Professional Documents
Culture Documents
Acidosis Alkalosis
Uploaded by
Arnaldo Manggala Rahardja0 ratings0% found this document useful (0 votes)
63 views4 pagesDifferintiate acidosis and alkalosis
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentDifferintiate acidosis and alkalosis
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
63 views4 pagesAcidosis Alkalosis
Uploaded by
Arnaldo Manggala RahardjaDifferintiate acidosis and alkalosis
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 4
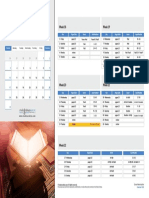
ACIDOSIS ALKALOSIS
The simple mechanism to diagnose whose having acid base disorder. The
table doesnt tell us yet about chronic and acute respiratory acid base
disorder
As you can see in the figure we have a significant elevation or decrease in
acute respiratory acid base disorder. This can be explain as follow :
Respiratory Alkalosis
Respiratory alkalosis is caused by hyperventilation, which results in
excessive loss of CO2. Hyperventilation can be caused by direct
stimulation of the medullary respiratory center, by hypoxemia (which
stimulates peripheral chemoreceptors), or by mechanical ventilation. The
arterial blood profile seen in respiratory alkalosis is
The following sequence of events occurs in the generation of respiratory
alkalosis to produce this blood profile :
1. Loss of CO2. Hyperventilation causes an excessive loss of CO2 and
a decrease in PCO2. The decreased PCO2 is the primary
disturbance in respiratory alkalosis and, as predicted by the
Henderson-Hasselbalch equation, causes an increase in pH (pH =
6.1 + log HCO3/CO2). The decreased PCO2, by mass action, also
causes a decreased concentration of HCO3.
2. Buffering. Buffering occurs exclusively in ICF, particularly in red
blood cells. In this case, CO2 leaves the cells and intracellular pH
increases.
3. Respiratory compensation. As with respiratory acidosis, there is
no respiratory compensation for respiratory alkalosis because
respiration is the cause of the disorder.
4. Renal compensation. Renal compensation for respiratory alkalosis
consists of decreased excretion of H+ as titratable acid and NH4+ and
decreased synthesis and reabsorption of new HCO3.
Decreased reabsorption of HCO3 decreases the HCO3
concentration even further than did the effect of mass action alone.
The Henderson-Hasselbalch equation can be used to understand
why the decreased HCO3 concentration is a compensatory
response:
This renal compensation differentiate acute and chronic
respiratory alkalosis :
In acute respiratory alkalosis, renal compensation has not yet
occurred and pH is quite high (there is a decrease in the denominator
of the Henderson- Hasselbalch equation but little decrease in the
numerator). In chronic respiratory alkalosis, renal compensation is
occurring, which further decreases the blood HCO3 concentration
and tends to normalize both the ratio of HCO3/CO2 and the pH. The
difference between acute and chronic respiratory alkalosis lies in renal
compensation. Again, on the basis of the absence or presence of renal
compensation, the renal rules give different calculations for the expected
change in HCO3 concentration in acute and chronic respiratory alkalosis.
Therefore chronic respiratory alkalosis often misintrepretate as a
normal condition which less elevate pH, less decrease in HCO3- ,
but significance decrease in PCO2.
Respiratory Acidosis
Respiratory acidosis is caused by hypoventilation, which results in
retention of CO2. The retention of CO2 can be caused by inhibition of the
medullary respiratory center, paralysis of respiratory muscles, airway
obstruction, or failure to exchange CO2 between pulmonary capillary blood
and alveolar gas. The arterial blood profile seen in respiratory acidosis is
The following sequence of events occurs in the generation of respiratory
acidosis to produce this blood profile:
1. Retention of CO2. Hypoventilation causes retention of CO2 and an
increase in PCO2. The increased PCO2 is the primary disturbance in
respiratory acidosis and, as predicted by the Henderson-Hasselbalch
equation, causes a decrease in pH (pH = 6.1 + log HCO3/CO2). The
increased PCO2, by mass action, also causes an increased
concentration of HCO3.
2. Buffering. Buffering of the excess CO2 occurs exclusively in ICF,
especially in red blood cells. To utilize these intracellular buffers,
CO2 diffuses across the cell membranes. Within the cells, CO2 is
converted to H+ and HCO3 and the H+ is buffered by intracellular
proteins (e.g., hemoglobin) and by organic phosphates.
3. Respiratory compensation. There is no respiratory compensation
for respiratory acidosis, since respiration is the cause of this
disorder.
4. Renal compensation. Renal compensation for respiratory acidosis
consists of increased H+ excretion as titratable acid and NH4+ and
increased synthesis and reabsorption of new HCO3.
