Professional Documents
Culture Documents
Chapter 2
Uploaded by
api-3044860520 ratings0% found this document useful (0 votes)
47 views2 pagesOriginal Title
chapter 2
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
47 views2 pagesChapter 2
Uploaded by
api-304486052Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 2
Term Definition
Active site Site on surface of the enzyme to which the substrate binds
Substrate A molecule which after combining with an enzyme is converted to a
product
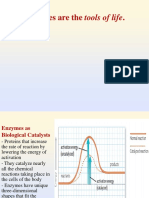
How do enzymes lower activation energy
- Enzyme binds to substrate
- Lowers activation energy
- By wearing bonds
- Making substrate more likely to react
Outline how lock and key model catalyse reactions
- It explains the ability of specific enzymes to bind to specific substrates
- Substrates fits exactly into the complementary shape to the active site of enzyme to form an
enzyme substrate complex by lowering the activation energy
- The active site can be changed by different chemicals/temperature/oH, so substrate can’t bind
Outline how induced fit model catalyse reactions
- It explains the ability of some enzymes to bind to several substrates
- Active site of enzyme binds to substrate, but they do not match up exactly
- Enzyme changes shape once bound, and this change in shape facilitates bond breaking by
reducing activation energy
- Once complete, products leave and enzyme works again
How active sites promotes enzyme-substrate specificity
- Shape of active site matches that of the substrate
- Active site can change to induce fit of substrate
Explain enzyme-substrate specificity
- Enzyme shape is specific to substrate
- Lock and key model
- Lipid and Lipase
- Has specific 3D shape essential to functioning
- Active site on enzyme binds to substrate
- Substrate and active site complementary due to structure
- Enzyme substrate complex forms
- Denaturation changes enzyme’s binding ability
Explain the effects of pH on enzyme catalysed reactions
- Enzymes has a optimum pH
- Active sites work best at this pH
- Activity decreases above and below the optimum
- By interfering with H-bonds
- Denaturing by extremes of pH so enzyme activity stops
- The formation of the enzyme-substrate complex and binding to active site
Effect to enzyme when exposed to increasing temperatures
- Rate of reaction increases when temperature increases as there is a greater proportion of
molecules with energy greater than the activation energy
- Maximum rate is achieved at optimum temperature
- Rate decreases above optimum temperature as enzymes are denatured at high temperature
- Heat causes vibration inside the enzyme which breaks bonds for maintaining the structure of
enzyme
- To form enzyme substrate, substrate should fit in active site of enzyme
- Since shape of active site has changed, substrate cannot bind to the enzyme
Explain the effect of change of pH, substrate concentration and
temperature on enzyme activity
pH:
- Enzymes have an optimum pH
- Activity increase as pH gets closer to optimum pH
- Extreme pH denatures enzymes by breaking bonds and altering structure
Substrate:
- As substrate concentration increases, activity increases
- As substrate concentration increases, the collisions between substrate and enzyme increases
- Till a plateau is reached and/or all active sites are occupied
Temperature:
- Enzymes have an optimum temperature
- Activity increases as it gets closer to optimum temperature
