Professional Documents
Culture Documents
Final Exam Review - 2011
Uploaded by
Elissa BakerOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Final Exam Review - 2011
Uploaded by
Elissa BakerCopyright:
Available Formats
Chem 14D Final Exam Review Packet P.
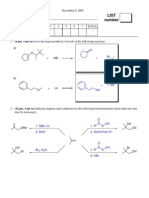
The final exam is cumulative, but with an emphasis on the more recent material (since exam 2). In this review packet, any questions that deal with Chapters 19/20 are shown in red font since there was not a separate problem set on these topics. 1. Draw the major product for each of the following reactions. Where applicable, answers without relative stereochemistry will be given zero credit. a.
Me Et H2, Pd/C EtOH
b.
1. O3, CH2Cl2 2. DMS
c.
CO2Me + CO2Me heat
d.
HgSO4 H2SO4, H2O
e.
O H+ MeOH (excess)
f.
O OMe H+ EtOH
g.
O OMe MeMgBr (2 equiv)
h.
Br2 FeBr3
Garg, Spring 2011
Chem 14D Final Exam Review Packet P.2
2. Provide the missing reagents for the transformations shown below. a.
OH OMe
b.
Br
c.
O OH
d.
OH
e.
O N Me
f.
O
g.
O O Me
h.
O OH OH
i.
O OH O Cl
j.
NH2 NO2
Garg, Spring 2011
Chem 14D Final Exam Review Packet P.3
3. Provide the missing reagents, products, and/or intermediates in the schemes below. a.
?
Br
?
? ?
EtNH2 !
SOCl2
?
O OH
O O
? ?
MeOH H+ O N
b.
O Cl AlCl3
NaBH4 EtOH
PBr3 CH2Cl2
O Zn(Hg) HCl Cl
1. Mg, Et2O 2. CO2 3. H+ work-up
CO2H
Garg, Spring 2011
Chem 14D Final Exam Review Packet P.4
4. Provide detailed arrow-pushing mechanisms for the following transformations. a.
Cl O MeO O AlCl3 MeO O O
b.
HNO3 H2SO4 NO2
c.
O OH EtOH H+, Heat O OEt
d.
O O H O NaOH, H2O Heat H
e.
O 1. LDA 2. Br O
f.
OH NaOH H2O O
Garg, Spring 2011
Chem 14D Final Exam Review Packet P.5
5. Provide syntheses of the following target molecules. You may use common reagents, solvents, fragments of 2-carbons or less (these fragments may contain atoms other than just carbon, including hydrogen, oxygen, MgBr, and Li), and any of the compounds shown in the Chem 14D Toolbox. It is not necessary to show mechanisms or a retrosynthetic analysis in your solution.
Chem 14D Toolbox
O Cl O H O H O Cl
a.
O OMe
OH
b.
O H
Br
c.
Me N
d.
MeO O O
e.
O OH
(ibuprofen)
Garg, Spring 2011
You might also like
- CHEM 14D/Garg Spring 2015 Problem Set #6Document10 pagesCHEM 14D/Garg Spring 2015 Problem Set #6Hugh BangNo ratings yet
- Practice Makes Perfect in Chemistry: Oxidation-ReductionFrom EverandPractice Makes Perfect in Chemistry: Oxidation-ReductionRating: 5 out of 5 stars5/5 (1)
- Do Not Open This Test Until Everyone Has OneDocument12 pagesDo Not Open This Test Until Everyone Has OneChemist MeNo ratings yet
- Practice Makes Perfect in Chemistry: Compounds, Reactions and Moles with AnswersFrom EverandPractice Makes Perfect in Chemistry: Compounds, Reactions and Moles with AnswersRating: 3 out of 5 stars3/5 (2)
- 231 F 2010 Practice MT4 - Pp1to12Document12 pages231 F 2010 Practice MT4 - Pp1to12Chemist MeNo ratings yet
- Practice Makes Perfect in Chemistry: Oxidation-Reduction with AnswersFrom EverandPractice Makes Perfect in Chemistry: Oxidation-Reduction with AnswersNo ratings yet
- Test1 342 PracticeV1Document5 pagesTest1 342 PracticeV1Camha NguyenNo ratings yet
- XXIVth International Congress of Pure and Applied Chemistry: Plenary and Main Section Lectures Presented at Hamburg, Federal Republic of Germany, 2–8 September 1973From EverandXXIVth International Congress of Pure and Applied Chemistry: Plenary and Main Section Lectures Presented at Hamburg, Federal Republic of Germany, 2–8 September 1973No ratings yet
- CHM 2210 Practice Exam 3Document8 pagesCHM 2210 Practice Exam 3Shaima MossamatNo ratings yet
- Organic Chemistry: Chem 210 Practice Exam 3BDocument13 pagesOrganic Chemistry: Chem 210 Practice Exam 3Bemmanferrer482No ratings yet
- College Organic Chemistry Semester II: Practice Questions with Detailed ExplanationsFrom EverandCollege Organic Chemistry Semester II: Practice Questions with Detailed ExplanationsNo ratings yet