Reabsorption of new HCO3 increases the HCO3 concentration even
further than the effect of mass action alone. The HendersonHasselbalch equation can be used tounderstand why the increased
HCO3 concentration is a compensatory response. Thus,
In acute respiratory acidosis, renal compensation has not yet
occurred, and the pH tends to be quite low (there is an increase in
the denominator in the Henderson-Hasselbalch equation but little increase
in the numerator). On the other hand, in chronic respiratory acidosis,
renal compensation is occurring, which increases the HCO3
concentration and tends to normalize both the ratio of HCO3/CO2 and
the pH. The difference between acute and chronic respiratory acidosis lies
in the renal compensation. the absence or presence of renal
compensation, the renal rules give different calculations for the expected
change in HCO3 concentration that occurs in acute and chronic
respiratory acidosis.
Therefore chronic respiratory acidosis often misintrepretate as a
normal condition which less decrease pH, less elevate in HCO3- ,
but significance elevation in PCO2.
You might also like
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5795)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (588)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (895)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (400)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (345)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (121)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- Carraro TLB1 - UP - 4WDDocument15 pagesCarraro TLB1 - UP - 4WDИрина CML75% (8)
- Arnis A Filipino Martial ArtDocument2 pagesArnis A Filipino Martial ArtAsliah Cawasa50% (2)
- enDocument40 pagesenuser100% (2)
- CVDocument2 pagesCVStef ChefNo ratings yet
- Discussion: Lesson 1: Fitness Tests Management For Sustainable HealthDocument16 pagesDiscussion: Lesson 1: Fitness Tests Management For Sustainable Healthjunalyn huquerizaNo ratings yet
- Game Modes in OverDrive! - Mantic GamesDocument5 pagesGame Modes in OverDrive! - Mantic GameslaurentstravaNo ratings yet
- Standing Yoga AsanasDocument5 pagesStanding Yoga AsanasmangaldasNo ratings yet
- Manny 2Document2 pagesManny 2unicronicNo ratings yet
- Frister Cub 7Document51 pagesFrister Cub 7Ian Black87% (15)
- Twirly ThingDocument4 pagesTwirly ThingKris KayNo ratings yet
- 6.03 Honors AssignmentDocument2 pages6.03 Honors AssignmentMariana JordanNo ratings yet
- Test Grilă La Limba Engleză - Profilul Maiştri Militari - Filiera Directă Varianta Nr. 2 Partea I: CITITDocument9 pagesTest Grilă La Limba Engleză - Profilul Maiştri Militari - Filiera Directă Varianta Nr. 2 Partea I: CITITAndreea Grecu100% (1)
- Piper PA-34-200T Seneca II Manual PDFDocument23 pagesPiper PA-34-200T Seneca II Manual PDFMuhammad Naveed0% (1)
- MSI MS-7823 Rev 3.2 PDFDocument33 pagesMSI MS-7823 Rev 3.2 PDFHACSNo ratings yet
- L-39 Parachute Packing ManualDocument35 pagesL-39 Parachute Packing ManualAnandeep_Pannu_3388100% (1)
- Studio Arabiya - Quran Memorization Calendar 2020 6Document1 pageStudio Arabiya - Quran Memorization Calendar 2020 6DarjeelingNo ratings yet
- Actions Song For KidsDocument2 pagesActions Song For KidsNurasmah Abdul HamidNo ratings yet
- Trinity Grade 5 QuestionsDocument2 pagesTrinity Grade 5 QuestionsMari Cruz JC100% (2)
- Sinotruk Tipper 6x4Document1 pageSinotruk Tipper 6x4Hugo RodriguezNo ratings yet
- Kolkata Knight RidersDocument2 pagesKolkata Knight RidersRitu SinghNo ratings yet
- Manual de Partes Pulsar - NS200 &AS 200 - SPC Abr16 PDFDocument90 pagesManual de Partes Pulsar - NS200 &AS 200 - SPC Abr16 PDFID339101No ratings yet
- Freedom Compatibility ListDocument6 pagesFreedom Compatibility Listandermall75% (4)
- Anatomy Upper ExtremityDocument17 pagesAnatomy Upper ExtremityJill LoveNo ratings yet
- Terminals of EcmDocument10 pagesTerminals of Ecmjorge luis guevara martinezNo ratings yet
- Physical Education and Health - Week 1-20Document186 pagesPhysical Education and Health - Week 1-20Christian Paul A. AsicoNo ratings yet
- The Odyssey TestDocument2 pagesThe Odyssey TestMichelle Dela Cruz0% (1)
- Storyboards and ScenariosDocument2 pagesStoryboards and ScenariosSiddharth JaiswalNo ratings yet
- Pink PantherDocument50 pagesPink Pantherkamy800No ratings yet
- Product CATLOG THAILAND - 1595282547Document39 pagesProduct CATLOG THAILAND - 1595282547walter sueroNo ratings yet