- High temperatures stop enzyme activity due to irreversible changes in structure by breaking
bonds
Explain the production of lactose-free milk
- Lactase added to milk
- Lactose hydrolysed into glucose and galactose
- For those who are lactose intolerant
- Increases sweetness and smooth texture
Discuss the use of lactase in the production of lactose-free milk
- Lactose is a disaccharide present in milk
- Lactase digests lactose into galactose and glucose
- Lactase produced naturally by yeast
- Biotechnology companies isolate lactase for use in food processing
- Lactase can be added to milk to reduce the level of lactose in the milk
- Immobilised enzymes may be used
- Lactose intolerant people cannot drink milk
- Galactose and glucose are sweeter than lactose
- So less sugar is need in food production
- Bacteria ferment glucose and galactose more quickly than lactose
- Galactose and glucose are more soluble so improve the texture of foods
You might also like
- Batch and Semi-batch Reactors: Practical Guides in Chemical EngineeringFrom EverandBatch and Semi-batch Reactors: Practical Guides in Chemical EngineeringNo ratings yet
- Biological Catalysts: Enzymes and MetabolismDocument2 pagesBiological Catalysts: Enzymes and MetabolismAlimm12No ratings yet
- Enzymes: Biological Catalysts and RegulationDocument31 pagesEnzymes: Biological Catalysts and RegulationdrhydrogenNo ratings yet
- 2 5 EnzymesDocument8 pages2 5 Enzymesapi-287708416No ratings yet
- Biocatalyse EnzymeDocument49 pagesBiocatalyse Enzymesyahida musthaffaNo ratings yet
- Enzymes - NotesDocument3 pagesEnzymes - Noteshiba.alkubaisyNo ratings yet
- Enzyme ChemistryDocument9 pagesEnzyme ChemistryVictor OmoloNo ratings yet
- Enzymes PDFDocument4 pagesEnzymes PDFrasha nada100% (1)
- Enzymes:: Catabolic ReactionsDocument5 pagesEnzymes:: Catabolic ReactionsSarahNo ratings yet
- STPM BIOLOGY Control in CellsDocument35 pagesSTPM BIOLOGY Control in Cellswkwhui100% (1)
- Medical Biochemistry: Enzymes: Mr. Genaro F. Alderite JR, MsermDocument58 pagesMedical Biochemistry: Enzymes: Mr. Genaro F. Alderite JR, MsermNash DeniegaNo ratings yet
- Enzymes As Biological CatalystsDocument23 pagesEnzymes As Biological CatalystsLailatul BadriyahNo ratings yet
- Flashcards - Topic 5 Enzymes - CAIE Biology IGCSEDocument25 pagesFlashcards - Topic 5 Enzymes - CAIE Biology IGCSEJane LeaNo ratings yet
- EnzymesDocument22 pagesEnzymeslovelykissNo ratings yet
- How Enzymes Speed Up Metabolic Reactions in CellsDocument23 pagesHow Enzymes Speed Up Metabolic Reactions in CellsscarlettNo ratings yet
- Enzymes:: The Nature's CatalystsDocument51 pagesEnzymes:: The Nature's Catalystskrk100% (1)
- EnzymesDocument2 pagesEnzymesArun DhirNo ratings yet
- LMRN Molecules of LifeDocument2 pagesLMRN Molecules of LifeVic AuNo ratings yet
- Enzymes Speed Up Reactions (StudentDocument5 pagesEnzymes Speed Up Reactions (StudentsushantNo ratings yet
- Classification and Factors Affecting Enzyme ActivityDocument25 pagesClassification and Factors Affecting Enzyme ActivitymiadelfiorNo ratings yet
- Biochemistry 1: - Biochemistry of Amino Acids - Biochemistry of Proteins - Portrait of Allosteric ProteinDocument55 pagesBiochemistry 1: - Biochemistry of Amino Acids - Biochemistry of Proteins - Portrait of Allosteric ProteinHiba N IkhmyesNo ratings yet
- EnzymesDocument14 pagesEnzymesCarl Agape DavisNo ratings yet
- Enzymes Key QuestionsDocument2 pagesEnzymes Key QuestionsNgọc KhuêNo ratings yet
