Professional Documents
Culture Documents
The Applications of Enzymes in Industry and Medicine
Uploaded by
Mary ThomasOriginal Description:
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
The Applications of Enzymes in Industry and Medicine
Uploaded by
Mary ThomasCopyright:
Available Formats
The Applications of Enzymes in Industry and Medicine
About Enzymes
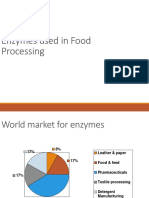
Enzymes are extracted from living organisms such as bacteria and moulds. They are biological catalysts capable of increasing efficiently the rate of a chemical reaction without using excessive energy, and remain unchanged once the reaction is complete. Minute quantities can accomplish large amounts of product at relatively low temperatures, for example approximately 30 g of pure crystalline pepsin can digest nearly 2 metric tons of egg white in a few hours. Natural enzymes are naturally present while industrial enzymes are extracted from bacteria or fungi and then added in specific quantities. There are numerous applications of enzymes in industry, for example in food, beverage, textiles and detergent processes.
Use in Baking
The wheat flour used for bread has naturally occurring enzymes that modify the starch, protein and fibre of the flour when water is added. Yeast added to the mixture also has enzymes, which ferment the maltose over time, to make the dough rise. In bakeries, the quality of the wheat flour varies, as a consequence of natural variation, time of year or inconsistencies in milling. To improve consistency and efficiency, extra enzymes (like xylanase, ?-amylase, protease, glucose oxidase and lipase) are used as supplements, enabling better handling of the dough and the control of certain characteristics in the finished bread. The interaction between different enzymes is complex and the wrong mixture of enzymes can be detrimental, for example, too much enzyme usually results in the failure of the bread to rise properly. The use of enzymes in bread making illustrates their value in quality control and efficiency of production.
Use in Alcohol
In the alcohol industry, fermentation depends on the action of enzymes synthesised by the yeasts and bacteria used in the production process. Beer brewing essentially involves the yeast action on barley, maize, sorghum, hops or rice. The yeast cells convert simple sugars into alcohol and carbon dioxide. However most sugar present is in the complex polysaccharide form such as starch and cannot readily be used. So these nutrients are "released" by malting in which enzymes are released, degrading starch and protein to simple reducing sugars and amino acids. The traditional malting process is an expensive inefficient way of manufacturing enzymes. So nowadays industrial enzymes such as amylases, glucanases and proteases are added to unmalted barley to produce the same products that malting would produce by more controlled means. Use of enzymes in the beverage industry allow it to be more economic and have consistent quality.
Use in Fruit Juices
Enzymes are used in the processing of fruit juices to maximize the production of clear or cloudy juice. Nearly all fruits contain pectin. The presence of soluble pectin in squeezed juice causes cloudiness. The addition of pectin degrading enzymes (pectin methyl esterase, polygalacturonase and pectin lyase) at the pressing stage increases the amount of juice produced and can reduce cloudiness. The desired flavour and colour of citrus juices especially orange depends on the insoluble, cloudy materials of the pressed juice. The pectin component is manipulated requiring a balance between pectin methyl esterase, to promote cloudiness by increasing the pectin/calcium complex formation and polygalacturonase, to break cloudiness by depolymerisation of the pectin.
The application of enzymes in these processes is cosmetic.
Use in Washing Powders
Principally protease digests on organic stains such as grass, blood, egg and human sweat and lipases are effective on stains resulting from fatty products and amylases are effective on removing starchy food deposits. Some powders contain cellulase to brighten colours and soften fabrics. Protease and amylase are also effective in dishwasher detergents, to remove food particles. These detergents are environmentally friendly with fewer bleaching agents and phosphates, allowing the enzymes to do more work and have beneficial effects on public and environmental health.
Use in the Textile Industry
Enzymes are used in the leather and the textile industries in finishing processes. Proteases help in the de-hairing of the animal hides and lipases are used for de-greasing. The correct application of a cellulase enzyme can give a smoother, glossier brighter fabric to cellulose fibres like cotton. This technique is known as bio-polishing. In the denim industry, cloth was traditionally stonewashed with pumice stones to fade the fabric. A small application of cellulase minimises damage to the garments and also to machinery. This technique is known as bio-stoning and can ensure greater fading without high abrasive damage to fabric and accessories (buttons, rivets). The use of enzymes in this area of industry illustrates their valuable technological contribution. Enzymes are also used in Contact Lense solution and in Pet Toothpaste.
