)
- Med SurgDocument179 pagesMed Surgvinwaleed100% (1)
- IV Solution Cheat Sheet: Type Description Osmolality Use MiscellaneousDocument1 pageIV Solution Cheat Sheet: Type Description Osmolality Use Miscellaneousivy_espesoNo ratings yet
- Mnemonics For NursesDocument52 pagesMnemonics For Nursessweetpearl27100% (3)
- NCLEX Fluids & ElectrolytesDocument6 pagesNCLEX Fluids & ElectrolytesNathalee Walker100% (1)
- Fluids & Electrolytes: A. 2 Body CompartmentsDocument5 pagesFluids & Electrolytes: A. 2 Body CompartmentsJULIUS ART VINCENT A. PADINITNo ratings yet
- Basic of Fluid Therapy ImaDocument69 pagesBasic of Fluid Therapy Imal Made ArtawanNo ratings yet
- Rudaina Alelyani MEV Assignment3.Document15 pagesRudaina Alelyani MEV Assignment3.Rudaina AlelyaniNo ratings yet
- Kantha Embroidery by Nanduri Asha, Shreya Gulati, Saloni PriyaDocument17 pagesKantha Embroidery by Nanduri Asha, Shreya Gulati, Saloni PriyaShreyaa Gulati100% (3)
- Bhsa Inggris'Document5 pagesBhsa Inggris'Dwi NovianaNo ratings yet
- Faculty Profile: Dr. Injam Siva ParvathiDocument9 pagesFaculty Profile: Dr. Injam Siva ParvathiMr. Y. RajeshNo ratings yet
- DEMU COUP - Leviat - 17 1 EDocument28 pagesDEMU COUP - Leviat - 17 1 EJianhua WuNo ratings yet
- Poster - Combur10 Test Parameters PDFDocument1 pagePoster - Combur10 Test Parameters PDFAde FeriyatnaNo ratings yet
- Accomplishment Report: Gass/Mfo Success Indicators Actual Accomplishments RemarksDocument1 pageAccomplishment Report: Gass/Mfo Success Indicators Actual Accomplishments RemarksGlenn HernandezNo ratings yet
- Fisher N551 ESVDocument2 pagesFisher N551 ESVWynn ChoNo ratings yet
- Fortified Rice FssaiDocument8 pagesFortified Rice FssaisaikumarNo ratings yet
- MHR PreCal 12 Textbook CH 1 SolutionsDocument57 pagesMHR PreCal 12 Textbook CH 1 SolutionsBryan LowNo ratings yet
- Photovoltaic Water Heater: The Ecological Revolution MADE IN ITALYDocument4 pagesPhotovoltaic Water Heater: The Ecological Revolution MADE IN ITALYDani Good VibeNo ratings yet
- Homework 3rd SteelDocument4 pagesHomework 3rd SteelPiseth HengNo ratings yet
- Animal Instinct (Em, Original) Acordes GuitarraDocument2 pagesAnimal Instinct (Em, Original) Acordes GuitarraGustavoNo ratings yet
- Carbohydrate-Related Diseases Term PaperDocument5 pagesCarbohydrate-Related Diseases Term Paperheiress comiaNo ratings yet
- Table of Content and PrefaceDocument5 pagesTable of Content and PrefaceHaiderEbrahimNo ratings yet
- Astm D6321-98-2004Document3 pagesAstm D6321-98-2004Thyagu LingamurthyNo ratings yet
- Plugs, Fuses and Household ElectricityDocument4 pagesPlugs, Fuses and Household ElectricityRonald HuynhNo ratings yet
- ISO 13920 - Tolerâncias para Juntas SoldadasDocument7 pagesISO 13920 - Tolerâncias para Juntas SoldadasRicardo RicardoNo ratings yet
- High Risk Neonatal Nursing CareDocument40 pagesHigh Risk Neonatal Nursing Carecarol_cezar100% (2)
- B.ing Wajib - XI IPA1-2Document3 pagesB.ing Wajib - XI IPA1-2iwan fals NurjaniNo ratings yet
- 09.3090 USTR2433b T Series Cassettes Omega IFU enDocument51 pages09.3090 USTR2433b T Series Cassettes Omega IFU enAdi SaputraNo ratings yet
- DH-IPC-HDBW1231E: 2MP WDR IR Mini-Dome Network CameraDocument3 pagesDH-IPC-HDBW1231E: 2MP WDR IR Mini-Dome Network CameraDeltaz AZNo ratings yet
- SRS Cheat CodesDocument9 pagesSRS Cheat CodesnurhayatiNo ratings yet
- Solution Manual For Laboratory Manual in Physical Geology 11th by American Geological InstituteDocument9 pagesSolution Manual For Laboratory Manual in Physical Geology 11th by American Geological InstituteKarenAcevedotkoi100% (40)
- Opti-Cal TPS1200 Terminal Mode QuickguideDocument4 pagesOpti-Cal TPS1200 Terminal Mode QuickguideClaudiu OvidiuNo ratings yet
- Lover Album LyricsDocument34 pagesLover Album LyricsMichael PlanasNo ratings yet
- Alum Rosin SizingDocument9 pagesAlum Rosin SizingAnkit JainNo ratings yet
- Routes of Medication AdministrationDocument2 pagesRoutes of Medication AdministrationTracy100% (6)
- The Singapore Engineer - September 2018 IssueDocument27 pagesThe Singapore Engineer - September 2018 Issuekrpt0tytNo ratings yet