- L8_EnzymesDocument26 pagesL8_Enzymesayaanali.voraNo ratings yet
- Biomolecules and Cells:: Mr. Derrick Banda MSC, BSCDocument69 pagesBiomolecules and Cells:: Mr. Derrick Banda MSC, BSCAmon Sangulube100% (1)
- Enzymes: Function, Structure, and Factors Affecting ActivityDocument5 pagesEnzymes: Function, Structure, and Factors Affecting ActivitySameer ButtNo ratings yet
- EnzymesDocument26 pagesEnzymeschangemakergrade8No ratings yet
- Enzyme: By. Yasmine Hadiastriani, S.SiDocument13 pagesEnzyme: By. Yasmine Hadiastriani, S.SiYasmine HadiastrianiNo ratings yet
- Dr. Kumari's Online Class on Enzyme MechanismsDocument13 pagesDr. Kumari's Online Class on Enzyme MechanismsPoonam ChauhanNo ratings yet
- 3.5 Enzymes 2Document64 pages3.5 Enzymes 2Alondra SagarioNo ratings yet
- Chapter 9Document14 pagesChapter 9katrinajen ferrerNo ratings yet
- Chapter 1 - EnzymesDocument84 pagesChapter 1 - EnzymesNorsuzianaNo ratings yet
- Enzymes: Cariño, Cordero, Magana, Samson, SantosDocument17 pagesEnzymes: Cariño, Cordero, Magana, Samson, SantosSandra SamsonNo ratings yet
- Properties of EnzymesDocument6 pagesProperties of EnzymesNida JavedNo ratings yet
- How Enzymes Work GuideDocument14 pagesHow Enzymes Work GuideX GamerNo ratings yet
- Enzymes:: Commented (1) : Im Going To Add The Notes From TheDocument5 pagesEnzymes:: Commented (1) : Im Going To Add The Notes From TheSarahNo ratings yet
- Chap 5 Bio IgcseDocument7 pagesChap 5 Bio Igcseananya.arumugarajanNo ratings yet
- C5 - EnzymesDocument6 pagesC5 - EnzymesThet Htar ZawNo ratings yet
- Enzymes: Structure, Function and Factors Affecting ActivityDocument5 pagesEnzymes: Structure, Function and Factors Affecting Activitytiyf mojaleedNo ratings yet
- Enzymes - 26 FebDocument30 pagesEnzymes - 26 FebTavimba FanetiNo ratings yet
- Topic 2.5Document15 pagesTopic 2.5hamad alwanNo ratings yet
- 12enzymes NewDocument39 pages12enzymes NewEman QawasmiNo ratings yet
- AP BIO Febraury Break With AnswersDocument12 pagesAP BIO Febraury Break With AnswersMehak BectorNo ratings yet
- Enzymes II CAPE Biology Unit 1Document24 pagesEnzymes II CAPE Biology Unit 1JayBigHarryNo ratings yet
- Human Nutrition EnzymesDocument7 pagesHuman Nutrition EnzymesMihika SpamNo ratings yet
- Summary Notes - Topic 5 Enzymes - CIE Biology IGCSEDocument2 pagesSummary Notes - Topic 5 Enzymes - CIE Biology IGCSEStudy GuyNo ratings yet
- Effect of Temperature on Enzyme ActivityDocument5 pagesEffect of Temperature on Enzyme ActivityKIARA B SAMUELNo ratings yet
- 10 - Enzymes - PPT - AutoRecoveredDocument65 pages10 - Enzymes - PPT - AutoRecoveredFaith WilliamsNo ratings yet
- Enzymes PPT BioDocument22 pagesEnzymes PPT Biovoyav37617No ratings yet
- EnzymesDocument40 pagesEnzymesVeronica HanyNo ratings yet
- Enzymes: Mode of Action and Factors Affecting RateDocument6 pagesEnzymes: Mode of Action and Factors Affecting RateNamra MazherNo ratings yet
- Enzymes and SubstratesDocument11 pagesEnzymes and Substratesapi-252152558No ratings yet
- 1.2 EnzymesDocument4 pages1.2 EnzymesMohammed RagabNo ratings yet
- Enzymes: Biological Catalysts Speed Up Chemical ReactionsDocument2 pagesEnzymes: Biological Catalysts Speed Up Chemical ReactionsKirusaanth SathiyathasanNo ratings yet
- Chapter EnzymesDocument2 pagesChapter EnzymesJaved SohawonNo ratings yet
- Enzymes Lower Activation Energy for Chemical ReactionsDocument3 pagesEnzymes Lower Activation Energy for Chemical ReactionsdetNo ratings yet