- Practice 3CDocument13 pagesPractice 3CCamha NguyenNo ratings yet
- 2423 e 2Document24 pages2423 e 2Agustin KurniatiNo ratings yet
- 432georgia Spring 2018 Practice MT 2 - REVISEDDocument14 pages432georgia Spring 2018 Practice MT 2 - REVISEDChemist MeNo ratings yet
- Massachusetts Institute of Technology 5.12, Spring 2005: Problem Set #4Document9 pagesMassachusetts Institute of Technology 5.12, Spring 2005: Problem Set #4Minh TieuNo ratings yet
- CHM 1321 Assignment 5 Answers: 1) Name The Following CompoundsDocument15 pagesCHM 1321 Assignment 5 Answers: 1) Name The Following CompoundsSara Yuen100% (1)
- Orgo 2 GuideDocument25 pagesOrgo 2 GuideRukiezillaNo ratings yet
- QuestionDocument12 pagesQuestionKiranSinglaNo ratings yet
- List Number: 1 2 3 4 5 6 7 TotalDocument5 pagesList Number: 1 2 3 4 5 6 7 Totaltcrupi1No ratings yet
- CHM556 Assignment1Document4 pagesCHM556 Assignment1Saidin AhmadNo ratings yet
- 2007 ADocument4 pages2007 AAmiro MayraNo ratings yet
- Q4 06710-12-14 Qs and AsDocument13 pagesQ4 06710-12-14 Qs and AsGhadeer M HassanNo ratings yet
- M.SC 1st Sem Unit-2 2015Document6 pagesM.SC 1st Sem Unit-2 2015bgroyNo ratings yet
- Practice 3ADocument12 pagesPractice 3ACamha NguyenNo ratings yet
- Pentadienyl CationDocument7 pagesPentadienyl CationAbhishek SardaNo ratings yet
- Additional Problems Final Exam Part 2 AnswersDocument10 pagesAdditional Problems Final Exam Part 2 AnswersJohn SmithNo ratings yet
- Tutorial 6 AlcoholDocument5 pagesTutorial 6 Alcoholwan arifahNo ratings yet
- Disconnection Approach To Synthesis: Illogical Two Group Disconnections I) 1,2-Dioxygenated PatternDocument27 pagesDisconnection Approach To Synthesis: Illogical Two Group Disconnections I) 1,2-Dioxygenated PatternhinhthoiNo ratings yet
- (15 Points) Predict The Products of The Following Reactions. Show Relative Stereochemistry (Only One Stereoisomer) Where AppropriateDocument6 pages(15 Points) Predict The Products of The Following Reactions. Show Relative Stereochemistry (Only One Stereoisomer) Where AppropriateparnaNo ratings yet
- Organic Chemistry Additional Problems Final Exam Part2Document6 pagesOrganic Chemistry Additional Problems Final Exam Part2John SmithNo ratings yet
- Chapter 8 PracticeQDocument20 pagesChapter 8 PracticeQKevin Peng0% (1)
- Gen Chem 1 Q1 M 5-6Document12 pagesGen Chem 1 Q1 M 5-6Joselito UbaldoNo ratings yet
- Practice Exam 2 1Document6 pagesPractice Exam 2 1api-197124028No ratings yet
- ExercisesDocument10 pagesExerciseslethanhdien2010No ratings yet
- Final Sim CorrDocument12 pagesFinal Sim CorrKeila BoisjolyNo ratings yet
- Chemistry HSSC-II Solution of 2nd Set Model Question PaperDocument15 pagesChemistry HSSC-II Solution of 2nd Set Model Question PaperIsha KhanNo ratings yet
- CHM 2210 Practice Exam IVDocument5 pagesCHM 2210 Practice Exam IVShaima MossamatNo ratings yet
- Iit 2011 FST2 QNS P1Document29 pagesIit 2011 FST2 QNS P1Sarvesh DubeyNo ratings yet
- Section 5.5: Standard Enthalpies of Formation: H N H N HDocument8 pagesSection 5.5: Standard Enthalpies of Formation: H N H N HhairtNo ratings yet
- Stuctural Indefication - POC ExerciseDocument22 pagesStuctural Indefication - POC ExercisemikcNo ratings yet
- Austrian National Chemistry Olympiad 1998Document21 pagesAustrian National Chemistry Olympiad 1998Popa ElenaNo ratings yet
- Exam I - sp18 KeyDocument6 pagesExam I - sp18 KeyJerry WangNo ratings yet
- Mid TermDocument12 pagesMid TermKaran PrabaNo ratings yet
- UAB Wang Recitation #1Document4 pagesUAB Wang Recitation #1OmarBilbeisiNo ratings yet
- XIX. Chemistry, High SchoolDocument25 pagesXIX. Chemistry, High SchoolJane TrinidadNo ratings yet
- Chem 114Document12 pagesChem 114lesliemarie272No ratings yet
- BaleqpracticeDocument4 pagesBaleqpracticeapi-272465522No ratings yet
- Test Problems For Chapter 18 With AnswersDocument27 pagesTest Problems For Chapter 18 With AnswersJosh Wishman50% (2)
- General Chemistry 1: CHEM 025, Section X, Fall 201X - Anthony DutoiDocument10 pagesGeneral Chemistry 1: CHEM 025, Section X, Fall 201X - Anthony DutoiJasmin GarciaNo ratings yet