Use in Medicine
Most genetic diseases are a result of a particular enzyme deficiency. Similarly certain bacteria are more pathogenic because of an enzyme activity they have. Uses of enzymes in medicine include: Analytical tests: Diabetics use strips of paper impregnated with glucose oxidase to monitor their blood sugar. The presence of enzymes where they should not be present can also help to diagnose disease. For example when the liver is diseased or damaged, enzymes leak into the bloodstream. Testing the blood for these enzymes can confirm liver damage. Therapeutic enzymes: Enzymes are sometimes used as medicines to replace enzyme deficiencies in patients like is the use of blood clotting factors to treat haemoplilia, or the opposite where proteases are used to degrade fibrin; to prevent the formation of dangerous blood clots. Nuclease is a possible therapy for cystic fibrosis, but it is not clear how commercialized and therapeutically successful this has been. Proteases are used to clean wounds and therefore accelerate the healing process. Drug manufacture: The chemical synthesis of complex drugs is often difficult and companies turn to enzymes to perform chemical conversions In a semi-therapeutic way; Enzymes are used to aid digestion, to supplement the natural amylase, lipase and protease produced by the pancreas. People with lactose intolerance lose the enzyme lactase. Lactase supplements help to avoid stomach upsets for these people.
Enzymes for Starch
In the early 19th century, it was discovered that by boiling starch with acid it could be converted
into a sweettasting substance, which consisted mainly of glucose. Since then acids have been used widely for breaking down starch into glucose. This technique does have a number of drawbacks. The DE (dextrose equivalent) value is used as an indication of the degree of hydrolysis of syrup. The DE value of starch is zero and that of dextrose is 100. In the last 25 years, as new enzymes are available, starch hydrolysis technology has move away from acids to enzymes. Enzymatic starch conversion, depending on the enzymes used, syrups with different compositions and physical properties of starch. There are three basic steps in enzymatic starch conversion: liquefaction, saccharification and isomerisation.
Starch liquefaction
Firstly, there is a liquefaction process. A starch suspension containing 30-40% dry matter is first gelatinised and liquefied. By using heat-stable bacterial alpha amylase, 'maltodextrin' is obtained which contains mainly different oligosaccharides and dextrins. Maltodextrins are only slightly sweet and they usually undergo further conversion. In most starch conversion plants, starch liquefaction takes place in a jet-cooking process. The heat stable alpha amylase is added to the starch slurry after pH adjustment, and the slurry is pumped through a jet cooker. Live steam is injected here to raise the temperature to 105?C, and the slurry is then passed through a series of holding tubes for 5-7 minutes, which is necessary to gelatinise the starch fully. Then the temperature of the partially liquefied starch is reduced to 90-100?C by flashing, and the enzyme is allowed to react further at this temperature for 1-2 hours until the required DE (Dextrose Equivalent) is obtained.
Starch Saccharification
Saccharification is the second step in the process. Depending of the desired end product, a glucoamylase or a fungal alpha amylase is used further break down the Maltodextrins. The glucoamylase can hydrolyse starch completely to glucose along with, a little maltose and isomaltose. A pullulanase is a de-branching enzyme that can also be used to aid saccharification. Fungal alpha amylases can also be added in order to produce syrups with a higher maltose content, which means high fermentability and a relatively high degree of sweetness.
Isomerisation
Further going one step ahead, a proportion of the glucose can be isomerised into fructose, which is about twice as sweet as glucose. An immobilized glucose isomerase is used. Maps offers a range of amylases and glucoamylase for starch conversion depending on the desired end product.
Palkolase HT Palkolase LT
Heat-stable alpha amylase for starch liquefaction Alpha amylase for starch liquefaction
Palkodex Palkoamylo
Glucoamylase for starch saccharification Fungal alpha amylase for starch saccharification
Enzymes for Animal Feed
Animal feed is composed of plant material, cereals and vegetable proteins, which cannot be fully digested and utilised by animals. However, feed utilization and digestion can often be increased by the addition of external enzymes to the feed. Many cereals have a proportion of their energy in the form of nonstarch polysaccharides (NSPs), more commonly known as fibre. Enzymes are to break down these NSPs, which lead in increase of metabolisable energy and protein utilisation. In some cereals, a large part of the NSP is soluble and causes high viscosity in the small intestine of a monogastric animal. As a result, digestion becomes impaired. Selected microbial enzymes can partially degrade this NSP, lowering viscosity in the intestine and improving feed utilisation. Many vegetable protein sources, such as soybean meal, also contain NSP. The addition of selected microbial enzymes can be used to break down the NSP and make it available to the animal. Just as with cereals, the metabolisable energy and protein utilisation for vegetable protein sources can be improved by using the correct combination of amylases and proteases. In almost all plant material used for animal feed, a large part of the mineral phosphorus is bound in the form of phytic acid, which cannot be degraded by monogastric animals. Phytase liberates part of the bound phosphorus and makes it possible to reduce the phosphorus content of the feed by 25-30%. The R & D center at Maps has successful produced phytase, and soon shall be commercially launched.