- O Level Biology Notes - EnzymesDocument3 pagesO Level Biology Notes - Enzymesdet100% (1)
- EnzymeDocument3 pagesEnzymedetNo ratings yet
- Enzymes: Definition, Characteristics, Examples and Mechanism of ActionDocument21 pagesEnzymes: Definition, Characteristics, Examples and Mechanism of ActionSaketh VNo ratings yet
- Chapter 1Document1 pageChapter 1api-304486052No ratings yet
- Chapter 1Document1 pageChapter 1api-304486052No ratings yet
- Chapter 2Document1 pageChapter 2api-304486052No ratings yet
- Chapter 2Document1 pageChapter 2api-304486052No ratings yet
- Chapter 1Document2 pagesChapter 1api-304486052No ratings yet
- Chapter 2Document1 pageChapter 2api-304486052No ratings yet
- Chapter 1Document2 pagesChapter 1api-304486052No ratings yet
- Chapter 1Document2 pagesChapter 1api-304486052No ratings yet
- Chapter 1Document2 pagesChapter 1api-304486052No ratings yet
- Chapter 2Document1 pageChapter 2api-304486052No ratings yet
- Chapter 2Document1 pageChapter 2api-304486052No ratings yet
- Chapter 2Document2 pagesChapter 2api-304486052No ratings yet
- Chapter 2Document1 pageChapter 2api-304486052No ratings yet
- Chapter 2Document2 pagesChapter 2api-304486052No ratings yet
- Chapter 3Document4 pagesChapter 3api-304486052No ratings yet
- Chapter 3Document4 pagesChapter 3api-304486052No ratings yet
- Chapter 3Document2 pagesChapter 3api-304486052No ratings yet
- Chapter 3Document4 pagesChapter 3api-304486052No ratings yet
- Chapter 3Document2 pagesChapter 3api-304486052No ratings yet
- Chapter 4Document3 pagesChapter 4api-304486052No ratings yet
- Chapter 5Document2 pagesChapter 5api-304486052No ratings yet
- Chapter 4Document2 pagesChapter 4api-304486052No ratings yet
- Chapter 5Document4 pagesChapter 5api-304486052No ratings yet
- Chapter 4Document3 pagesChapter 4api-304486052No ratings yet
- Chapter 5Document3 pagesChapter 5api-304486052No ratings yet
- Chapter 6Document2 pagesChapter 6api-304486052No ratings yet
- Chapter 4Document1 pageChapter 4api-304486052No ratings yet
- Chapter 5Document2 pagesChapter 5api-304486052No ratings yet
- Chapter 6Document6 pagesChapter 6api-304486052No ratings yet
- The Fermentation of Sugars Using YeastDocument7 pagesThe Fermentation of Sugars Using YeastJames BelciñaNo ratings yet
- Lactose Intolerance GRP 7 E-1Document39 pagesLactose Intolerance GRP 7 E-1Carl Angelo Suaybaguio100% (1)
- Applied Chemistry Activity Sheet Quarter 4 - MELC 3 Week 7-9 Organic CompoundsDocument12 pagesApplied Chemistry Activity Sheet Quarter 4 - MELC 3 Week 7-9 Organic CompoundsRACHEL GRACE SALVANTE100% (1)
- Lactose: The Hidden Culprit in Medication Intolerance?: Orthopedics September 2007Document4 pagesLactose: The Hidden Culprit in Medication Intolerance?: Orthopedics September 2007Sophia ClaireNo ratings yet
- Cell composition chapter 1 questionsDocument41 pagesCell composition chapter 1 questionsrohaizi rastamNo ratings yet
- Biology Extended Essay Madeeha Kalsekar: (I) State A Null Hypothesis That The Student Could Make For This InvestigationDocument6 pagesBiology Extended Essay Madeeha Kalsekar: (I) State A Null Hypothesis That The Student Could Make For This InvestigationMadeeha KalsekarNo ratings yet
- BWV 24A COMP Lactase - Action - CO2Document7 pagesBWV 24A COMP Lactase - Action - CO2Carla DionisioNo ratings yet
- Chapter 6: Physiology Questionbank Summary: Enzym E Source Optimum PH Substrate ProductsDocument3 pagesChapter 6: Physiology Questionbank Summary: Enzym E Source Optimum PH Substrate ProductsAshmita KumarNo ratings yet