- Exam Chemistry 2014-05-27 StudentsDocument6 pagesExam Chemistry 2014-05-27 StudentsAli PliegoNo ratings yet
- Organic Exam Answer.Document11 pagesOrganic Exam Answer.S JNo ratings yet
- Final Exam For Organic II 200pts (Weighted As 300) : ROH ROR RCNDocument23 pagesFinal Exam For Organic II 200pts (Weighted As 300) : ROH ROR RCNUmmi Khairani UrfaNo ratings yet
- Chem 110 Test 2 Solution (2012)Document9 pagesChem 110 Test 2 Solution (2012)Chima C. UgwuegbuNo ratings yet
- Assignment 4 Reactions of Aromatic Compounds AnswersDocument11 pagesAssignment 4 Reactions of Aromatic Compounds AnswersJonathan Yeung100% (1)
- Concerto in E Sharp Minor by Alfred ReedDocument62 pagesConcerto in E Sharp Minor by Alfred ReedMalcolm TanNo ratings yet
- KIM 101E - Week 3 - BDocument70 pagesKIM 101E - Week 3 - Baliyasin200000No ratings yet
- Answer The Following Questions For Chemistry ReviewDocument6 pagesAnswer The Following Questions For Chemistry ReviewDwyne Kaiser GallegoNo ratings yet
- Aqa Chm6w W QP Jun02Document18 pagesAqa Chm6w W QP Jun02Diksha KoossoolNo ratings yet
- Is That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeFrom EverandIs That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeRating: 5 out of 5 stars5/5 (4)
- Periodic Tales: A Cultural History of the Elements, from Arsenic to ZincFrom EverandPeriodic Tales: A Cultural History of the Elements, from Arsenic to ZincRating: 3.5 out of 5 stars3.5/5 (137)
- The Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactFrom EverandThe Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactRating: 5 out of 5 stars5/5 (5)
- Sodium Bicarbonate: Nature's Unique First Aid RemedyFrom EverandSodium Bicarbonate: Nature's Unique First Aid RemedyRating: 5 out of 5 stars5/5 (21)
- Taste: Surprising Stories and Science About Why Food Tastes GoodFrom EverandTaste: Surprising Stories and Science About Why Food Tastes GoodRating: 3 out of 5 stars3/5 (20)
- Guidelines for Chemical Process Quantitative Risk AnalysisFrom EverandGuidelines for Chemical Process Quantitative Risk AnalysisRating: 5 out of 5 stars5/5 (1)
- It's Elemental: The Hidden Chemistry in EverythingFrom EverandIt's Elemental: The Hidden Chemistry in EverythingRating: 4 out of 5 stars4/5 (10)
- Handbook of Formulating Dermal Applications: A Definitive Practical GuideFrom EverandHandbook of Formulating Dermal Applications: A Definitive Practical GuideNo ratings yet
- Guidelines for Defining Process Safety Competency RequirementsFrom EverandGuidelines for Defining Process Safety Competency RequirementsRating: 3 out of 5 stars3/5 (1)
- Chemistry for Breakfast: The Amazing Science of Everyday LifeFrom EverandChemistry for Breakfast: The Amazing Science of Everyday LifeRating: 4.5 out of 5 stars4.5/5 (90)
- Chemistry for Breakfast: The Amazing Science of Everyday LifeFrom EverandChemistry for Breakfast: The Amazing Science of Everyday LifeRating: 4.5 out of 5 stars4.5/5 (14)
- The Periodic Table: A Very Short IntroductionFrom EverandThe Periodic Table: A Very Short IntroductionRating: 4.5 out of 5 stars4.5/5 (3)
- AP® Chemistry Crash Course, For the 2020 Exam, Book + Online: Get a Higher Score in Less TimeFrom EverandAP® Chemistry Crash Course, For the 2020 Exam, Book + Online: Get a Higher Score in Less TimeRating: 5 out of 5 stars5/5 (1)
- The Disappearing Spoon: And Other True Tales of Madness, Love, and the History of the World from the Periodic Table of the ElementsFrom EverandThe Disappearing Spoon: And Other True Tales of Madness, Love, and the History of the World from the Periodic Table of the ElementsRating: 4 out of 5 stars4/5 (146)
- AP Chemistry Flashcards, Fourth Edition: Up-to-Date Review and PracticeFrom EverandAP Chemistry Flashcards, Fourth Edition: Up-to-Date Review and PracticeNo ratings yet
- Tribology: Friction and Wear of Engineering MaterialsFrom EverandTribology: Friction and Wear of Engineering MaterialsRating: 5 out of 5 stars5/5 (1)