Enzyme for poultry feed
The main cost in the raising of poultry is the feed, which is mainly cereal-based. Maize (corn) has a low content of soluble NSPs and is considered to be an ideal cereal. Other cereals contain higher amounts of NSPs that normally impair feed utilisation. It is possible to partially degrading these NSPs with selected enzymes acting on specific raw materials. For example, when using a standard dose of the Maps xylanase enzymes for poultry feed, the metabolisable energy value for wheat increases. Some cereals are also avoided in poultry diets due to the adverse effects caused by NSP. Barley is a prime example because inclusion of more than 10% barley in broiler diets gives rise to wet and sticky droppings as well as reduced growth rates. This is due to beta glucan, a soluble, high viscosity NSP located in the cell walls of the barley grain. By adding microbial enzymes to the barley based feed, the NSP could be degraded giving an improvement in droppings, better feed utilisation and faster growth rate. Maps offers a number of enzymes, each with its own special properties. These can be used either alone or in combination in order to obtain faster growth of the animal; better feed utilisation (feed conversion ratio); and more uniform production. Palkofeed AP Mixture of alpha amylase and protease
Palkofeed APG Palkofeed PHY Palkofeed Multizyme Palkofeed XCG
Mixture of alpha amylase, protease and beta glucanase Phytase Mixture of various enzymes Mixture of xylanase, cellulase and beta glucanase
Enzyme kinetics
Dihydrofolate reductase from E. coli with its two substrates, dihydrofolate (right) and NADPH (left), bound in the active site. The protein is shown as a ribbon diagram, with alpha helices in red, beta sheets in yellow and loops in blue. Generated from 7DFR. Enzyme kinetics is the study of the chemical reactions that are catalysed by enzymes. In enzyme kinetics the reaction rate is measured and the effects of varying the conditions of the reaction investigated. Studying an enzyme's kinetics in this way can reveal the catalytic mechanism of this enzyme, its role in metabolism, how its activity is controlled, and how a drug or a poison might inhibit the enzyme. Enzymes are usually protein molecules that manipulate other molecules the enzymes' substrates. These target molecules bind to an enzyme's active site and are transformed into products through a series of steps known as the enzymatic mechanism. These mechanisms can be divided into singlesubstrate and multiple-substrate mechanisms. Kinetic studies on enzymes that only bind one substrate, such as triosephosphate isomerase, aim to measure the affinity with which the enzyme binds this substrate and the turnover rate. When enzymes bind multiple substrates, such as dihydrofolate reductase (shown right), enzyme kinetics can also show the sequence in which these substrates bind and the sequence in which products are released. An example of enzymes that bind a single substrate and release multiple products are proteases, which cleave one protein substrate into two polypeptide products. Others join two
substrates together, such as DNA polymerase linking a nucleotide to DNA. Although these mechanisms are often a complex series of steps, there is typically one rate-determining step that determines the overall kinetics. This rate-determining step may be a chemical reaction or a conformational change of the enzyme or substrates, such as those involved in the release of product(s) from the enzyme. Knowledge of the enzyme's structure is helpful in interpreting kinetic data. For example, the structure can suggest how substrates and products bind during catalysis; what changes occur during the reaction; and even the role of particular amino acid residues in the mechanism. Some enzymes change shape significantly during the mechanism; in such cases, it is helpful to determine the enzyme structure with and without bound substrate analogs that do not undergo the enzymatic reaction. Not all biological catalysts are protein enzymes; RNA-based catalysts such as ribozymes and ribosomes are essential to many cellular functions, such as RNA splicing and translation. The main difference between ribozymes and enzymes is that RNA catalysts are composed of nucleotides, whereas enzymes are composed of amino acids. Ribozymes also perform a more limited set of reactions, although their reaction mechanisms and kinetics can be analysed and classified by the same methods.