- Study Case Lactose Intolerance Sofia MontoyaDocument4 pagesStudy Case Lactose Intolerance Sofia MontoyaMaría Sofía MontoyaNo ratings yet
- PGD CNAD 607 Meal Management and Planning Disease SheetDocument14 pagesPGD CNAD 607 Meal Management and Planning Disease SheetHasaan QaziNo ratings yet
- Cambridge International AS & A Level: BIOLOGY 9700/35Document16 pagesCambridge International AS & A Level: BIOLOGY 9700/35photopour13sNo ratings yet
- 3.1 Macronutrients - Carbohydrate PDFDocument40 pages3.1 Macronutrients - Carbohydrate PDFNur Ayu Nadhirah Bt YahyaNo ratings yet
- IAL Enzyme PP (Set 1)Document26 pagesIAL Enzyme PP (Set 1)JjrlNo ratings yet
- Lactose Properties and CharacteristicsDocument12 pagesLactose Properties and CharacteristicsQasim AkhtarNo ratings yet
- 2.5 EnzymesDocument42 pages2.5 EnzymesHelena GlanvilleNo ratings yet
- NutritionofSquirrels 1Document15 pagesNutritionofSquirrels 1Julka MadejNo ratings yet
- Structure and function of sucroseDocument73 pagesStructure and function of sucrosetess_15No ratings yet
- Dr. Ashish Kumar Singh: Senior Scientist Dairy Technology Division NDRI, KarnalDocument60 pagesDr. Ashish Kumar Singh: Senior Scientist Dairy Technology Division NDRI, KarnalshalambikaNo ratings yet
- h420 02 Biological DiversityDocument48 pagesh420 02 Biological Diversityirene9tan9ailianNo ratings yet
- Lactose IntoleranceDocument5 pagesLactose IntoleranceNader Smadi100% (1)
- Got Lactase - FillableDocument7 pagesGot Lactase - FillableElizabeth WardenNo ratings yet
- 3.2 Cell Transport PDFDocument146 pages3.2 Cell Transport PDFChryssa EconomouNo ratings yet
- Lactase, Pepsin, AmylaseDocument3 pagesLactase, Pepsin, AmylaseYume AkamaiNo ratings yet
- The Biotechnological Utilization of Cheese Whey: A ReviewDocument11 pagesThe Biotechnological Utilization of Cheese Whey: A ReviewEduar Moreno LondoñoNo ratings yet
- GCSE Biology Enzymes NotesDocument33 pagesGCSE Biology Enzymes NotesMeera ParaNo ratings yet
- Bio 201 Laboratory ReportDocument5 pagesBio 201 Laboratory Reportapi-252855115No ratings yet
- Maxilact TechBulletinDocument9 pagesMaxilact TechBulletinandres mNo ratings yet
- Aqa Bly2h QP Jan13Document16 pagesAqa Bly2h QP Jan13ApplemeisterNo ratings yet
- Water and Carbohydrates Exam Qs & MSDocument10 pagesWater and Carbohydrates Exam Qs & MSKiswal AliNo ratings yet
- Jurnal Intoleransi LaktosaDocument5 pagesJurnal Intoleransi LaktosaUpitFlowNo ratings yet
- Is That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeFrom EverandIs That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeRating: 4.5 out of 5 stars4.5/5 (3)
- Stuff Matters: Exploring the Marvelous Materials That Shape Our Man-Made WorldFrom EverandStuff Matters: Exploring the Marvelous Materials That Shape Our Man-Made WorldRating: 4 out of 5 stars4/5 (289)
- Chemistry for Breakfast: The Amazing Science of Everyday LifeFrom EverandChemistry for Breakfast: The Amazing Science of Everyday LifeRating: 4.5 out of 5 stars4.5/5 (14)
- Guidelines for Asset Integrity ManagementFrom EverandGuidelines for Asset Integrity ManagementRating: 5 out of 5 stars5/5 (1)
- Monkeys, Myths, and Molecules: Separating Fact from Fiction in the Science of Everyday LifeFrom EverandMonkeys, Myths, and Molecules: Separating Fact from Fiction in the Science of Everyday LifeRating: 4 out of 5 stars4/5 (9)