You might also like
- (Jacobi Carl) Jungle Wires (BookFi)Document8 pages(Jacobi Carl) Jungle Wires (BookFi)Em RaNo ratings yet
- Vanish Magic Magazine Paul Romhany Edition 26Document238 pagesVanish Magic Magazine Paul Romhany Edition 26Marcelo Jorge Abel rossi100% (1)
- Best Chicken Biryani RecipeDocument8 pagesBest Chicken Biryani RecipeAbhinavNo ratings yet
- Food Analysis ManualDocument70 pagesFood Analysis ManualRishad KalappurakkalNo ratings yet
- Tax2 Meeting 2 - 17novemberDocument108 pagesTax2 Meeting 2 - 17novemberBobby Olavides SebastianNo ratings yet
- Sub Zero Operations ManualDocument57 pagesSub Zero Operations Manualpascal rosasNo ratings yet
- CHBH13 - Laboratory Manual 4Document8 pagesCHBH13 - Laboratory Manual 4Ysa DienteNo ratings yet
- Microbial Enzymes Improve Foods BeveragesDocument75 pagesMicrobial Enzymes Improve Foods BeveragesIna Ardan50% (2)
- Chapter 5 Microbial BiotechnologyDocument75 pagesChapter 5 Microbial BiotechnologymichaelNo ratings yet
- Applied and Industrial MicrobiologyDocument57 pagesApplied and Industrial MicrobiologyWinstonSwaby100% (1)
- Gail Faulkner - Slip Knot EllDocument138 pagesGail Faulkner - Slip Knot EllRally Mateeva80% (5)
- Enzymatic Starch HydrolysisDocument107 pagesEnzymatic Starch HydrolysisManuel Rojas100% (1)
- Form 4: Uses of Enzymes in Daily Life and IndustryDocument1 pageForm 4: Uses of Enzymes in Daily Life and IndustrySiva Raj100% (1)
- Industrial Uses of Enzymes: Dr. Reda Mahgoub MohamedDocument23 pagesIndustrial Uses of Enzymes: Dr. Reda Mahgoub MohamedIslam ImadNo ratings yet
- Ensymm Invert Sugar Production Abstract-1Document10 pagesEnsymm Invert Sugar Production Abstract-1swa bumNo ratings yet
- Application of Enzymes in Industry: by Manisha A. DhotreDocument16 pagesApplication of Enzymes in Industry: by Manisha A. DhotreBio ChimieNo ratings yet
- Chapter 5 Microbial BiotechnologyDocument75 pagesChapter 5 Microbial BiotechnologySyamala Natarajan100% (1)
- Industrial Enzymes Seminar SummaryDocument13 pagesIndustrial Enzymes Seminar SummaryRamu RamuNo ratings yet
- Enzymes in Bread Baking: What They Are, How They Work & Solutions To TryDocument20 pagesEnzymes in Bread Baking: What They Are, How They Work & Solutions To TryTrinh Lam100% (1)
- Assignment On Enzymes Used As Food AdditivesDocument11 pagesAssignment On Enzymes Used As Food AdditivesRupiya ChakmaNo ratings yet
- ArticleDocument11 pagesArticlesukiwaniNo ratings yet
- Application of Enzymes in Food IndustriesDocument16 pagesApplication of Enzymes in Food IndustriesjawwadNo ratings yet
- Folio Biology: Name: Abd Majid Bin MaarofDocument14 pagesFolio Biology: Name: Abd Majid Bin MaarofZeti Ahtar Abdullah SaparNo ratings yet
- Enzyme in Beverage IndustryDocument3 pagesEnzyme in Beverage IndustryFong Cai YingNo ratings yet
- Enzymes in Food ProcessingDocument39 pagesEnzymes in Food ProcessingKarthikShivaramNo ratings yet
- Fermentation TBGFinalDocument4 pagesFermentation TBGFinalPravar TrivediNo ratings yet
- Applied Enzyme CatalystsDocument64 pagesApplied Enzyme CatalystsHugo WizenbergNo ratings yet
- Enzymes As PreservativeDocument21 pagesEnzymes As PreservativejoseNo ratings yet
- Commercial Applications of EnzymesDocument3 pagesCommercial Applications of EnzymesJudy Ann BoseNo ratings yet
- Lipase hydrolyzes triglycerides into free fatty acids- Free fatty acids are further metabolized by microbes- Final products contain less fat and caloriesDocument58 pagesLipase hydrolyzes triglycerides into free fatty acids- Free fatty acids are further metabolized by microbes- Final products contain less fat and caloriesQuoc KhanhNo ratings yet