- The Disappearing Spoon: And Other True Tales of Madness, Love, and the History of the World from the Periodic Table of the ElementsFrom EverandThe Disappearing Spoon: And Other True Tales of Madness, Love, and the History of the World from the Periodic Table of the ElementsRating: 4 out of 5 stars4/5 (146)
- Science Goes Viral: Captivating Accounts of Science in Everyday LifeFrom EverandScience Goes Viral: Captivating Accounts of Science in Everyday LifeRating: 5 out of 5 stars5/5 (1)
- Coating and Drying Defects: Troubleshooting Operating ProblemsFrom EverandCoating and Drying Defects: Troubleshooting Operating ProblemsRating: 5 out of 5 stars5/5 (1)
- Organic Chemistry for Schools: Advanced Level and Senior High SchoolFrom EverandOrganic Chemistry for Schools: Advanced Level and Senior High SchoolNo ratings yet
- An Introduction to the Periodic Table of Elements : Chemistry Textbook Grade 8 | Children's Chemistry BooksFrom EverandAn Introduction to the Periodic Table of Elements : Chemistry Textbook Grade 8 | Children's Chemistry BooksRating: 5 out of 5 stars5/5 (1)
- Guidelines for Defining Process Safety Competency RequirementsFrom EverandGuidelines for Defining Process Safety Competency RequirementsRating: 3 out of 5 stars3/5 (1)
- Napoleon's Buttons: 17 Molecules That Changed HistoryFrom EverandNapoleon's Buttons: 17 Molecules That Changed HistoryRating: 4 out of 5 stars4/5 (25)
- The Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsFrom EverandThe Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsRating: 5 out of 5 stars5/5 (3)
- Essential Oil Chemistry Formulating Essential Oil Blends that Heal - Aldehyde - Ketone - Lactone: Healing with Essential OilFrom EverandEssential Oil Chemistry Formulating Essential Oil Blends that Heal - Aldehyde - Ketone - Lactone: Healing with Essential OilRating: 5 out of 5 stars5/5 (1)
- It's Elemental: The Hidden Chemistry in EverythingFrom EverandIt's Elemental: The Hidden Chemistry in EverythingRating: 4 out of 5 stars4/5 (10)
- Meltdown: Nuclear disaster and the human cost of going criticalFrom EverandMeltdown: Nuclear disaster and the human cost of going criticalRating: 5 out of 5 stars5/5 (5)
- Chemical Elements Pocket Guide: Detailed Summary of the Periodic TableFrom EverandChemical Elements Pocket Guide: Detailed Summary of the Periodic TableNo ratings yet
- Chemistry: a QuickStudy Laminated Reference GuideFrom EverandChemistry: a QuickStudy Laminated Reference GuideRating: 5 out of 5 stars5/5 (1)
- The Periodic Table: A Very Short IntroductionFrom EverandThe Periodic Table: A Very Short IntroductionRating: 4.5 out of 5 stars4.5/5 (3)
- Chemistry for Breakfast: The Amazing Science of Everyday LifeFrom EverandChemistry for Breakfast: The Amazing Science of Everyday LifeRating: 4.5 out of 5 stars4.5/5 (90)
- The Elements We Live By: How Iron Helps Us Breathe, Potassium Lets Us See, and Other Surprising Superpowers of the Periodic TableFrom EverandThe Elements We Live By: How Iron Helps Us Breathe, Potassium Lets Us See, and Other Surprising Superpowers of the Periodic TableRating: 3.5 out of 5 stars3.5/5 (22)
- Guidelines for Integrating Process Safety into Engineering ProjectsFrom EverandGuidelines for Integrating Process Safety into Engineering ProjectsNo ratings yet
- Monkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeFrom EverandMonkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeRating: 4 out of 5 stars4/5 (1)