- Kuliah 2 EnzymetechnologyDocument23 pagesKuliah 2 EnzymetechnologyrhabarberrhabarberNo ratings yet
- AmylaseDocument13 pagesAmylaseDr. Kalavati PrajapatiNo ratings yet
- L12 Enzymes in Food ProcessingDocument38 pagesL12 Enzymes in Food ProcessingGirupha NishanthNo ratings yet
- Application of Enzymes in BioprocessDocument5 pagesApplication of Enzymes in BioprocessPriyank LashkariNo ratings yet
- Some Important Enzymes and Their Industrial ImportanceDocument13 pagesSome Important Enzymes and Their Industrial ImportanceBOMMIDI JAHNAVI (RA2132001010057)No ratings yet
- Folio Biology EnzymeDocument10 pagesFolio Biology EnzymeIzZati YazidNo ratings yet
- Enzymes in Action Textbook PagesDocument4 pagesEnzymes in Action Textbook Pagesjpop_doverbroecksNo ratings yet
- Applications of EnzDocument11 pagesApplications of EnzALI ASHRAFNo ratings yet
- Applications of EnzymesDocument11 pagesApplications of EnzymesNadish Namish BhradwajNo ratings yet
- Midterm Enzyme Fermentation 1Document20 pagesMidterm Enzyme Fermentation 1Thuỳ TrangNo ratings yet
- Application of Enzymes in Food IndustriesDocument38 pagesApplication of Enzymes in Food IndustriesRoshan JainNo ratings yet
- Invertase Enzyme Preparation 100 MLPDHNPDocument3 pagesInvertase Enzyme Preparation 100 MLPDHNPrelishtv35No ratings yet
- Enzyme Biotechnology in Everyday LifeDocument9 pagesEnzyme Biotechnology in Everyday LifeIrwani AzizNo ratings yet
- Enzymes Used in Food Industry: Roll No. 3 M. SC Part 1Document34 pagesEnzymes Used in Food Industry: Roll No. 3 M. SC Part 1bin ngaNo ratings yet
- Molasses Types and Its Utilization1Document16 pagesMolasses Types and Its Utilization1Nur AprilianiNo ratings yet
- Enzymes in Brewing1Document9 pagesEnzymes in Brewing1zigobasNo ratings yet
- InTech-Gel Electrophoresis For Investigating Enzymes With Biotechnological ApplicationDocument15 pagesInTech-Gel Electrophoresis For Investigating Enzymes With Biotechnological Applicationchavi1310No ratings yet
- Enzymes Used in Food IndustriesDocument38 pagesEnzymes Used in Food Industriesanon_183990983No ratings yet
- Bio-Chemical Engineering: CHE-422 Date: 13/03/2018Document53 pagesBio-Chemical Engineering: CHE-422 Date: 13/03/2018Atif MehfoozNo ratings yet
- What Is Catalytic Action of Enzyme?Document4 pagesWhat Is Catalytic Action of Enzyme?Temesgen MuletaNo ratings yet
- 231lecture18,19 UpdateDocument19 pages231lecture18,19 UpdateRana AbdullahNo ratings yet
- Biotechnology and Genetic EngineeringDocument22 pagesBiotechnology and Genetic EngineeringitsmebyeNo ratings yet
- Gusti Raka Divananda 1206292093Document2 pagesGusti Raka Divananda 1206292093robotrobot13No ratings yet
- The Industrial Production of EnzymesDocument4 pagesThe Industrial Production of EnzymesAnh Thư NguyễnNo ratings yet
- Food TechDocument41 pagesFood TechMD Ishteyaque AhmadNo ratings yet
- Kinetic Modeling of Simultaneous Saccharification and Fermentation of Corn Starch For Ethanol ProductionDocument10 pagesKinetic Modeling of Simultaneous Saccharification and Fermentation of Corn Starch For Ethanol ProductionSeba GelsuminoNo ratings yet
- Food and Beverage BiotechnologyDocument10 pagesFood and Beverage BiotechnologyHayderNo ratings yet
- EnzymesDocument10 pagesEnzymesAlok KumarNo ratings yet
- StarchDocument12 pagesStarchNahusenay TewodrosNo ratings yet
- Industrial Applications of Enzymes in Dairy Industry: Archa DaveDocument21 pagesIndustrial Applications of Enzymes in Dairy Industry: Archa DaveTín PhạmNo ratings yet
- Polarimeter Invert SugarDocument3 pagesPolarimeter Invert SugarMeranaid LogisticNo ratings yet
- ENZYMESDocument2 pagesENZYMESLennon BarrosNo ratings yet
- Enzymes: Proteins That Speed Up MetabolismDocument13 pagesEnzymes: Proteins That Speed Up MetabolismTemesgen MuletaNo ratings yet
- Enzymes Used in Food ProcessingDocument14 pagesEnzymes Used in Food Processingthings strangerNo ratings yet
- AmylaseDocument7 pagesAmylasesoumya vermaNo ratings yet
- Bartending Ncii Reviewe1Document3 pagesBartending Ncii Reviewe1Mark Bryan CervantesNo ratings yet
- PESTLE and Porter's Five Forces Analysis of the Microbrewery IndustryDocument5 pagesPESTLE and Porter's Five Forces Analysis of the Microbrewery IndustryYayaakshi ShokeenNo ratings yet
- Science4 - 1st QTR - Mod 3 - MixDocument42 pagesScience4 - 1st QTR - Mod 3 - MixJAYDEN PAULO CASTILLONo ratings yet
- Historical/Significant Sites in Azumini, Ndoki: UminiDocument16 pagesHistorical/Significant Sites in Azumini, Ndoki: UminiBright SamuelNo ratings yet
- Daniel Fast Flat BreadsDocument2 pagesDaniel Fast Flat Breadsphomolebonolo100% (1)
- Caso Starbucks PDFDocument2 pagesCaso Starbucks PDFmuazNo ratings yet
- Vorwerk CARPET CAREDocument5 pagesVorwerk CARPET CAREDan George IIINo ratings yet
- 2019 Maryland Hop Growers GuideDocument32 pages2019 Maryland Hop Growers GuideFlying Dog Brewery80% (5)
- Goof Proof GrammarDocument140 pagesGoof Proof GrammarJosué Maximin ANDÉNo ratings yet
- Resep MAKANANDocument3 pagesResep MAKANANLKP Amerta BaktiNo ratings yet
- Linguistics and Archaeology in The Americas. The Historization of Language and SocietyDocument40 pagesLinguistics and Archaeology in The Americas. The Historization of Language and SocietyDebora MinervaNo ratings yet
- Management StructureDocument7 pagesManagement StructureHong MinhNo ratings yet
- TLE Macaballug Cookery 10Document16 pagesTLE Macaballug Cookery 10Jake MacaballugNo ratings yet
- Bamberg Rauch Liquid Malt Extract Specification (1) - WeyermannDocument2 pagesBamberg Rauch Liquid Malt Extract Specification (1) - WeyermannCarmineD'AnielloNo ratings yet
- Biotechnology Exam CUTTING EDGE UNIT 1-2-3 Starter Midterm (Autoguardado)Document4 pagesBiotechnology Exam CUTTING EDGE UNIT 1-2-3 Starter Midterm (Autoguardado)Marilu Plasencia RubioNo ratings yet
- Past-Perfect-Past-Tense - My HomeworkDocument1 pagePast-Perfect-Past-Tense - My HomeworkNewspaper Translation with Mr. HongNo ratings yet
- Phys347 Exam 3Document6 pagesPhys347 Exam 3Araq InfoNo ratings yet
- SongsDocument43 pagesSongsapi-246339394No ratings yet
- Poland HRI Sector Offers Growth for U.S. Food ExportersDocument13 pagesPoland HRI Sector Offers Growth for U.S. Food ExportersCrueLargoNo ratings yet
- Preparation of Soymilk Using Different Methods: Journal of Food and Nutrition SciencesDocument7 pagesPreparation of Soymilk Using Different Methods: Journal of Food and Nutrition SciencesTaha RahmanNo ratings yet
- Advantium 120 Cooking GuideDocument4 pagesAdvantium 120 Cooking GuideqwertypolakNo ratings yet
- U.S. Dairy Ingredients in Yogurt and Yogurt BeveragesDocument20 pagesU.S. Dairy Ingredients in Yogurt and Yogurt BeveragesNguyễn Tiến DũngNo ratings yet
- F&B NC 11 CBLM Providing Link Between Kitchen & Service AreasDocument27 pagesF&B NC 11 CBLM Providing Link Between Kitchen & Service AreasCarlos Baul David0% (1)
- Tempting Season's Greetings - 2010Document1 pageTempting Season's Greetings - 2010JurgenJanssensNo ratings yet